Get your patient on Adenosine - Adenosine injection (Adenosine)
Adenosine - Adenosine injection prescribing information
Indications and Usage
Intravenous adenosine injection is indicated for the following.
Conversion to sinus rhythm of paroxysmal supraventricular tachycardia (PSVT), including that associated with accessory bypass tracts (Wolff-Parkinson-White Syndrome). When clinically advisable, appropriate vagal maneuvers (e.g., Valsalva maneuver), should be attempted prior to adenosine injection administration.
It is important to be sure the adenosine injection solution actually reaches the systemic circulation (see DOSAGE AND ADMINISTRATION ).
Adenosine Injection does not convert atrial flutter, atrial fibrillation, or ventricular tachycardia to normal sinus rhythm. In the presence of atrial flutter or atrial fibrillation, a transient modest slowing of ventricular response may occur immediately following adenosine injection administration.
Dosage and Administration
For rapid bolus intravenous use only.
Adenosine Injection, USP should be given as a rapid bolus by the peripheral intravenous route. To be certain the solution reaches the systemic circulation, it should be administered either directly into a vein or, if given into an IV line, it should be given as close to the patient as possible and followed by a rapid saline flush.
Adult Patients
The dose recommendation is based on clinical studies with peripheral venous bolus dosing. Central venous (CVP or other) administration of adenosine injection has not been systematically studied.
The recommended intravenous doses for adults are as follows:
Initial dose: 6 mg given as a rapid intravenous bolus (administered over a 1 to 2 second period).
Repeat administration: If the first dose does not result in elimination of the supraventricular tachycardia within 1 to 2 minutes, 12 mg should be given as a rapid intravenous bolus. This 12 mg dose may be repeated a second time if required.
Pediatric Patients
The dosages used in neonates, infants, children and adolescents were equivalent to those administered to adults on a weight basis.
Pediatric Patients with a Body Weight < 50 kg:
Initial dose: Give 0.05 to 0.1 mg/kg as a rapid IV bolus given either centrally or peripherally. A saline flush should follow.
Repeat administration: If conversion of PSVT does not occur within 1-2 minutes, additional bolus injections of adenosine can be administered at incrementally higher doses, increasing the amount given by 0.05 to 0.1 mg/kg. Follow each bolus with a saline flush. This process should continue until sinus rhythm is established or a maximum single dose of 0.3 mg/kg is used.
Pediatric Patients with a Body Weight ≥ 50 kg: Administer the adult dose.
Doses greater than 12 mg are not recommended for adult and pediatric patients.
NOTE: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
Contraindications
Intravenous adenosine injection is contraindicated in:
- Second- or third-degree A-V block (except in patients with a functioning artificial pacemaker).
- Sinus node disease, such as sick sinus syndrome or symptomatic bradycardia (except in patients with a functioning artificial pacemaker).
- Known hypersensitivity to adenosine.
Adverse Reactions
The following reactions were reported with intravenous adenosine injection used in controlled U.S. clinical trials. The placebo group had a less than 1% rate of all of these reactions.
Cardiovascular
Facial flushing (18%), headache (2%), sweating, palpitations, chest pain, hypotension (less than 1%).
Respiratory
Shortness of breath/dyspnea (12%), chest pressure (7%), hyperventilation, head pressure (less than 1%).
Central Nervous System
Lightheadedness (2%), dizziness, tingling in arms, numbness (1%), apprehension, blurred vision, burning sensation, heaviness in arms, neck and back pain (less than 1%).
Gastrointestinal
Nausea (3%), metallic taste, tightness in throat, pressure in groin (less than 1%).
Post Marketing Experience (see WARNINGS )
The following adverse events have been reported from marketing experience with adenosine injection. Because these events are reported voluntarily from a population of uncertain size, are associated with concomitant diseases and multiple drug therapies and surgical procedures, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions to include these events in labeling are typically based on one or more of the following factors: (1) seriousness of the event, (2) frequency of the reporting, (3) strength of causal connection to the drug, or a combination of these factors.
Cardiovascular
Prolonged asystole, ventricular tachycardia, ventricular fibrillation, transient increase in blood pressure, bradycardia, atrial fibrillation, and Torsade de Pointes
Respiratory
Bronchospasm
Central Nervous System
Seizure activity, including tonic clonic (grand mal) seizures, and loss of consciousness.
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals, Inc. at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Intravenous adenosine injection has been effectively administered in the presence of other cardioactive drugs, such as quinidine, beta-adrenergic blocking agents, calcium channel blocking agents, and angiotensin converting enzyme inhibitors, without any change in the adverse reaction profile. Digoxin and verapamil use may be rarely associated with ventricular fibrillation when combined with adenosine injection (see WARNINGS ). Because of the potential for additive or synergistic depressant effects on the SA and AV nodes, however, adenosine injection should be used with caution in the presence of these agents. The use of adenosine injection in patients receiving digitalis may be rarely associated with ventricular fibrillation (see WARNINGS ).
The effects of adenosine are antagonized by methylxanthines such as caffeine and theophylline. In the presence of these methylxanthines, larger doses of adenosine may be required or adenosine may not be effective. Adenosine effects are potentiated by dipyridamole. Thus, smaller doses of adenosine may be effective in the presence of dipyridamole. Carbamazepine has been reported to increase the degree of heart block produced by other agents. As the primary effect of adenosine is to decrease conduction through the A-V node, higher degrees of heart block may be produced in the presence of carbamazepine.
Description
Adenosine is an endogenous nucleoside occurring in all cells of the body. It is chemically 6-amino-9-β-D-ribofuranosyl-9-H-purine and has the following structural formula:
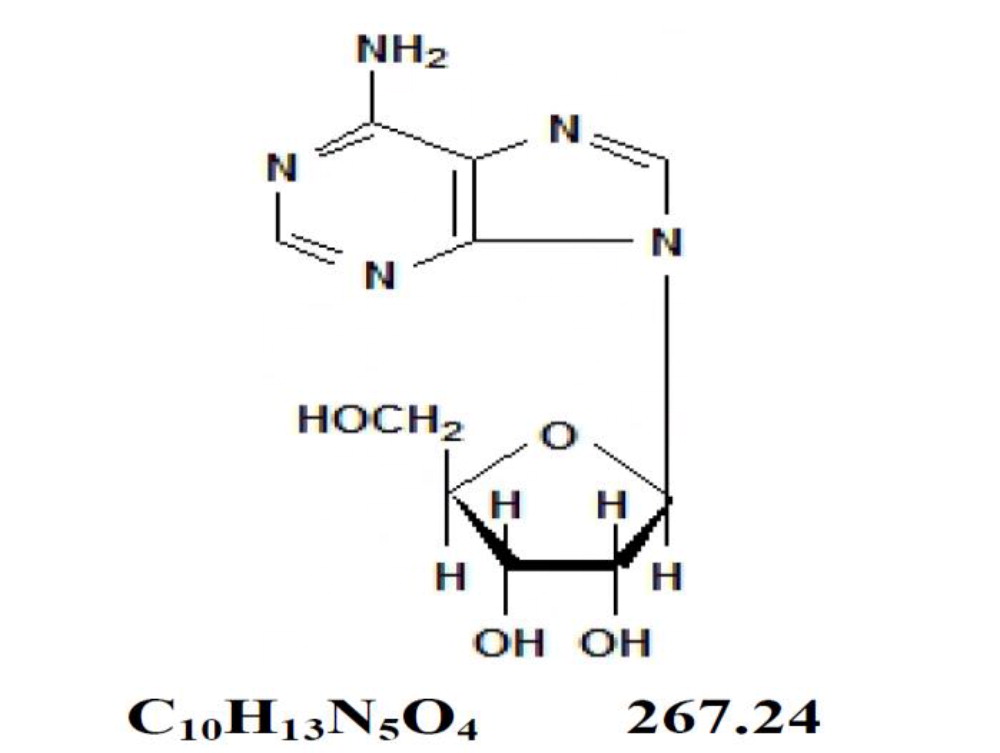
Adenosine is a white crystalline powder. It is soluble in water and practically insoluble in alcohol. Solubility increases by warming and lowering the pH. Adenosine is not chemically related to other antiarrhythmic drugs. Adenosine Injection is a sterile, nonpyrogenic solution for rapid bolus intravenous injection. Each mL contains 3 mg adenosine and 9 mg sodium chloride in Water for Injection. The pH of the solution is between 4.5 and 7.5.
Clinical Pharmacology
Mechanism of Action
Adenosine Injection slows conduction time through the A-V node, can interrupt the reentry pathways through the A-V node, and can restore normal sinus rhythm in patients with paroxysmal supraventricular tachycardia (PSVT), including PSVT associated with Wolff-Parkinson-White Syndrome.
Adenosine Injection is antagonized competitively by methylxanthines such as caffeine and theophylline, and potentiated by blockers of nucleoside transport such as dipyridamole. Adenosine Injection is not blocked by atropine.
Hemodynamics
The intravenous bolus dose of 6 or 12 mg adenosine injection usually has no systemic hemodynamic effects. When larger doses are given by infusion, adenosine decreases blood pressure by decreasing peripheral resistance.
Pharmacokinetics
Intravenously administered adenosine is rapidly cleared from the circulation via cellular uptake, primarily by erythrocytes and vascular endothelial cells. This process involves a specific transmembrane nucleoside carrier system that is reversible, nonconcentrative, and bidirectionally symmetrical. Intracellular adenosine is rapidly metabolized either via phosphorylation to adenosine monophosphate by adenosine kinase, or via deamination to inosine by adenosine deaminase in the cytosol. Since adenosine kinase has a lower K m and V max than adenosine deaminase, deamination plays a significant role only when cytosolic adenosine saturates the phosphorylation pathway. Inosine formed by deamination of adenosine can leave the cell intact or can be degraded to hypoxanthine, xanthine, and ultimately uric acid. Adenosine monophosphate formed by phosphorylation of adenosine is incorporated into the high-energy phosphate pool. While extracellular adenosine is primarily cleared by cellular uptake with a half-life of less than 10 seconds in whole blood, excessive amounts may be deaminated by an ecto-form of adenosine deaminase. As adenosine injection requires no hepatic or renal function for its activation or inactivation, hepatic and renal failure would not be expected to alter its effectiveness or tolerability.
Clinical Trial Results
In controlled studies in the United States, bolus doses of 3, 6, 9, and 12 mg were studied. A cumulative 60% of patients with paroxysmal supraventricular tachycardia had converted to normal sinus rhythm within one minute after an intravenous bolus dose of 6 mg adenosine injection (some converted on 3 mg and failures were given 6 mg), and a cumulative 92% converted after a bolus dose of 12 mg. Seven to sixteen percent of patients converted after 1 to 4 placebo bolus injections. Similar responses were seen in a variety of patient subsets, including those using or not using digoxin, those with Wolff-Parkinson-White Syndrome, males, females, blacks, Caucasians, and Hispanics.
Adenosine is not effective in converting rhythms other than PSVT, such as atrial flutter, atrial fibrillation, or ventricular tachycardia, to normal sinus rhythm.
How Supplied
Adenosine Injection, USP is supplied as a sterile non-pyrogenic solution in normal saline.
| NDC | Adenosine Injection, USP (3 mg per mL) | Package Factor |
| 25021-318-02 | 6 mg per 2 mL Single-Dose Vial | 10 vials per carton |
| 25021-318-04 | 12 mg per 4 mL Single-Dose Vial | 10 vials per carton |
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
DO NOT REFRIGERATE as crystallization may occur. If crystallization has occurred, dissolve crystals by warming to room temperature. The solution must be clear at the time of use.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
Mechanism of Action
Adenosine Injection slows conduction time through the A-V node, can interrupt the reentry pathways through the A-V node, and can restore normal sinus rhythm in patients with paroxysmal supraventricular tachycardia (PSVT), including PSVT associated with Wolff-Parkinson-White Syndrome.
Adenosine Injection is antagonized competitively by methylxanthines such as caffeine and theophylline, and potentiated by blockers of nucleoside transport such as dipyridamole. Adenosine Injection is not blocked by atropine.