Get your patient on Acyclovir - Acyclovir tablet (Acyclovir)
Acyclovir - Acyclovir tablet prescribing information
INDICATIONS & USAGE
Herpes Zoster Infections:
Acyclovir Tablet is indicated for the acute treatment of herpes zoster (shingles).
Genital Herpes:
Acyclovir Tablet is indicated for the treatment of initial episodes and the management of recurrent episodes of genital herpes.
Chickenpox:Acyclovir Tablet is indicated for the treatment of chickenpox (varicella).
DOSAGE & ADMINISTRATION
Acute Treatment of Herpes Zoster: 800 mg every 4 hours orally, 5 times daily for 7 to 10 days. Genital Herpes: Treatment of Initial Genital Herpes: 200 mg every 4 hours, 5 times daily for 10 days. Chronic Suppressive Therapy for Recurrent Disease: 400 mg 2 times daily for up to 12 months, followed by re-evaluation. Alternative regimens have included doses ranging from 200 mg 3 times daily to 200 mg 5 times daily. The frequency and severity of episodes of untreated genital herpes may change over time. After 1 year of therapy, the frequency and severity of the patient’s genital herpes infection should be re-evaluated to assess the need for continuation of therapy with acyclovir. Intermittent Therapy: 200 mg every 4 hours, 5 times daily for 5 days. Therapy should be initiated at the earliest sign or symptom (prodrome) of recurrence. Treatment of Chickenpox: Children (2 years of age and older): 20 mg/kg per dose orally 4 times daily (80 mg/kg/day) for 5 days. Children over 40 kg should receive the adult dose for chickenpox. Adults and Children over 40 kg: 800 mg 4 times daily for 5 days. Intravenous acyclovir is indicated for the treatment of varicella-zoster infections in immunocompromised patients. When therapy is indicated, it should be initiated at the earliest sign or symptom of chickenpox. There is no information about the efficacy of therapy initiated more than 24 hours after onset of signs and symptoms. Patients With Acute or Chronic Renal Impairment: In patients with renal impairment, the dose of Acyclovir Tablets should be modified as shown in Table 3.
| Table 3. Dosage Modification for Renal Impairment | |||
| Normal Dosage Regimen | Creatinine Clearance (mL/min/1.73 m 2 ) | Adjusted Dosage Regimen | |
| Dose (mg) | Dosing Interval | ||
| 200 mg every 4 hours | >10 0-10 | 200 200 | every 4 hours, 5x daily every 12 hours |
| 400 mg every 12 hours | >10 0-10 | 400 200 | every 12 hours every 12 hours |
| 800 mg every 4 hours | >25 10-25 0-10 | 800 800 800 | every 4 hours, 5x daily every 8 hours every 12 hours |
Hemodialysis: For patients who require hemodialysis, the mean plasma half-life of acyclovir during hemodialysis is approximately 5 hours. This results in a 60% decrease in plasma concentrations following a 6-hour dialysis period. Therefore, the patient’s dosing schedule should be adjusted so that an additional dose is administered after each dialysis. Peritoneal Dialysis : No supplemental dose appears to be necessary after adjustment of the dosing interval. Bioequivalence of Dosage Forms: One Acyclovir 800-mg tablet was shown to be bioequivalent to four Acyclovir 200-mg capsules (n = 24).
CONTRAINDICATIONS
Acyclovir is contraindicated for patients who develop hypersensitivity to acyclovir or valacyclovir.
ADVERSE REACTIONS
Herpes Simplex:
Short-Term Administration: The most frequent adverse events reported during clinical trials of treatment of genital herpes with acyclovir 200 mg administered orally 5 times daily every 4 hours for 10 days were nausea and/or vomiting in 8 of 298 patient treatments (2.7%). Nausea and/or vomiting occurred in 2 of 287 (0.7%) patients who received placebo.
Long-Term Administration: The most frequent adverse events reported in a clinical trial for the prevention of recurrences with continuous administration of 400 mg (two 200-mg capsules) 2 times daily for 1 year in 586 patients treated with acyclovir were nausea (4.8%) and diarrhea (2.4%). The 589 control patients receiving intermittent treatment of recurrences with acyclovir for 1 year reported diarrhea (2.7%), nausea (2.4%), and headache (2.2%). Herpes Zoster: The most frequent adverse event reported during 3 clinical trials of treatment of herpes zoster (shingles) with 800 mg of oral acyclovir 5 times daily for 7 to 10 days in 323 patients was malaise (11.5%). The 323 placebo recipients reported malaise (11.1%). Chickenpox: The most frequent adverse event reported during 3 clinical trials of treatment of chickenpox with oral acyclovir at doses of 10 to 20 mg/kg 4 times daily for 5 to 7 days or 800 mg 4 times daily for 5 days in 495 patients was diarrhea (3.2%). The 498 patients receiving placebo reported diarrhea (2.2%). Observed During Clinical Practice: In addition to adverse events reported from clinical trials, the following events have been identified during post-approval use of acyclovir. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to either their seriousness, frequency of reporting, potential causal connection to acyclovir, or a combination of these factors. General: Anaphylaxis, angioedema, fever, headache, pain, peripheral edema. Nervous: Aggressive behavior, agitation, ataxia, coma, confusion, decreased consciousness, delirium, dizziness, dysarthria, encephalopathy, hallucinations, paresthesia, psychosis, seizure, somnolence, tremors. These symptoms may be marked, particularly in older adults or in patients with renal impairment (see PRECAUTIONS ). Digestive: Diarrhea, gastrointestinal distress, nausea. Hematologic and Lymphatic: Anemia, leukocytoclastic vasculitis, leukopenia, lymphadenopathy, thrombocytopenia. Hepatobiliary Tract and Pancreas: Elevated liver function tests, hepatitis, hyperbilirubinemia, jaundice. Musculoskeletal: Myalgia. Skin: Alopecia, erythema multiforme, photosensitive rash, pruritus, rash, Stevens-Johnson syndrome, toxic epidermal necrolysis, urticaria. Special Senses: Visual abnormalities. Urogenital: Renal failure, renal pain (may be associated with renal failure), elevated blood urea nitrogen, elevated creatinine, hematuria (see WARNINGS ).
Drug Interactions
See CLINICAL PHARMACOLOGY : Pharmacokinetics.
DESCRIPTION
Acyclovir is a synthetic nucleoside analogue active against herpesviruses. Acyclovir Tablets are for oral administration. Each Acyclovir Tablet contains 400 mg or 800-mg of acyclovir. In addition, each tablet contains the inactive ingredients: magnesium stearate, microcrystalline cellulose, povidone, sodium starch glycolate and talc. The 800mg tablet also contains FD&C Blue No. 2 Lake.
Acyclovir is a white, crystalline powder with the molecular formula C 8 H 11 N 5 O 3 and a molecular weight of 225. The maximum solubility in water at 37°C is 2.5 mg/mL. The pka’s of acyclovir are 2.27 and 9.25.
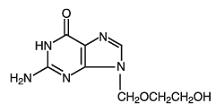
The chemical name of acyclovir is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]-6 H -purin-6-one; it has the following structural formula:
VIROLOGY
Mechanism of Antiviral Action: Acyclovir is a synthetic purine nucleoside analogue with in vitro and in vivo inhibitory activity against herpes simplex virus types 1 (HSV-1), 2 (HSV-2), and varicella-zoster virus (VZV).
The inhibitory activity of acyclovir is highly selective due to its affinity for the enzyme thymidine kinase (TK) encoded by HSV and VZV. This viral enzyme converts acyclovir into acyclovir monophosphate, a nucleotide analogue. The monophosphate is further converted into diphosphate by cellular guanylate kinase and into triphosphate by a number of cellular enzymes. In vitro, acyclovir triphosphate stops replication of herpes viral DNA. This is accomplished in 3 ways: 1) competitive inhibition of viral DNA polymerase, 2) incorporation into and termination of the growing viral DNA chain, and 3) inactivation of the viral DNA polymerase. The greater antiviral activity of acyclovir against HSV compared with VZV is due to its more efficient phosphorylation by the viral TK.
Antiviral Activities:
The quantitative relationship between the in vitro susceptibility of herpes viruses to antivirals and the clinical response to therapy has not been established in humans, and virus sensitivity testing has not been standardized. Sensitivity testing results, expressed as the concentration of drug required to inhibit by 50% the growth of virus in cell culture (IC 50 ), vary greatly depending upon a number of factors. Using plaque-reduction assays, the IC 50 against herpes simplex virus isolates ranges from 0.02 to 13.5 mcg/mL for HSV-1 and from 0.01 to 9.9 mcg/mL for HSV-2. The IC 50 for acyclovir against most laboratory strains and clinical isolates of VZV ranges from 0.12 to 10.8 mcg/mL. Acyclovir also demonstrates activity against the Oka vaccine strain of VZV with a mean IC 50 of 1.35 mcg/mL.
Drug Resistance: Resistance of HSV and VZV to acyclovir can result from qualitative and quantitative changes in the viral TK and/or DNA polymerase. Clinical isolates of HSV and VZV with reduced susceptibility to acyclovir have been recovered from immunocompromised patients, especially with advanced HIV infection. While most of the acyclovir-resistant mutants isolated thus far from immunocompromised patients have been found to be TK-deficient mutants, other mutants involving the viral TK gene (TK partial and TK altered) and DNA polymerase have been isolated. TK-negative mutants may cause severe disease in infants and immunocompromised adults. The possibility of viral resistance to acyclovir should be considered in patients who show poor clinical response during therapy.
CLINICAL PHARMACOLOGY
Pharmacokinetics:
The pharmacokinetics of acyclovir after oral administration have been evaluated in healthy volunteers and in immunocompromised patients with herpes simplex or varicella-zoster virus infection. Acyclovir pharmacokinetic parameters are summarized in Table 1.
| Table 1. Acyclovir Pharmacokinetic Characteristics (Range) | |
| Parameter | Range |
| Plasma protein binding | 9% to 33% |
| Plasma elimination half-life | 2.5 to 3.3 hr |
| Average oral bioavailability | 10% to 20%• |
•Bioavailability decreases with increasing dose.
In one multiple-dose, crossover study in healthy subjects (n = 23), it was shown that increases in plasma acyclovir concentrations were less than dose proportional with increasing dose, as shown in Table 2. The decrease in bioavailability is a function of the dose and not the dosage form.
| Table 2. AcyclovirPeak and Trough Concentrations at Steady State | |||
| Parameter | 200 mg | 400 mg | 800 mg |
| C ss max | 0.83 mcg/mL | 1.21 mcg/mL | 1.61 mcg/mL |
| C ss trough | 0.46 mcg/mL | 0.63 mcg/mL | 0.83 mcg/mL |
There was no effect of food on the absorption of acyclovir (n = 6); therefore, Acyclovir Tablets may be administered with or without food.
The only known urinary metabolite is 9-[(carboxymethoxy)methyl]guanine.
Special Populations: Adults With Impaired Renal Function: The half-life and total body clearance of acyclovir are dependent on renal function. A dosage adjustment is recommended for patients with reduced renal function (see DOSAGE AND ADMINISTRATION ). Geriatrics: Acyclovir plasma concentrations are higher in geriatric patients compared with younger adults, in part due to age-related changes in renal function. Dosage reduction may be required in geriatric patients with underlying renal impairment (see PRECAUTIONS: Geriatric Use ). Pediatrics: In general, the pharmacokinetics of acyclovir in pediatric patients is similar to that of adults. Mean half-life after oral doses of 300 mg/m 2 and 600 mg/m 2 in pediatric patients aged 7 months to 7 years was 2.6 hours (range 1.59 to 3.74 hours). Drug Interactions:
Coadministration of probenecid with intravenous acyclovir has been shown to increase the mean acyclovir half-life and the area under the concentration-time curve. Urinary excretion and renal clearance were correspondingly reduced.
Clinical Trials: Initial Genital Herpes: Double-blind, placebo-controlled studies have demonstrated that orally administered acyclovir significantly reduced the duration of acute infection and duration of lesion healing. The duration of pain and new lesion formation was decreased in some patient groups.
Recurrent Genital Herpes: Double-blind, placebo-controlled studies in patients with frequent recurrences (6 or more episodes per year) have shown that orally administered acyclovir given daily for 4 months to 10 years prevented or reduced the frequency and/or severity of recurrences in greater than 95% of patients.
In a study of patients who received acyclovir 400 mg twice daily for 3 years, 45%, 52%, and 63% of patients remained free of recurrences in the first, second, and third years, respectively. Serial analyses of the 3-month recurrence rates for the patients showed that 71% to 87% were recurrence free in each quarter.
Herpes Zoster Infections: In a double-blind, placebo-controlled study of immunocompetent patients with localized cutaneous zoster infection, acyclovir (800 mg 5 times daily for 10 days) shortened the times to lesion scabbing, healing, and complete cessation of pain, and reduced the duration of viral shedding and the duration of new lesion formation.
In a similar double-blind, placebo-controlled study, acyclovir (800 mg 5 times daily for 7 days) shortened the times to complete lesion scabbing, healing, and cessation of pain; reduced the duration of new lesion formation; and reduced the prevalence of localized zoster-associated neurologic symptoms (paresthesia, dysesthesia, or hyperesthesia).
Treatment was begun within 72 hours of rash onset and was most effective if started within the first 48 hours.
Adults greater than 50 years of age showed greater benefit. Chickenpox: Three randomized, double-blind, placebo-controlled trials were conducted in 993 pediatric patients aged 2 to 18 years with chickenpox. All patients were treated within 24 hours after the onset of rash. In 2 trials, acyclovir was administered at 20 mg/kg 4 times daily (up to 3,200 mg per day) for 5 days. In the third trial, doses of 10, 15, or 20 mg/kg were administered 4 times daily for 5 to 7 days. Treatment with acyclovir shortened the time to 50% healing; reduced the maximum number of lesions; reduced the median number of vesicles; decreased the median number of residual lesions on day 28; and decreased the proportion of patients with fever, anorexia, and lethargy by day 2. Treatment with acyclovir did not affect varicella-zoster virus-specific humoral or cellular immune responses at 1 month or 1 year following treatment.
HOW SUPPLIED
Acyclovir Tablets, USP 400 mg is White to off white, oval, biconvex, uncoated tablets, debossed with ‘CP114’ on one side and ‘400’ on other side. It is available as follows:
Bottle of 100 (NDC 72241-026-05)
Bottle of 500 (NDC 72241-026-10)
Acyclovir Tablets, USP 800 mg is Light blue to blue, oval, biconvex, uncoated tablets, debossed with ‘CP113’ on one side and ‘800’ on the other side. It is available as follows:
Bottle of 100 (NDC 72241-027-05)
Bottle of 500 (NDC 72241-027-10)
Store at 15° to 25°C (59° to 77°F) and protect from light and moisture. Dispense in tight, light-resistant container as defined in the USP.
Made in India by:
Cadila Pharmaceuticals Limited,
1389, Dholka, District - Ahmedabad,
Gujarat State, INDIA
Manufactured for:
Modavar Pharmaceuticals LLC.
1016, 16th Street, Suite 602,
Washington, DC 20036
Toll free: 800 688 4697
Revised: June 2021