Get your patient on Acarbose - Acarbose tablet (Acarbose)
Acarbose - Acarbose tablet prescribing information
INDICATIONS AND USAGE
Acarbose tablets, USP are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
DOSAGE AND ADMINISTRATION
There is no fixed dosage regimen for the management of diabetes mellitus with acarbose tablets or any other pharmacologic agent. Dosage of acarbose tablets must be individualized on the basis of both effectiveness and tolerance while not exceeding the maximum recommended dose of 100 mg t.i.d. Acarbose tablets should be taken three times daily at the start (with the first bite) of each main meal. Acarbose tablets should be started at a low dose, with gradual dose escalation as described below, both to reduce gastrointestinal side effects and to permit identification of the minimum dose required for adequate glycemic control of the patient. If the prescribed diet is not observed, the intestinal side effects may be intensified. If strongly distressing symptoms develop in spite of adherence to the diabetic diet prescribed, the doctor must be consulted and the dose temporarily or permanently reduced.
During treatment initiation and dose titration (see below), one-hour postprandial plasma glucose may be used to determine the therapeutic response to acarbose tablets and identify the minimum effective dose for the patient. Thereafter, glycosylated hemoglobin should be measured at intervals of approximately three months. The therapeutic goal should be to decrease both postprandial plasma glucose and glycosylated hemoglobin levels to normal or near normal by using the lowest effective dose of acarbose tablets, either as monotherapy or in combination with sulfonylureas, insulin or metformin.
Initial Dosage
The recommended starting dosage of acarbose tablets is 25 mg given orally three times daily at the start (with the first bite) of each main meal. However, some patients may benefit from more gradual dose titration to minimize gastrointestinal side effects. This may be achieved by initiating treatment at 25 mg once per day and subsequently increasing the frequency of administration to achieve 25 mg t.i.d.
Maintenance Dosage
Once a 25 mg t.i.d. dosage regimen is reached, dosage of acarbose tablets should be adjusted at 4-8 week intervals based on one-hour postprandial glucose or glycosylated hemoglobin levels, and on tolerance. The dosage can be increased from 25 mg t.i.d. to 50 mg t.i.d. Some patients may benefit from further increasing the dosage to 100 mg t.i.d. The maintenance dose ranges from 50 mg t.i.d. to 100 mg t.i.d. However, since patients with low body weight may be at increased risk for elevated serum transaminases, only patients with body weight > 60 kg should be considered for dose titration above 50 mg t.i.d. (see PRECAUTIONS ). If no further reduction in postprandial glucose or glycosylated hemoglobin levels is observed with titration to 100 mg t.i.d., consideration should be given to lowering the dose. Once an effective and tolerated dosage is established, it should be maintained.
Maximum Dosage
The maximum recommended dose for patients ≤ 60 kg is 50 mg t.i.d. The maximum recommended dose for patients > 60 kg is 100 mg t.i.d.
Patients Receiving Sulfonylureas or Insulin
Sulfonylurea agents or insulin may cause hypoglycemia. Acarbose tablets given in combination with a sulfonylurea or insulin will cause a further lowering of blood glucose and may increase the potential for hypoglycemia. If hypoglycemia occurs, appropriate adjustments in the dosage of these agents should be made.
CONTRAINDICATIONS
Acarbose tablets are contraindicated in patients with known hypersensitivity to the drug. Acarbose tablets are contraindicated in patients with diabetic ketoacidosis or cirrhosis. Acarbose tablets are also contraindicated in patients with inflammatory bowel disease, colonic ulceration, partial intestinal obstruction or in patients predisposed to intestinal obstruction. In addition, acarbose tablets are contraindicated in patients who have chronic intestinal diseases associated with marked disorders of digestion or absorption and in patients who have conditions that may deteriorate as a result of increased gas formation in the intestine.
ADVERSE REACTIONS
Digestive Tract
Gastrointestinal symptoms are the most common reactions to acarbose tablets. In U.S. placebo-controlled trials, the incidences of abdominal pain, diarrhea, and flatulence were 19%, 31%, and 74% respectively in 1255 patients treated with acarbose tablets 50-300 mg t.i.d., whereas the corresponding incidences were 9%, 12%, and 29% in 999 placebo-treated patients. In a one-year safety study, during which patients kept diaries of gastrointestinal symptoms, abdominal pain and diarrhea tended to return to pretreatment levels over time, and the frequency and intensity of flatulence tended to abate with time. The increased gastrointestinal tract symptoms in patients treated with acarbose tablets are a manifestation of the mechanism of action of acarbose and are related to the presence of undigested carbohydrate in the lower GI tract.
If the prescribed diet is not observed, the intestinal side effects may be intensified. If strongly distressing symptoms develop in spite of adherence to the diabetic diet prescribed, the doctor must be consulted and the dose temporarily or permanently reduced.
Elevated Serum Transaminase Levels
See PRECAUTIONS .
Other Abnormal Laboratory Findings
Small reductions in hematocrit occurred more often in acarbose-treated patients than in placebo-treated patients but were not associated with reductions in hemoglobin. Low serum calcium and low plasma vitamin B6 levels were associated with acarbose tablets therapy but are thought to be either spurious or of no clinical significance.
Postmarketing Adverse Event Reports
Additional adverse events reported from worldwide postmarketing experience include fulminant hepatitis with fatal outcome, hypersensitive skin reactions (for example rash, erythema, exanthema and uticaria), edema, ileus/subileus, jaundice and/or hepatitis and associated liver damage, thrombocytopenia, and pneumatosis cystoides intestinalis (see PRECAUTIONS ).
Pneumatosis Cystoides Intestinalis
There have been rare postmarketing reports of pneumatosis cystoides intestinalis associated with the use of alpha-glucosidase inhibitors, including acarbose tablets.Pneumatosis cystoides intestinalis may present with symptoms of diarrhea, mucus discharge, rectal bleeding, and constipation.Complications may include pneumoperitoneum, volvulus, intestinal obstruction, intussusception, intestinal hemorrhage, and intestinal perforation. If pneumatosis cystoides intestinalis is suspected, discontinue acarbose tablets and perform the appropriate diagnostic imaging.
Drug Interactions
Certain drugs tend to produce hyperglycemia and may lead to loss of blood glucose control. These drugs include the thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel-blocking drugs, and isoniazid. When such drugs are administered to a patient receiving acarbose tablets, the patient should be closely observed for loss of blood glucose control. When such drugs are withdrawn from patients receiving acarbose tablets in combination with sulfonylureas or insulin, patients should be observed closely for any evidence of hypoglycemia.
Patients Receiving Sulfonylureas or Insulin: Sulfonylurea agents or insulin may cause hypoglycemia. Acarbose tablets given in combination with a sulfonylurea or insulin may cause a further lowering of blood glucose and may increase the potential for hypoglycemia.
If hypoglycemia occurs, appropriate adjustments in the dosage of these agents should be made. Very rarely, individual cases of hypoglycemic shock have been reported in patients receiving acarbose tablets therapy in combination with sulfonylureas and/or insulin.
Intestinal adsorbents (for example, charcoal) and digestive enzyme preparations containing carbohydrate-splitting enzymes (for example, amylase, pancreatin) may reduce the effect of acarbose tablets and should not be taken concomitantly.
Acarbose tablets has been shown to change the bioavailability of digoxin when they are co administered, which may require digoxin dose adjustment. (See CLINICAL PHARMACOLOGY , Drug-Drug Interactions ).
DESCRIPTION
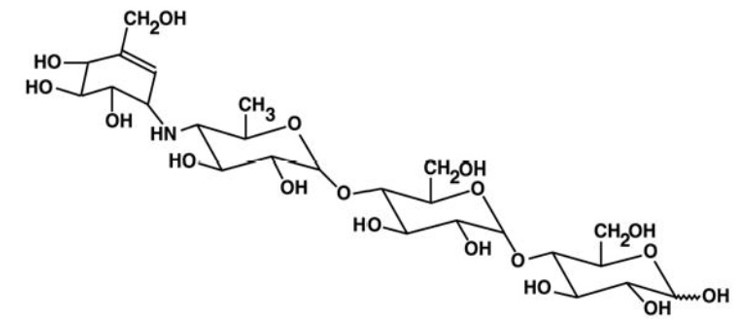
Acarbose Tablets, USP are an oral alpha-glucosidase inhibitor for use in the management of type 2 diabetes mellitus. Acarbose is an oligosaccharide which is obtained from fermentation processes of a microorganism, Actinoplanes utahensis , and is chemically known as O-4,6-dideoxy- 4-[[(1S,4R,5S,6S)-4,5,6-trihydroxy-3-(hydroxymethyl)-2-cyclohexen-1-yl] amino]- α-D-glucopyranosyl-(1 → 4)-O-α -D-glucopyranosyl-(1 → 4)-D-glucose. It is a white to off-white powder with a molecular weight of 645.6. Acarbose is soluble in water and has a pKa of 5.1. Its empirical formula is C 25 H 43 NO 18 and its chemical structure is as follows:
Acarbose Tablets, USP are available as 25 mg, 50 mg and 100 mg tablets for oral use. The inactive ingredients are starch (corn starch), microcrystalline cellulose, magnesium stearate, and colloidal silicon dioxide.
CLINICAL PHARMACOLOGY
Acarbose is a complex oligosaccharide that delays the digestion of ingested carbohydrates, thereby resulting in a smaller rise in blood glucose concentration following meals. As a consequence of plasma glucose reduction, acarbose tablets reduce levels of glycosylated hemoglobin in patients with type 2 diabetes mellitus. Systemic non-enzymatic protein glycosylation, as reflected by levels of glycosylated hemoglobin, is a function of average blood glucose concentration over time.
Mechanism of Action
In contrast to sulfonylureas, acarbose tablets do not enhance insulin secretion. The antihyperglycemic action of acarbose results from a competitive, reversible inhibition of pancreatic alpha-amylase and membrane-bound intestinal alpha-glucoside hydrolase enzymes. Pancreatic alpha-amylase hydrolyzes complex starches to oligosaccharides in the lumen of the small intestine, while the membrane-bound intestinal alpha-glucosidases hydrolyze oligosaccharides, trisaccharides, and disaccharides to glucose and other monosaccharides in the brush border of the small intestine. In diabetic patients, this enzyme inhibition results in a delayed glucose absorption and a lowering of postprandial hyperglycemia. Because its mechanism of action is different, the effect of acarbose tablets to enhance glycemic control is additive to that of sulfonylureas, insulin or metformin when used in combination. In addition, acarbose tablets diminish the insulinotropic and weight-increasing effects of sulfonylureas.
Acarbose has no inhibitory activity against lactase and consequently would not be expected to induce lactose intolerance.
Pharmacokinetics:
Absorption
In a study of 6 healthy men, less than 2% of an oral dose of acarbose was absorbed as active drug, while approximately 35% of total radioactivity from a 14C-labeled oral dose was absorbed. An average of 51% of an oral dose was excreted in the feces as unabsorbed drug-related radioactivity within 96 hours of ingestion. Because acarbose acts locally within the gastrointestinal tract, this low systemic bioavailability of parent compound is therapeutically desired. Following oral dosing of healthy volunteers with 14C-labeled acarbose, peak plasma concentrations of radioactivity were attained 14-24 hours after dosing, while peak plasma concentrations of active drug were attained at approximately 1 hour. The delayed absorption of acarbose-related radioactivity reflects the absorption of metabolites that may be formed by either intestinal bacteria or intestinal enzymatic hydrolysis.
Metabolism
Acarbose is metabolized exclusively within the gastrointestinal tract, principally by intestinal bacteria, but also by digestive enzymes. A fraction of these metabolites (approximately 34% of the dose) was absorbed and subsequently excreted in the urine. At least 13 metabolites have been separated chromatographically from urine specimens. The major metabolites have been identified as 4-methylpyrogallol derivatives (that is, sulfate, methyl, and glucuronide conjugates). One metabolite (formed by cleavage of a glucose molecule from acarbose) also has alpha-glucosidase inhibitory activity. This metabolite, together with the parent compound, recovered from the urine, accounts for less than 2% of the total administered dose.
Excretion
The fraction of acarbose that is absorbed as intact drug is almost completely excreted by the kidneys. When acarbose was given intravenously, 89% of the dose was recovered in the urine as active drug within 48 hours. In contrast, less than 2% of an oral dose was recovered in the urine as active (that is, parent compound and active metabolite) drug. This is consistent with the low bioavailability of the parent drug. The plasma elimination half-life of acarbose activity is approximately 2 hours in healthy volunteers. Consequently, drug accumulation does not occur with three times a day (t.i.d.) oral dosing.
Special Populations
The mean steady-state area under the curve (AUC) and maximum concentrations of acarbose were approximately 1.5 times higher in elderly compared to young volunteers; however, these differences were not statistically significant. Patients with severe renal impairment (Clcr < 25 mL/min/1.73m2) attained about 5 times higher peak plasma concentrations of acarbose and 6 times larger AUCs than volunteers with normal renal function. No studies of acarbose pharmacokinetic parameters according to race have been performed. In U.S. controlled clinical studies of acarbose tablets in patients with type 2 diabetes mellitus, reductions in glycosylated hemoglobin levels were similar in Caucasians (n=478) and African-Americans (n=167), with a trend toward a better response in Latinos (n=132).
Drug-Drug Interactions
Studies in healthy volunteers have shown that acarbose tablets have no effect on either the pharmacokinetics or pharmacodynamics of nifedipine, propranolol, or ranitidine. Acarbose tablets did not interfere with the absorption or disposition of the sulfonylurea glyburide in diabetic patients. Acarbose tablets may affect digoxin bioavailability and may require dose adjustment of digoxin by 16% (90% confidence interval: 8-23%), decrease mean C max of digoxin by 26% (90% confidence interval: 16-34%) and decreases mean trough concentrations of digoxin by 9% (90% confidence limit: 19% decrease to 2% increase). (See PRECAUTIONS , Drug Interactions. ) The amount of metformin absorbed while taking acarbose tablets was bioequivalent to the amount absorbed when taking placebo, as indicated by the plasma AUC values. However, the peak plasma level of metformin was reduced by approximately 20% when taking acarbose due to a slight delay in the absorption of metformin. There is little if any clinically significant interaction between acarbose and metformin.
HOW SUPPLIED
Acarbose Tablets, USP are supplied as follows:
For 25 mg: White to off white, round, biconvex tablets, debossed with "HP" on one side and "147" on other side.
For 50 mg: White to off white, round, biconvex tablets, debossed with "HP" and "148" on one side and plain on other side.
For 100 mg: White to off white, round, biconvex tablets, debossed with "HP" and "149" on one side and plain on other side.
Acarbose Tablets, USP 25 mg/50 mg/100 mg are available in Bottles of 90.
Strength | NDC | Tablet Identification | |
Bottles of 90 with child-resistance closure | 25 mg | 62135-453-90 | "HP" on one side and "147" on other side. |
Bottles of 90 with child-resistance closure | 50 mg | 62135-454-90 | "HP" and "148" on one side and plain on other side. |
Bottles of 90 with child-resistance closure | 100 mg | 62135-455-90 | "HP" and "149" on one side and plain on other side. |
Store at 20˚C to 25˚C (68˚F to 77˚F) [see USP Controlled Room Temperature]. Protect from moisture. For bottles, keep container tightly closed.
Manufactured by:
RA CHEM PHARMA LIMITED
Hyderabad – 500 076, INDIA
Manufactured for: Chartwell RX, LLC.
Congers, NY 10920
L71215
Revised: 01/2023