Get your patient on Albuterol Sulfate - Albuterol Sulfate aerosol, Metered (Albuterol Sulfate)
Albuterol Sulfate - Albuterol Sulfate aerosol, Metered prescribing information
INDICATIONS AND USAGE
Albuterol Sulfate Inhalation Aerosol is indicated in adults and children 4 years of age and older for the treatment or prevention of bronchospasm with reversible obstructive airway disease and for the prevention of exercise-induced bronchospasm.
DOSAGE AND ADMINISTRATION
For treatment of acute episodes of bronchospasm or prevention of asthmatic symptoms, the usual dosage for adults and children 4 years of age and older is two inhalations repeated every 4 to 6 hours. More frequent administration or a larger number of inhalations is not recommended. In some patients, one inhalation every 4 hours may be sufficient. Each actuation of Albuterol Sulfate Inhalation Aerosol delivers 108 mcg of albuterol sulfate (equivalent to 90 mcg of albuterol base) from the mouthpiece. It is recommended to prime the inhaler before using for the first time and in cases where the inhaler has not been used for more than 2 weeks by releasing four “test sprays” into the air, away from the face.
Exercise Induced Bronchospasm Prevention: The usual dosage for adults and children 4 years of age and older is two inhalations 15 to 30 minutes before exercise.
To maintain proper use of this product, it is important that the mouthpiece be washed and dried thoroughly at least once a week. The inhaler may cease to deliver medication if not properly cleaned and dried thoroughly (see PRECAUTIONS, Information for Patients section). Keeping the plastic mouthpiece clean is very important to prevent medication buildup and blockage. The inhaler may cease to deliver medication if not properly cleaned and air dried thoroughly. If the mouthpiece becomes blocked, washing the mouthpiece will remove the blockage.
If a previously effective dose regimen fails to provide the usual response, this may be a marker of destabilization of asthma and requires reevaluation of the patient and the treatment regimen, giving special consideration to the possible need for anti-inflammatory treatment, e.g., corticosteroids.
CONTRAINDICATIONS
Albuterol Sulfate Inhalation Aerosol is contraindicated in patients with a history of hypersensitivity to albuterol or any other Albuterol Sulfate Inhalation Aerosol components.
ADVERSE REACTIONS
Adverse reaction information concerning Albuterol Sulfate Inhalation Aerosol is derived from a 12-week, double-blind, double-dummy study which compared Albuterol Sulfate Inhalation Aerosol, a CFC 11/12 propelled albuterol inhaler, and an HFA-134a placebo inhaler in 565 asthmatic patients. The following table lists the incidence of all adverse events (whether considered by the investigator drug related or unrelated to drug) from this study which occurred at a rate of 3% or greater in the Albuterol Sulfate Inhalation Aerosol treatment group and more frequently in the Albuterol Sulfate Inhalation Aerosol treatment group than in the placebo group. Overall, the incidence and nature of the adverse reactions reported for Albuterol Sulfate Inhalation Aerosol and a CFC 11/12 propelled albuterol inhaler were comparable.
•This table includes all adverse events (whether considered by the investigator drug related or unrelated to drug) which occurred at an incidence rate of at least 3% in the Albuterol Sulfate Inhalation Aerosol group and more frequently in the Albuterol Sulfate Inhalation Aerosol group than in the HFA-134a placebo inhaler group. | ||||
| Body System/ Adverse Event (Preferred Term) | Albuterol Sulfate Inhalation Aerosol (N=193) | CFC 11/12 Propelled Albuterol Inhaler (N=186) | HFA-134a Placebo Inhaler (N=186) | |
| Application Site Disorders | Inhalation Site Sensation | 6 | 9 | 2 |
| Inhalation Taste Sensation | 4 | 3 | 3 | |
| Body as a Whole | Allergic Reaction/Symptoms | 6 | 4 | <1 |
| Back Pain | 4 | 2 | 3 | |
| Fever | 6 | 2 | 5 | |
| Central and Peripheral Nervous System | Tremor | 7 | 8 | 2 |
| Gastrointestinal System | Nausea | 10 | 9 | 5 |
| Vomiting | 7 | 2 | 3 | |
| Heart Rate and Rhythm Disorder | Tachycardia | 7 | 2 | <1 |
| Psychiatric Disorders | Nervousness | 7 | 9 | 3 |
| Respiratory System Disorders | Respiratory Disorder (unspecified) | 6 | 4 | 5 |
| Rhinitis | 16 | 22 | 14 | |
| Upper Resp Tract Infection | 21 | 20 | 18 | |
| Urinary System Disorder | Urinary Tract Infection | 3 | 4 | 2 |
Adverse events reported by less than 3% of the patients receiving Albuterol Sulfate Inhalation Aerosol, and by a greater proportion of Albuterol Sulfate Inhalation Aerosol patients than placebo patients, which have the potential to be related to Albuterol Sulfate Inhalation Aerosol include: dysphonia, increased sweating, dry mouth, chest pain, edema, rigors, ataxia, leg cramps, hyperkinesia, eructation, flatulence, tinnitus, diabetes mellitus, anxiety, depression, somnolence, rash. Palpitation and dizziness have also been observed with Albuterol Sulfate Inhalation Aerosol.
Adverse events reported in a 4-week pediatric clinical trial comparing Albuterol Sulfate Inhalation Aerosol and a CFC 11/12 propelled albuterol inhaler occurred at a low incidence rate and were similar to those seen in the adult trials.
In small, cumulative dose studies, tremor, nervousness, and headache appeared to be dose related.
Rare cases of urticaria, angioedema, rash, bronchospasm, and oropharyngeal edema have been reported after the use of inhaled albuterol. In addition, albuterol, like other sympathomimetic agents, can cause adverse reactions such as hypertension, angina, vertigo, central nervous system stimulation, insomnia, headache, metabolic acidosis, and drying or irritation of the oropharynx.
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc., at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
- Beta-Blockers: Beta-adrenergic-receptor blocking agents not only block the pulmonary effect of beta-agonists, such as Albuterol Sulfate Inhalation Aerosol, but may produce severe bronchospasm in asthmatic patients. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-adrenergic blocking agents in patients with asthma. In this setting, cardioselective beta-blockers should be considered, although they should be administered with caution.
- Diuretics: The ECG changes and/or hypokalemia which may result from the administration of nonpotassium-sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the coadministration of beta-agonists with nonpotassium-sparing diuretics.
- Albuterol-Digoxin: Mean decreases of 16% and 22% in serum digoxin levels were demonstrated after single-dose intravenous and oral administration of albuterol, respectively, to normal volunteers who had received digoxin for 10 days. The clinical significance of these findings for patients with obstructive airway disease who are receiving albuterol and digoxin on a chronic basis is unclear; nevertheless, it would be prudent to carefully evaluate the serum digoxin levels in patients who are currently receiving digoxin and albuterol.
- Monoamine Oxidase Inhibitors or Tricyclic Antidepressants: Albuterol Sulfate Inhalation Aerosol should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents, because the action of albuterol on the cardiovascular system may be potentiated.
DESCRIPTION
The active component of Albuterol Sulfate Inhalation Aerosol is albuterol sulfate, USP racemic α 1 [( tert- Butylamino)methyl]-4-hydroxy- m -xylene-α,α'-diol sulfate (2:1)(salt), a relatively selective beta 2 -adrenergic bronchodilator having the following chemical structure:
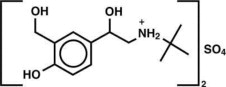
Albuterol sulfate is the official generic name in the United States. The World Health Organization recommended name for the drug is salbutamol sulfate. The molecular weight of albuterol sulfate is 576.7, and the empirical formula is (C 13 H 21 NO 3 ) 2 •H 2 SO 4 . Albuterol sulfate is a white to off-white crystalline solid. It is soluble in water and slightly soluble in ethanol. Albuterol Sulfate Inhalation Aerosol is a pressurized metered-dose aerosol unit for oral inhalation. It contains a microcrystalline suspension of albuterol sulfate in propellant HFA-134a (1,1,1,2-tetrafluoroethane), ethanol, and oleic acid.
Each actuation delivers 120 mcg albuterol sulfate, USP from the valve and 108 mcg albuterol sulfate, USP from the mouthpiece (equivalent to 90 mcg of albuterol base from the mouthpiece). Each canister provides 200 inhalations. It is recommended to prime the inhaler before using for the first time and in cases where the inhaler has not been used for more than 2 weeks by releasing four “test sprays” into the air, away from the face.
This product does not contain chlorofluorocarbons (CFCs) as the propellant.
CLINICAL PHARMACOLOGY
Mechanism of Action In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta 2 -adrenergic receptors compared with isoproterenol. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors on bronchial smooth muscle, data indicate that there is a population of beta 2 -receptors in the human heart existing in a concentration between 10% and 50% of cardiac beta-adrenergic receptors. The precise function of these receptors has not been established. (See WARNINGS, Cardiovascular Effects section.)
Activation of beta 2 -adrenergic receptors on airway smooth muscle leads to the activation of adenylcyclase and to an increase in the intracellular concentration of cyclic-3',5'-adenosine monophosphate (cyclic AMP). This increase of cyclic AMP leads to the activation of protein kinase A, which inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in relaxation. Albuterol relaxes the smooth muscles of all airways, from the trachea to the terminal bronchioles. Albuterol acts as a functional antagonist to relax the airway irrespective of the spasmogen involved, thus protecting against all bronchoconstrictor challenges. Increased cyclic AMP concentrations are also associated with the inhibition of release of mediators from mast cells in the airway.
Albuterol has been shown in most clinical trials to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes.
Preclinical Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood-brain barrier and reaches brain concentrations amounting to approximately 5% of the plasma concentrations. In structures outside the blood-brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those in the whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta 2 -agonist and methylxanthines were administered concurrently. The clinical significance of these findings is unknown.
Propellant HFA-134a is devoid of pharmacological activity except at very high doses in animals (380-1300 times the maximum human exposure based on comparisons of AUC values), primarily producing ataxia, tremors, dyspnea, or salivation. These are similar to effects produced by the structurally related chlorofluorocarbons (CFCs), which have been used extensively in metered dose inhalers.
In animals and humans, propellant HFA-134a was found to be rapidly absorbed and rapidly eliminated, with an elimination half-life of 3 to 27 minutes in animals and 5 to 7 minutes in humans. Time to maximum plasma concentration (Tmax) and mean residence time are both extremely short, leading to a transient appearance of HFA-134a in the blood with no evidence of accumulation.
Pharmacokinetics In a single-dose bioavailability study which enrolled six healthy, male volunteers, transient low albuterol levels (close to the lower limit of quantitation) were observed after administration of two puffs from both Albuterol Sulfate Inhalation Aerosol and a CFC 11/12 propelled albuterol inhaler. No formal pharmacokinetic analyses were possible for either treatment, but systemic albuterol levels appeared similar.
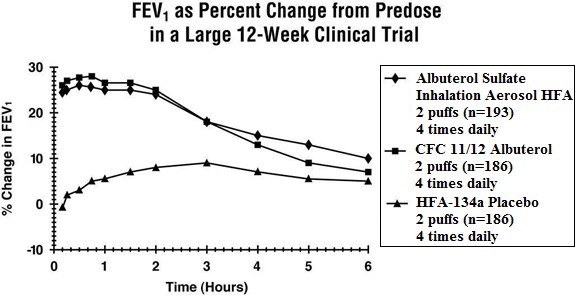
Clinical Trials In a 12-week, randomized, double-blind, double-dummy, active- and placebo-controlled trial, 565 patients with asthma were evaluated for the bronchodilator efficacy of Albuterol Sulfate Inhalation Aerosol (193 patients) in comparison to a CFC 11/12 propelled albuterol inhaler (186 patients) and an HFA-134a placebo inhaler (186 patients).
Serial FEV 1 measurements (shown below as percent change from test-day baseline) demonstrated that two inhalations of Albuterol Sulfate Inhalation Aerosol produced significantly greater improvement in pulmonary function than placebo and produced outcomes which were clinically comparable to a CFC 11/12 propelled albuterol inhaler.
The mean time to onset of a 15% increase in FEV 1 was 6 minutes and the mean time to peak effect was 50 to 55 minutes. The mean duration of effect as measured by a 15% increase in FEV 1 was 3 hours. In some patients, duration of effect was as long as 6 hours.
In another clinical study in adults, two inhalations of Albuterol Sulfate Inhalation Aerosol taken 30 minutes before exercise prevented exercise-induced bronchospasm as demonstrated by the maintenance of FEV 1 within 80% of baseline values in the majority of patients.
In a 4-week, randomized, open-label trial, 63 children, 4 to 11 years of age, with asthma were evaluated for the bronchodilator efficacy of Albuterol Sulfate Inhalation Aerosol (33 pediatric patients) in comparison to a CFC 11/12 propelled albuterol inhaler (30 pediatric patients).
Serial FEV 1 measurements as percent change from test-day baseline demonstrated that two inhalations of Albuterol Sulfate Inhalation Aerosol produced outcomes which were clinically comparable to a CFC 11/12 propelled albuterol inhaler.
The mean time to onset of a 12% increase in FEV 1 for Albuterol Sulfate Inhalation Aerosol was 7 minutes and the mean time to peak effect was approximately 50 minutes. The mean duration of effect as measured by a 12% increase in FEV 1 was 2.3 hours. In some pediatric patients, duration of effect was as long as 6 hours.
In another clinical study in pediatric patients, two inhalations of Albuterol Sulfate Inhalation Aerosol taken 30 minutes before exercise provided comparable protection against exercise-induced bronchospasm as a CFC 11/12 propelled albuterol inhaler.
HOW SUPPLIED
Albuterol Sulfate Inhalation Aerosol is supplied as a pressurized aluminum canister, with an attached dose indicator, a yellow plastic actuator and orange dust cap each in boxes of one. Each actuation delivers 120 mcg of albuterol sulfate from the valve and 108 mcg of albuterol sulfate from the mouthpiece (equivalent to 90 mcg of albuterol base). Canisters with a labeled net weight of 6.7 g contain 200 inhalations (NDC 0781-7296-85).
Rx only. Store between 15° to 25°C (59° to 77°F). Store the inhaler with the mouthpiece down. For best results, canister should be at room temperature before use.
SHAKE WELL BEFORE USING.
The yellow actuator supplied with Albuterol Sulfate Inhalation Aerosol should not be used with any other product canisters, and actuator from other products should not be used with a Albuterol Sulfate Inhalation Aerosol canister. The correct amount of medication in each canister cannot be assured after 200 actuations and when the dose indicator display window shows zero, even though the canister is not completely empty the canister is not completely empty. The canister should be discarded when the labeled number of actuations have been used.
WARNING: Avoid spraying in eyes. Contents under pressure. Do not puncture or incinerate. Exposure to temperatures above 120°F may cause bursting. Keep out of reach of children.
Albuterol Sulfate Inhalation Aerosol does not contain chlorofluorocarbons (CFCs) as the propellant.
Developed and Manufactured by:
Kindeva Drug Delivery L.P.
Northridge, CA 91324, USA
or
Kindeva Drug Delivery Limited
Loughborough, UK
Distributed by: Sandoz Inc. Princeton, NJ 08540
Copyright © 1996, 2011, 2012, 2017, 2018, 2020, 2025 All rights reserved.
The trademarks depicted in this piece are owned by their respective companies.
Revised: 03/2025 3200001123
INSTRUCTIONS FOR USE Albuterol Sulfate (al-BYOO-ter-ole) Inhalation Aerosol with Dose Indicator
Read this Instructions for Use before you start using Albuterol Sulfate Inhalation Aerosol and eachtime you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or treatment. Your doctor should show you how your child should use Albuterol Sulfate Inhalation Aerosol.
Important Information:
- Albuterol Sulfate Inhalation Aerosol is for oral inhalation use only.
- Take Albuterol Sulfate Inhalation Aerosol exactly as your doctor tells you to.
Albuterol Sulfate Inhalation Aerosol comes as a canister with a dose indicator. The dose indicator is located on the top of the canister that fits into an actuator (See Figure A ). The dose indicator display window will show you how many puffs of medicine you have left. A puff of medicine is released each time you press the center of the dose indicator.
- Do not use the Albuterol Sulfate Inhalation Aerosol actuator with a canister of medicine from any other inhaler.
- Do not use the Albuterol Sulfate Inhalation Aerosol canister with an actuator from any other inhaler.
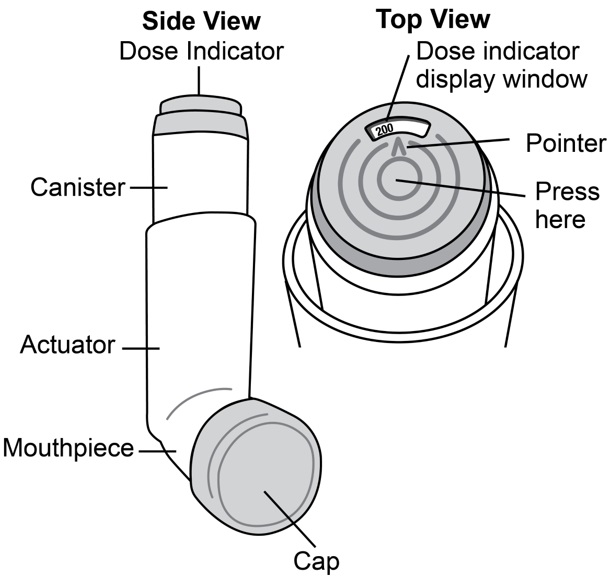
Figure A
Before you use Albuterol Sulfate Inhalation Aerosol for the first time make sure that the pointer on the dose indicator is pointing to the right of the “200” inhalation mark in the dose indicator display window (See Figure A ) .
Each canister of Albuterol Sulfate Inhalation Aerosol contains 200 puffs of medicine. This does not include the sprays of medicine used for priming your inhaler.
- The dose indicator display window will continue to move after every 10 puffs.
- The number in the dose indicator display window will continue to change after every 20 puffs.
- The color in the dose indicator display window will change to red, as shown in the shaded area, when there are only 20 puffs of medicine left in your inhaler (See Figure B ) . This is when you need to refill your prescription or ask your doctor if you need another prescription for Albuterol Sulfate Inhalation Aerosol.
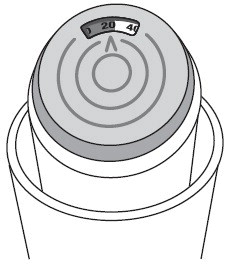
Figure B
Before using your Albuterol Sulfate Inhalation Aerosol for the first time, you should prime your inhaler. If you do not use your Albuterol Sulfate Inhalation Aerosol for more than 2 weeks , you should re-prime it before use.
- Remove the cap from the mouthpiece ( See Figure C ). Check inside the mouthpiece for objects before use.
- Make sure the canister is fully inserted into the actuator.
- Hold the inhaler in an upright position away from your face and shake the inhaler well .
- Press down fully on the center of the dose indicator to release a spray of medicine. You may hear a soft click from the dose indicator as it counts down during use.
- Repeat the priming step 3 more times to release a total of 4 sprays of medicine . Shake the inhaler well before each priming spray.
- After the 4 priming sprays, the dose indicator should be pointing to 200. There are now 200 puffs of medicine left in the canister.
- Your inhaler is now ready to use.
Using your Albuterol Sulfate Inhalation Aerosol inhaler:
Step 1: Shake the inhaler well before each use. Remove the cap from the mouthpiece ( See Figure C ). Check inside the mouthpiece for objects before use. Make sure the canister is fully inserted into the actuator.
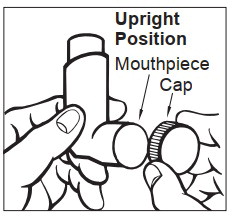
Figure C
Step 2: Breathe out as fully as you comfortably can through your mouth. Hold the inhaler in the upright position with the mouthpiece pointing towards you and place the mouthpiece fully into the mouth ( See Figure D ). Close your lips around the mouthpiece.
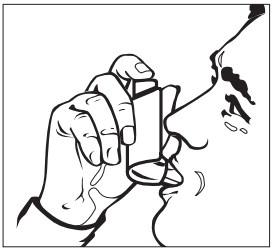
Figure D
Step 3: While breathing in deeply and slowly, press down on the center of the dose indicator with your index finger until the canister stops moving in the actuator and a puff of medicine has been released ( See Figure D ). Then stop pressing the dose indicator.
Step 4: Hold your breath as long as you comfortably can, up to 10 seconds. Remove the inhaler from your mouth, and then breathe out.
Step 5: If your doctor has prescribed additional puffs of Albuterol Sulfate Inhalation Aerosol , wait 1 minute then shake the inhaler well. Repeat steps 3 through 5 in the section “ Using your Albuterol Sulfate Inhalation Aerosol inhaler ”.
Step 6: Replace the cap right away after use.
Cleaning your Albuterol Sulfate Inhalation Aerosol inhaler:
It is very important that you keep the mouthpiece clean so that medicine will not build up and block the spray through the mouthpiece. Clean the mouthpiece 1 time each week or if your mouthpiece becomes blocked ( See Figure F ).
Step 1: Remove the canister from the actuator and take the cap off the mouthpiece. Do not clean the metal canister or let it get wet.
Step 2: Wash the mouthpiece through the top and bottom with warm running water for 30 seconds ( See Figure E ).
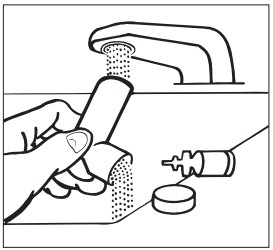
Figure E
Step 3 : Shake off as much water from the mouthpiece as you can.
Step 4: Look in the mouthpiece to make sure any medicine buildup has been completely washed away. If the mouthpiece is blocked with buildup, little to no medicine will come out of the mouthpiece ( See Figure F ). If there is any buildup, repeat Steps 2 through 4 in the section “ Cleaning your Albuterol Sulfate Inhalation Aerosol inhaler ”.
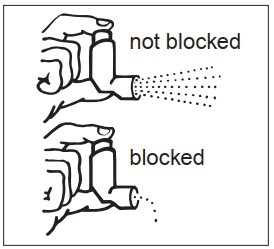
Figure F
Step 5: Let the mouthpiece air-dry such as overnight ( Figure G ). Do not put the canister back into the actuator if it is still we
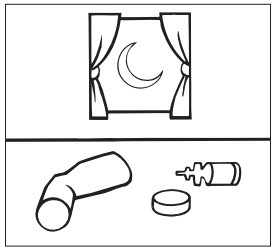
Figure G
Step 6: When the mouthpiece is dry, put the canister back in the actuator and put the cap on the mouthpiece.
Note: If you need to use your Albuterol Sulfate Inhalation Aerosol inhaler before it is completely dry, put the canister back in the actuator and shake the inhaler well. Press down on the center of the dose indicator 2 times to release a total of 2 sprays into the air, away from your face. Take your dose as prescribed then clean and air-dry your inhaler as described in the section “ Cleaning your Albuterol Sulfate Inhalation Aerosol inhaler ”.
How should I store Albuterol Sulfate Inhalation Aerosol?
- Store Albuterol Sulfate Inhalation Aerosol at room temperature between 59°F and 77°F (15°C and 25°C).
- Store with the mouthpiece down.
- Avoid exposing Albuterol Sulfate Inhalation Aerosol to extreme heat and cold.
- Do not puncture or burn the canister.
- Keep your Albuterol Sulfate Inhalation Aerosol inhaler and all medicines out of the reach of children.
Developed and Manufactured by: Kindeva Drug Delivery L.P. Northridge, CA 91324, USA
or
Kindeva Drug Delivery Limited
Loughborough, UK
Distributed by: Sandoz Inc. Princeton, NJ 08540
Copyright © 1996, 2011, 2012, 2017, 2018, 2020, 2025
All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 03/2025 3200001123
Mechanism of Action In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta 2 -adrenergic receptors compared with isoproterenol. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors on bronchial smooth muscle, data indicate that there is a population of beta 2 -receptors in the human heart existing in a concentration between 10% and 50% of cardiac beta-adrenergic receptors. The precise function of these receptors has not been established. (See WARNINGS, Cardiovascular Effects section.)
Activation of beta 2 -adrenergic receptors on airway smooth muscle leads to the activation of adenylcyclase and to an increase in the intracellular concentration of cyclic-3',5'-adenosine monophosphate (cyclic AMP). This increase of cyclic AMP leads to the activation of protein kinase A, which inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in relaxation. Albuterol relaxes the smooth muscles of all airways, from the trachea to the terminal bronchioles. Albuterol acts as a functional antagonist to relax the airway irrespective of the spasmogen involved, thus protecting against all bronchoconstrictor challenges. Increased cyclic AMP concentrations are also associated with the inhibition of release of mediators from mast cells in the airway.
Albuterol has been shown in most clinical trials to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes.
Preclinical Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood-brain barrier and reaches brain concentrations amounting to approximately 5% of the plasma concentrations. In structures outside the blood-brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those in the whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta 2 -agonist and methylxanthines were administered concurrently. The clinical significance of these findings is unknown.
Propellant HFA-134a is devoid of pharmacological activity except at very high doses in animals (380-1300 times the maximum human exposure based on comparisons of AUC values), primarily producing ataxia, tremors, dyspnea, or salivation. These are similar to effects produced by the structurally related chlorofluorocarbons (CFCs), which have been used extensively in metered dose inhalers.
In animals and humans, propellant HFA-134a was found to be rapidly absorbed and rapidly eliminated, with an elimination half-life of 3 to 27 minutes in animals and 5 to 7 minutes in humans. Time to maximum plasma concentration (Tmax) and mean residence time are both extremely short, leading to a transient appearance of HFA-134a in the blood with no evidence of accumulation.