Get your patient on Acitretin - Acitretin capsule (Acitretin)
Acitretin - Acitretin capsule prescribing information
CONTRAINDICATIONS AND WARNINGS: Pregnancy
Acitretin must not be used by females who are pregnant, or who intend to become pregnant during therapy or at any time for at least 3 years following discontinuation of therapy. Acitretin also must not be used by females who may not use reliable contraception while undergoing treatment and for at least 3 years following discontinuation of treatment. Acitretin is a metabolite of etretinate (TEGISON), and major human fetal abnormalities have been reported with the administration of acitretin and etretinate. Potentially, any fetus exposed can be affected.
Clinical evidence has shown that concurrent ingestion of acitretin and ethanol has been associated with the formation of etretinate, which has a significantly longer elimination half-life than acitretin. Because the longer elimination half-life of etretinate would increase the duration of teratogenic potential for female patients, ethanol must not be ingested by female patients of childbearing potential either during treatment with acitretin or for 2 months after cessation of therapy. This allows for elimination of acitretin, thus removing the substrate for transesterification to etretinate. The mechanism of the metabolic process for conversion of acitretin to etretinate has not been fully defined. It is not known whether substances other than ethanol are associated with transesterification.
Acitretin has been shown to be embryotoxic and/or teratogenic in rabbits, mice, and rats at oral doses of 0.6, 3, and 15 mg per kg, respectively. These doses are approximately 0.2, 0.3, and 3 times the maximum recommended therapeutic dose, respectively, based on a mg-per-m 2 comparison.
Major human fetal abnormalities associated with acitretin and/or etretinate administration have been reported including meningomyelocele; meningoencephalocele; multiple synostoses; facial dysmorphia; syndactyly; absence of terminal phalanges; malformations of hip, ankle, and forearm; low-set ears; high palate; decreased cranial volume; cardiovascular malformation; and alterations of the skull and cervical vertebrae.
Acitretin should be prescribed only by those who have special competence in the diagnosis and treatment of severe psoriasis, are experienced in the use of systemic retinoids, and understand the risk of teratogenicity.
Because of the teratogenicity of acitretin, a program called the H.A.E.R.T. program, Healthier Acitretin Education and Remission Treatment, has been developed to educate women of childbearing potential and their healthcare providers about the serious risks associated with acitretin and to help prevent pregnancies from occurring with the use of this drug and for 3 years after its discontinuation. The H.A.E.R.T. program requirements are described below and program materials are available at https://www.alembicusa.com/AcitretinHAERT or may be requested by calling 1-866-210-9797 (see also PRECAUTIONS section).
Important Information for Women of Childbearing Potential: Acitretin should be considered only for women with severe psoriasis unresponsive to other therapies or whose clinical condition contraindicates the use of other treatments.
Females of reproductive potential must not be given a prescription for acitretin until pregnancy is excluded. Acitretin are contraindicated in females of reproductive potential unless the patient meets ALL of the following conditions :
• Must have had 2 negative urine or serum pregnancy tests with a sensitivity of at least 25 mIU per mL before receiving the initial prescription for acitretin. The first test (a screening test) is obtained by the prescriber when the decision is made to pursue therapy with acitretin. The second pregnancy test (a confirmation test) should be done during the first 5 days of the menstrual period immediately preceding the beginning of therapy with acitretin. For patients with amenorrhea, the second test should be done at least 11 days after the last act of unprotected sexual intercourse (without using 2 effective forms of contraception [birth control] simultaneously). If the second pregnancy test is negative, initiation of treatment with acitretin should begin within 7 days of the specimen collection. Acitretin should be limited to a monthly supply.
• Must have a pregnancy test with a sensitivity of at least 25 mIU per mL repeated every month during treatment with acitretin. The patient must have a negative result from a urine or serum pregnancy test before receiving a prescription for acitretin. To encourage compliance with this recommendation, a monthly supply of the drug should be prescribed. For at least 3 years after discontinuing therapy with acitretin, a pregnancy test must be repeated every 3 months.
• Must have selected and have committed to use 2 effective forms of contraception (birth control) simultaneously, at least 1 of which must be a primary form, unless absolute abstinence is the chosen method, or the patient has undergone a hysterectomy or is clearly postmenopausal.
• Patients must use 2 effective forms of contraception (birth control) simultaneously for at least 1 month prior to initiation of therapy with acitretin, during therapy with acitretin, and for at least 3 years after discontinuing therapy with acitretin. Counseling about contraception and behaviors associated with an increased risk of pregnancy must be repeated on a monthly basis by the prescriber during therapy with acitretin and every 3 months for at least 3 years following discontinuation of acitretin.
Effective forms of contraception include both primary and secondary forms of contraception. Primary forms of contraception include: tubal ligation, partner’s vasectomy, intrauterine devices, birth control pills, and injectable/implantable/insertable/topical hormonal birth control products. Secondary forms of contraception include condoms (with or without spermicide), diaphragms and cervical caps (which must be used with a spermicide), and vaginal sponges (contains spermicide).
Any birth control method can fail. Therefore, it is critically important that women of childbearing potential use 2 effective forms of contraception (birth control) simultaneously. It has not been established if there is a pharmacokinetic interaction between acitretin and combined oral contraceptives. However, it has been established that acitretin interferes with the contraceptive effect of microdosed progestin preparations. 1 Microdosed “minipill” progestin preparations are not recommended for use with acitretin. It is not known whether other progestin¬only contraceptives, such as implants and injectables, are adequate methods of contraception during acitretin therapy. Prescribers are advised to consult the package insert of any medication administered concomitantly with hormonal contraceptives, since some medications may decrease the effectiveness of these birth control products. Patients should be prospectively cautioned not to self-medicate with the herbal supplement St. John’s wort because a possible interaction has been suggested with hormonal contraceptives based on reports of breakthrough bleeding on oral contraceptives shortly after starting St. John’s wort. Pregnancies have been reported by users of combined hormonal contraceptives who also used some form of St. John’s wort (see PRECAUTIONS).
• Must have signed a Patient Agreement/Informed Consent for Female Patients that contains warnings about the risk of potential birth defects if the fetus is exposed to acitretin, about contraceptive failure, about the fact that they must not ingest beverages or products containing ethanol while taking acitretin and for 2 months after treatment with acitretin has been discontinued, and about preventing pregnancy while taking acitretin and for at least 3 years after discontinuing acitretin. If pregnancy does occur during therapy with acitretin or at any time for at least 3 years following discontinuation of acitretin, the prescriber and patient should discuss the possible effects on the pregnancy. The available information is as follows:
Acitretin, the active metabolite of etretinate, is teratogenic and is contraindicated during pregnancy. The risk of severe fetal malformations is well established when systemic retinoids are taken during pregnancy. Pregnancy must also be prevented after stopping acitretin therapy, while the drug is being eliminated to below a threshold blood concentration that would be associated with an increased incidence of birth defects. Because this threshold has not been established for acitretin in humans and because elimination rates vary among patients, the duration of posttherapy contraception to achieve adequate elimination cannot be calculated precisely. It is strongly recommended that contraception be continued for at least 3 years after stopping treatment with acitretin, based on the following considerations:
o In the absence of transesterification to form etretinate, greater than 98% of the acitretin would be eliminated within 2 months, assuming a mean elimination half-life of 49 hours. o In cases where etretinate is formed, as has been demonstrated with concomitant administration of acitretin and ethanol, ♦ greater than 98% of the etretinate formed would be eliminated in 2 years, assuming a mean elimination half-life of 120 days. ♦ greater than 98% of the etretinate formed would be eliminated in 3 years, based on the longest demonstrated elimination half-life of 168 days. However, etretinate was found in plasma and subcutaneous fat in one patient reported to have had sporadic alcohol intake, 52 months after she stopped acitretin therapy. 2
• Severe birth defects have been reported where conception occurred during the time interval when the patient was being treated with acitretin and/or etretinate. In addition, severe birth defects have also been reported when conception occurred after the mother completed therapy. These cases have been reported both prospectively (before the outcome was known) and retrospectively (after the outcome was known). The events below are listed without distinction as to whether the reported birth defects are consistent with retinoid-induced embryopathy or not.
♦ There have been 318 prospectively reported cases involving pregnancies and the use of etretinate, acitretin, or both. In 238 of these cases, the conception occurred after the last dose of etretinate (103 cases), acitretin (126), or both (9). Fetal outcome remained unknown in approximately one-half of these cases, of which 62 were terminated and 14 were spontaneous abortions. Fetal outcome is known for the other 118 cases and 15 of the outcomes were abnormal (including cases of absent hand/wrist, clubfoot, GI malformation, hypocalcemia, hypotonia, limb malformation, neonatal apnea/anemia, neonatal ichthyosis, placental disorder/death, undescended testicle, and 5 cases of premature birth). In the 126 prospectively reported cases where conception occurred after the last dose of acitretin only, 43 cases involved conception at least 1 year but less than 2 years after the last dose. There were 3 reports of abnormal outcomes out of these 43 cases (involving limb malformation, GI tract malformations, and premature birth). There were only 4 cases where conception occurred at least 2 years after the last dose but there were no reports of birth defects in these cases.
♦ There is also a total of 35 retrospectively reported cases where conception occurred at least 1 year after the last dose of etretinate, acitretin, or both. From these cases there are 3 reports of birth defects when the conception occurred at least 1 year but less than 2 years after the last dose of acitretin (including heart malformations, Turner’s Syndrome, and unspecified congenital malformations) and 4 reports of birth defects when conception occurred 2 or more years after the last dose of acitretin (including foot malformation, cardiac malformations [2 cases], and unspecified neonatal and infancy disorder). There were 3 additional abnormal outcomes in cases where conception occurred 2 or more years after the last dose of etretinate (including chromosome disorder, forearm aplasia, and stillbirth).
♦ Females who have taken TEGISON (etretinate) must continue to follow the contraceptive recommendations for TEGISON. TEGISON is no longer marketed in the US; for information, call Alembic Pharmaceuticals Limited at 1-866-210-9797. ♦ Patients should not donate blood during and for at least 3 years following the completion of therapy with acitretin because women of childbearing potential must not receive blood from patients being treated with acitretin.
Important Information for Males Taking Acitretin: Patients should not donate blood during and for at least 3 years following therapy with acitretin because women of childbearing potential must not receive blood from patients being treated with acitretin.
• Samples of seminal fluid from 3 male patients treated with acitretin and 6 male patient treated with etretinate have been assayed for the presence of acitretin. The maximum concentration of acitretin observed in the seminal fluid of these men was 12.5 ng per mL. Assuming an ejaculate volume of 10 mL, the amount of drug transferred in semen would be 125 ng, which is 1/200,000 of a single 25-mg capsule. Thus, although it appears that residual acitretin in seminal fluid poses little, if any, risk to a fetus while a male patient is taking the drug or after it is discontinued, the no-effect limit for teratogenicity is unknown and there is no registry for birth defects associated with acitretin. The available data are as follows:
There have been 25 cases of reported conception when the male partner was taking acitretin. The pregnancy outcome is known in 13 of these 25 cases. Of these, 9 reports were retrospective and 4 were prospective (meaning the pregnancy was reported prior to knowledge of the outcome) 3 .
| Timing of Paternal Acitretin Treatment Relative to Conception | Delivery of Healthy Neonate | Spontaneous Abortion | Induced Abortion | Total |
| At time of conception | 5 a | 5 | 1 | 11 |
| Discontinued ~4 weeks prior | 0 | 0 | 1 b | 1 |
| Discontinued ~6 to 8 months prior | 0 | 1 | 0 | 1 |
a Four of 5 cases were prospective b With malformation pattern not typical of retinoid embryopathy (bilateral cystic hygromas of neck. hypoplasia of lungs bilateral. VSD with overriding truncus arteriosus).
For All Patients : AN ACITRETIN MEDICATION GUIDE MUST BE GIVEN TO THE PATIENT EACH TIME ACITRETIN IS DISPENSED, AS REQUIRED BY LAW.
INDICATIONS AND USAGE
Acitretin capsules are indicated for the treatment of severe psoriasis in adults. Because of significant adverse effects associated with its use, acitretin capsules should be prescribed only by those knowledgeable in the systemic use of retinoids. In females of reproductive potential, acitretin capsules should be reserved for non-pregnant patients who are unresponsive to other therapies or whose clinical condition contraindicates the use of other treatments (see boxed CONTRAINDICATIONS AND WARNINGS — acitretin capsules can cause severe birth defects).
Most patients experience relapse of psoriasis after discontinuing therapy. Subsequent courses, when clinically indicated, have produced efficacy results similar to the initial course of therapy.
DOSAGE AND ADMINISTRATION
There is intersubject variation in the pharmacokinetics, clinical efficacy, and incidence of side effects with acitretin capsules. A number of the more common side effects are dose-related. Individualization of dosage is required to achieve sufficient therapeutic response while minimizing side effects. Therapy with acitretin capsules should be initiated at 25 to 50 mg per day, given as a single dose with the main meal. Maintenance doses of 25 to 50 mg per day may be given dependent upon an individual patient’s response to initial treatment. Relapses may be treated as outlined for initial therapy.
When acitretin capsules are used with phototherapy, the prescriber should decrease the phototherapy dose, dependent on the patient’s individual response (see PRECAUTIONS: General ).
Females who have taken TEGISON (etretinate) must continue to follow the contraceptive recommendations for TEGISON. TEGISON is no longer marketed in the US; for information, call Alembic Pharmaceuticals Limited at 1-866-210-9797 .
Information for Pharmacists
Acitretin capsules must only be dispensed in no more than a monthly supply. An acitretin capsules Medication Guide must be given to the patient each time acitretin capsules are dispensed, as required by law.
CONTRAINDICATIONS
Pregnancy : See boxed CONTRAINDICATIONS AND WARNINGS.
Acitretin is contraindicated in patients with severely impaired liver or kidney function and in patients with chronic abnormally elevated blood lipid values (see boxed WARNINGS: Hepatotoxicity, WARNINGS: Lipids and Possible Cardiovascular Effects, and PRECAUTIONS ).
An increased risk of hepatitis has been reported to result from combined use of methotrexate and etretinate. Consequently, the combination of methotrexate with acitretin is also contraindicated (see PRECAUTIONS: Drug Interactions ).
Since both acitretin and tetracyclines can cause increased intracranial pressure, their combined use is contraindicated (see WARNINGS: Pseudotumor Cerebri ).
Acitretin is contraindicated in cases of hypersensitivity (e.g., angioedema, urticaria) to the preparation (acitretin or excipients) or to other retinoids .
ADVERSE REACTIONS
Hypervitaminosis A produces a wide spectrum of signs and symptoms primarily of the mucocutaneous, musculoskeletal, hepatic, neuropsychiatric, and central nervous systems. Many of the clinical adverse reactions reported to date with administration of acitretin resemble those of the hypervitaminosis A syndrome. Adverse Events/Postmarketing Reports In addition to the events listed in the tables for the clinical trials, the following adverse events have been identified during postapproval use of acitretin. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Cardiovascular : Acute myocardial infarction, thromboembolism (see WARNINGS ), stroke. Immune System Disorders : Hypersensitivity, including angioedema and urticaria (see CONTRAINDICATIONS ). Nervous System : Myopathy with peripheral neuropathy has been reported during therapy with acitretin. Both conditions improved with discontinuation of the drug. Psychiatric : Aggressive feelings and/or suicidal thoughts have been reported. These events, including self-injurious behavior, have been reported in patients taking other systemically administered retinoids, as well as in patients taking acitretin. Since other factors may have contributed to these events, it is not known if they are related to acitretin (see PRECAUTIONS ). Reproductive : Vulvo-vaginitis due to Candida albicans. Skin and Appendages : Thinning of the skin, skin fragility, and scaling may occur all over the body, particularly on the palms and soles; nail fragility is frequently observed. Madarosis and exfoliative dermatitis/erythroderma have been reported (see WARNINGS ). Vascular Disorders : Capillary leak syndrome (see WARNINGS ).
Clinical Trials During clinical trials with acitretin, 513 of 525 (98%) subjects reported a total of 3,545 adverse events. One-hundred sixteen subjects (22%) left trials prematurely, primarily because of adverse experiences involving the mucous membranes and skin. Three subjects died. Two of the deaths were not drug-related (pancreatic adenocarcinoma and lung cancer); the other subject died of an acute myocardial infarction, considered remotely related to drug therapy. In clinical trials, acitretin was associated with elevations in liver function test results or triglyceride levels and hepatitis .
The tables below list by body system and frequency the adverse events reported during clinical trials of 525 subjects with psoriasis. Table 3. Adverse Events Frequently Reported during Clinical Trials Percent of Subjects Reporting (N = 525)
| Body System | >75% | 50% to 75% | 25% to 50% | 10% to 25% |
| CNS | Rigors | |||
| Eye Disorders | Xerophthalmia | |||
| Mucous Membranes | Cheilitis | Rhinitis | Dry mouth Epistaxis | |
| Musculoskeletal | Arthralgia Spinal hyperostosis (progression of existing lesions) | |||
| Skin and Appendages | Alopecia Skin peeling | Dry skin Nail disorder Pruritus | Erythematous rash Hyperesthesia Paresthesia Paronychia Skin atrophy Sticky skin |
Table 4. Adverse Events Less Frequently Reported during Clinical Trials (Some of Which May Bear No Relationship to Therapy) Percent of Subjects Reporting (N = 525)
| Body System | 1% to 10% | <1% | |||
|---|---|---|---|---|---|
| Body as a Whole | Anorexia Edema Fatigue Hot flashes Increased appetite | Alcohol intolerance Dizziness Fever Influenza-like symptoms | Malaise Moniliasis Muscle weakness Weight increase | ||
| Cardiovascular | Flushing | Chest pain Cyanosis Increased bleeding time | Intermittent claudication Peripheral ischemia | ||
| CNS (also see Psychiatric) | Headache Pain | Abnormal gait Migraine Neuritis | Pseudotumor cerebri (intracranial hypertension) | ||
| Eye Disorders | Abnormal/ blurred vision Blepharitis Conjunctivitis/ irritation Corneal epithelial abnormality | Decreased night vision/ night blindness Eye abnormality Eye pain Photophobia | Abnormal lacrimation Chalazion Conjunctival hemorrhage Corneal ulceration Diplopia Ectropion | Itchy eyes and lids Papilledema Recurrent sties Subepithelial corneal lesions | |
| Gastrointestinal | Abdominal pain Diarrhea Nausea Tongue disorder | Constipation Dyspepsia Esophagitis Gastritis Gastroenteritis | Glossitis Hemorrhoids Melena Tenesmus Tongue ulceration | ||
| Liver and Biliary | Hepatic function abnormal Hepatitis Jaundice | ||||
| Mucous Membranes | Gingival bleeding Gingivitis Increased saliva | Stomatitis Thirst Ulcerative stomatitis | Altered saliva Hemorrhage Anal disorder Pharyngitis Gum hyperplasia | ||
| Musculoskeletal | Arthritis Arthrosis Back pain Hypertonia Myalgia | Osteodynia Peripheral joint hyperostosis (progression of existing lesions) | Bone disorder Olecranon bursitis Spinal hyperostosis (new lesions) Tendonitis | ||
| Psychiatric | Depression Insomnia Somnolence | Anxiety Dysphonia Libido decreased Nervousness | |||
| Reproductive | Atrophic vaginitis Leukorrhea | ||||
| Respiratory | Sinusitis | Coughing Increased sputum Laryngitis | |||
| Skin and Appendages | Abnormal skin odor Abnormal hair texture Bullous eruption Cold/clammy skin Dermatitis Increased sweating Infection | Psoriasiform rash Purpura Pyogenic granuloma Rash Seborrhea Skin fissures Skin ulceration Sunburn | Acne Breast pain Cyst Eczema Fungal infection Furunculosis Hair discoloration Herpes simplex Hyperkeratosis Hypertrichosis Hypoesthesia Impaired healing Otitis media | Otitis externa Photosensitivity reaction Psoriasis aggravated Scleroderma Skin nodule Skin hypertrophy Skin disorder Skin irritation Sweat gland disorder Urticaria Verrucae | |
| Special Senses/ Other | Earache Taste perversion Tinnitus | Ceruminosis Deafness Taste loss | |||
| Urinary | Abnormal urine Dysuria Penis disorder | ||||
Laboratory: Therapy with acitretin induces changes in liver function tests in a significant number of patients. Elevations of AST (SGOT), ALT (SGPT) or LDH were experienced by approximately 1 in 3 subjects treated with acitretin. In most subjects, elevations were slight to moderate and returned to normal either during continuation of therapy or after cessation of treatment. In subjects receiving acitretin during clinical trials, 66% and 33% experienced elevation in triglycerides and chlesterol, respectively. Decreased high density lipoproteins (HDL) occurred in 40% (see WARNINGS ). Transient, usually reversible elevations of alkaline phosphatase have been observed.
Table 5 lists the laboratory abnormalities reported during clinical trials.
Table 5. Abnormal Laboratory Test Results Reported during Clinical Trials Percent of Subjects Reporting
| Body System | 50% to 75% | 25% to 50% | 10% to 25% | 1% to 10% |
| Electrolytes | Increased: –Phosphorus –Potassium –Sodium Increased and decreased: –Magnesium | Decreased: –Phosphorus –Potassium –Sodium Increased and decreased: –Calcium –Chloride | ||
| Hematologic | Increased: –Reticulocytes | Decreased: –Hematocrit –Hemoglobin –WBC Increased: –Haptoglobin –Neutrophils –WBC | Increased: –Bands –Basophils –Eosinophils –Hematocrit –Hemoglobin –Lymphocytes –Monocytes Decreased: –Haptoglobin –Lymphocytes –Neutrophils –Reticulocytes Increased or decreased: –Platelets –RBC | |
| Hepatic | Increased: –Cholesterol –LDH –SGOT –SGPT Decreased: –HDL cholesterol | Increased: –Alkaline phosphatase –Direct bilirubin –GGTP | Increased: –Globulin –Total bilirubin –Total protein Increased and decreased: –Serum albumin | |
| Miscellaneous | Increased: –Triglycerides | Increased: –CPK –Fasting blood sugar | Decreased: –Fasting blood sugar –High occult blood | Increased and decreased: –Iron |
| Renal | Increased: –Uric acid | Increased: –BUN –Creatinine | ||
| Urinary | WBC in urine | Acetonuria Hematuria RBC in urine | Glycosuria Proteinuria |
To report SUSPECTED ADVERSE REACTIONS, contact Alembic Pharmaceuticals Limited at 1-866-210-9797 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
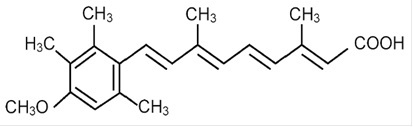
Acitretin, USP a retinoid, is available in 10-mg, 17.5-mg, and 25-mg gelatin capsules for oral administration. Chemically, acitretin is all-trans-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoic acid. It is a metabolite of etretinate and is related to both retinoic acid and retinol (vitamin A). It is a yellow to greenish-yellow crystalline powder with a molecular weight of 326.44. The structural formula is:
Each capsule contains acitretin, microcrystalline cellulose, maltodextrin, sodium ascorbate, sodium lauryl sulfate, colloidal silicon dioxide and magnesium stearate.
Gelatin capsule shells contain gelatin, iron oxide yellow, iron oxide red, titanium dioxide and sodium lauryl sulfate. Additionally, 10 mg and 25 mg gelatin capsule shells contains iron oxide black.
The gelatin capsule shells are printed with edible black ink containing shellac, iron oxide black and potassium hydroxide.
FDA approved dissolution test specifications differ from USP.
CLINICAL PHARMACOLOGY
The mechanism of action of acitretin is unknown.
Pharmacokinetics: Absorption: Oral absorption of acitretin is optimal when given with food. For this reason, acitretin was given with food in all of the following trials. After administration of a single 50-mg oral dose of acitretin to 18 healthy subjects, maximum plasma concentrations ranged from 196 to 728 ng per mL (mean: 416 ng per mL) and were achieved in 2 to 5 hours (mean: 2.7 hours). The oral absorption of acitretin is linear and proportional with increasing doses from 25 to 100 mg. Approximately 72% (range: 47% to 109%) of the administered dose was absorbed after a single 50-mg dose of acitretin was given to 12 healthy subjects.
Distribution: Acitretin is more than 99.9% bound to plasma proteins, primarily albumin.
Metabolism: (See Pharmacokinetic Drug Interactions: Ethanol.) Following oral absorption, acitretin undergoes extensive metabolism and interconversion by simple isomerization to its 13-cis form (cis-acitretin). The formation of cis-acitretin relative to parent compound is not altered by dose or fed/fast conditions of oral administration of acitretin. Both parent compound and isomer are further metabolized into chain-shortened breakdown products and conjugates, which are excreted. Following multiple-dose administration of acitretin, steady-state concentrations of acitretin and cis-acitretin in plasma are achieved within approximately 3 weeks.
Elimination: The chain-shortened metabolites and conjugates of acitretin and cis-acitretin are ultimately excreted in the feces (34% to 54%) and urine (16% to 53%). The terminal elimination half-life of acitretin following multiple-dose administration is 49 hours (range: 33 to 96 hours), and that of cis-acitretin under the same conditions is 63 hours (range: 28 to 157 hours). The accumulation ratio of the parent compound is 1.2; that of cis-acitretin is 6.6.
Special Populations: Psoriasis: In an 8-week trial of acitretin pharmacokinetics in subjects with psoriasis, mean steady-state trough concentrations of acitretin increased in a dose-proportional manner with dosages ranging from 10 to 50 mg daily. Acitretin plasma concentrations were nonmeasurable (<4 ng per mL) in all subjects 3 weeks after cessation of therapy.
Elderly: In a multiple-dose trial in healthy young (n = 6) and elderly (n = 8) subjects, a 2-fold increase in acitretin plasma concentrations were seen in elderly subjects, although the elimination half-life did not change.
Renal Failure: Plasma concentrations of acitretin were significantly (59.3%) lower in subjects with end-stage renal failure (n = 6) when compared with age-matched controls, following single 50-mg oral doses. Acitretin was not removed by hemodialysis in these subjects.
Pharmacokinetic Drug Interactions (see also boxed CONTRAINDICATIONS AND WARNINGS and PRECAUTIONS: Drug Interactions): In studies of in vivo pharmacokinetic drug interactions, no interaction was seen between acitretin and cimetidine, digoxin, phenprocoumon, or glyburide.
Ethanol: Clinical evidence has shown that etretinate (a retinoid with a much longer half-life, see below) can be formed with concurrent ingestion of acitretin and ethanol. In a 2-way crossover trial, all 10 subjects formed etretinate with concurrent ingestion of a single 100-mg oral dose of acitretin during a 3-hour period of ethanol ingestion (total ethanol, approximately 1.4 g per kg body weight). A mean peak etretinate concentration of 59 ng per mL (range: 22 to 105 ng per mL) was observed, and extrapolation of AUC values indicated that the formation of etretinate in this trial was comparable to a single 5-mg oral dose of etretinate. There was no detectable formation of etretinate when a single 100-mg oral dose of acitretin was administered without concurrent ethanol ingestion, although the formation of etretinate without concurrent ethanol ingestion cannot be excluded (see boxed CONTRAINDICATIONS AND WARNINGS ). Of 93 evaluable psoriatic subjects on acitretin therapy in several foreign trials (10 to 80 mg per day), 16% had measurable etretinate levels (>5 ng per mL).
Etretinate has a much longer elimination half-life compared with that of acitretin. In one trial the apparent mean terminal half-life after 6 months of therapy was approximately 120 days (range: 84 to 168 days). In another trial of 47 subjects treated chronically with etretinate, 5 had detectable serum drug levels (in the range of 0.5 to 12 ng per mL) 2.1 to 2.9 years after therapy was discontinued. The long half-life appears to be due to storage of etretinate in adipose tissue.
Progestin-only Contraceptives: It has not been established if there is a pharmacokinetic interaction between acitretin and combined oral contraceptives. However, it has been established that acitretin interferes with the contraceptive effect of microdosed progestin preparations. 1 Microdosed “minipill” progestin preparations are not recommended for use with acitretin. It is not known whether other progestin-only contraceptives, such as implants and injectables, are adequate methods of contraception during acitretin therapy.
CLINICAL STUDIES
In 2 double-blind, placebo-controlled trials, acitretin was administered once daily to subjects with severe psoriasis (e.g., covering at least 10% to 20% of the body surface area). At 8 weeks (see Table 1) subjects treated in Trial A with 50 mg of acitretin per day showed significant improvements ( P ≤0.05) relative to baseline and to placebo in the physician’s global evaluation and in the mean ratings of severity of psoriasis (scaling, thickness, and erythema). In Trial B, differences from baseline and from placebo were statistically significant ( P ≤0.05) for all variables at both the 25-mg and 50-mg doses; it should be noted for Trial B that no statistical adjustment for multiplicity was carried out.
Table 1. Summary of the Efficacy Results of the 8-Week Double-Blind Phase of Trials A and B of Acitretin
| Efficacy Variables | Trial A | Trial B | |||
|---|---|---|---|---|---|
| Total Daily Dose | Total Daily Dose | ||||
| Placebo (N = 29) | 50 mg (N = 29) | Placebo (N = 72) | 25 mg (N = 74) | 50 mg (N = 71) | |
| Physician’s Global Evaluation | |||||
| Baseline | 4.62 | 4.55 | 4.43 | 4.37 | 4.49 |
| Mean Change After 8 Weeks | −0.29 | −2 a | −0.06 | −1.06 a | −1.57 a |
| Scaling | |||||
| Baseline | 4.1 | 3.76 | 3.97 | 4.11 | 4.1 |
| Mean Change After 8 Weeks | −0.22 | −1.62 a | −0.21 | −1.5 a | −1.78 a |
| Thickness | |||||
| Baseline | 4.1 | 4.1 | 4.03 | 4.11 | 4.2 |
| Mean Change After 8 Weeks | −0.39 | −2.1 a | −0.18 | −1.43 a | −2.11 a |
| Erythema | |||||
| Baseline | 4.21 | 4.59 | 4.42 | 4.24 | 4.45 |
| Mean Change After 8 Weeks | −0.33 | −2.1 a | −0.37 | −1.12 a | −1.65 a |
a Values were statistically significantly different from placebo and from baseline ( P ≤0.05). No adjustment for multiplicity was done for Trial B.
The efficacy variables consisted of: the mean severity rating of scale, lesion thickness, erythema, and the physician’s global evaluation of the current status of the disease. Ratings of scaling, erythema, and lesion thickness, and the ratings of the global assessments were made using a 7-point scale (0 = none, 1 = trace, 2 = mild, 3 = mild-moderate, 4 = moderate, 5 = moderate-severe, 6 = severe).
A subset of 141 subjects from both pivotal Trials A and B continued to receive acitretin in an open fashion for up to 24 weeks. At the end of the treatment period, all efficacy variables, as indicated in Table 2, were significantly improved ( P ≤0.01) from baseline, including extent of psoriasis, mean ratings of psoriasis severity, and physician’s global evaluation.
Table 2. Summary of the First Course of Therapy with Acitretin (24 Weeks)
| Variables | Trial A | Trial B |
|---|---|---|
| Mean Total Daily Dose of Acitretin (mg) | 42.8 | 43.1 |
| Mean Duration of Therapy (Weeks) | 21.1 | 22.6 |
| Physician’s Global Evaluation | N = 39 | N = 98 |
| Baseline | 4.51 | 4.43 |
| Mean Change from Baseline | −2.26 a | −2.6 a |
| Scaling | N = 59 | N = 132 |
| Baseline | 3.97 | 4.07 |
| Mean Change from Baseline | −2.15 a | −2.42 a |
| Thickness | N = 59 | N = 132 |
| Baseline | 4 | 4.12 |
| Mean Change from Baseline | −2.44 a | −2.66 a |
| Erythema | N = 59 | N = 132 |
| Baseline | 4.35 | 4.33 |
| Mean Change from Baseline | −2.31 a | −2.29 a |
a Indicates that the difference from baseline was statistically significant (P ≤0.01).
The efficacy variables consisted of: the mean severity rating of scale, lesion thickness, erythema, and the physician’s global evaluation of the current status of the disease. Ratings of scaling, erythema, and lesion thickness, and the ratings of the global assessments were made using a 7-point scale (0 = none, 1 = trace, 2 = mild, 3 = mild-moderate, 4 = moderate, 5 = moderate-severe, 6 = severe).
All efficacy variables improved significantly in a subset of 55 subjects from Trial A treated for a second, 6-month maintenance course of therapy (for a total of 12 months of treatment); a small subset of subjects (n = 4) from Trial A continued to improve after a third 6-month course of therapy (for a total of 18 months of treatment).
HOW SUPPLIED
Acitretin capsules, USP are available as follows:
10 mg, opaque brown cap / opaque white body hard gelatin capsules size “4” having imprinting “A” on cap and “232” on body with black ink filled with yellow granular powder.
Bottle of 30 capsules with child resistant closure, NDC 62332-741-30
17.5 mg, opaque yellow cap / opaque yellow body hard gelatin capsules size “2” having imprinting “A” on cap and “233” on body with black ink filled with yellow granular powder.
Bottle of 30 capsules with child resistant closure, NDC 62332-742-30
25 mg, opaque brown cap / opaque yellow body hard gelatin capsules size “1” having imprinting “A” on cap and “235” on body with black ink filled with yellow granular powder.
Bottle of 30 capsules with child resistant closure, NDC 62332-743-30
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Protect from light. Avoid exposure to high temperatures and humidity after the bottle is opened.