Get your patient on Alprazolam xr - Alprazolam tablet, Extended Release (Alprazolam)
Alprazolam xr - Alprazolam tablet, Extended Release prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs for patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation [see Warnings and Precautions (5.1) , Drug Interactions (7.1) ] .
- The use of benzodiazepines, including alprazolam XR, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing alprazolam XR and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction [see Warnings and Precautions (5.2) ] .
- The continued use of benzodiazepines, including alprazolam XR, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of alprazolam XR after continued use may precipitate acute withdrawal reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue alprazolam XR or reduce the dosage [see Dosage and Administration (2.2) , Warnings and Precautions (5.3) ] .
Warnings and Precautions (5.8 ) 1/2023
INDICATIONS AND USAGE
Alprazolam XR is indicated for the treatment of panic disorder with or without agoraphobia, in adults.
DOSAGE AND ADMINISTRATION
- Recommended starting oral dosage is 0.5 mg to 1 mg once daily (preferably in the morning). Depending on the response, the dose may be increased at intervals of 3 to 4 days in increments of no more than 1 mg daily. (2.1 )
- Recommended total daily dosage is 3 mg to 6 mg daily. (2.1 )
- Swallow tablets whole; do not divide, crush, or chew. (2.1 )
- When tapering, decrease dosage by no more than 0.5 mg every 3 days. Some patients may require an even slower dosage reduction. (2.2 , 5.2 )
- See the Full Prescribing Information for the recommended dosage in geriatric patients, patients with hepatic impairment, and with use with ritonavir. (2.3 , 2.4 , 2.5 )
Recommended Dosage
Administer alprazolam XR orally once daily, preferably in the morning. Swallow tablets whole; do not divide, crush, or chew.
The recommended starting oral dosage for alprazolam XR is 0.5 mg to 1 mg once daily. Depending on the response, the dosage may be adjusted at intervals of every 3 to 4 days in increments of no more than 1 mg daily. The recommended dosage range is 3 mg to 6 mg once daily.
Controlled trials of alprazolam XR for the treatment of panic disorder included dosages in the range of 1 mg to 10 mg per day. Most patients showed a response in the dosage range of 3 mg to 6 mg per day. Occasional patients required as much as 10 mg per day.
The longer-term efficacy of alprazolam XR has not been systematically evaluated. If alprazolam XR is used for periods longer than 8 weeks, the healthcare provider should periodically reassess the usefulness of the drug for the individual patient.
After a period of extended freedom from panic attacks, a carefully supervised tapered discontinuation may be attempted, but there is evidence that this may often be difficult to accomplish without recurrence of symptoms and/or the manifestation of withdrawal phenomena [see Dosage and Administration (2.2) , Warnings and Precautions (5.2) ] .
Discontinuation or Dosage Reduction of Alprazolam XR
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue alprazolam XR or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly [see Warnings and Precautions (5.3) , Drug Abuse and Dependence (9.3) ] .
Reduce the dosage by no more than 0.5 mg every three days. Some patients may benefit from an even more gradual discontinuation. Some patients may prove resistant to all discontinuation regimens.
In a controlled postmarketing discontinuation study of panic disorder patients which compared the recommended taper schedule with a slower taper schedule, no difference was observed between the groups in the proportion of patients who tapered to zero dose; however, the slower schedule was associated with a reduction in symptoms associated with a withdrawal syndrome.
Dosage Recommendations in Geriatric Patients
In geriatric patients, the recommended starting dosage of alprazolam XR is 0.5 mg once daily. This may be gradually increased if needed and tolerated. Geriatric patients may be sensitive to the effects of benzodiazepines [see Use in Specific Populations (8.5) , Clinical Pharmacology (12.3) ] .
Dosage Recommendations in Patients with Hepatic Impairment
In patients with hepatic impairment, the recommended starting dosage of alprazolam XR is 0.5 mg once daily. This may be gradually increased if needed and tolerated [see Use in Specific Populations (8.6) , Clinical Pharmacology (12.3) ] .
Dosage Modifications for Drug Interactions
Alprazolam XR should be reduced to half of the recommended dosage when a patient is started on ritonavir and alprazolam XR together, or when ritonavir is added to a patient treated with alprazolam XR. Increase alprazolam XR dosage to the target dose after 10 to 14 days of dosing ritonavir and alprazolam XR together. It is not necessary to reduce alprazolam XR dosage in patients who have been taking ritonavir for more than 10 to 14 days.
Alprazolam XR is contraindicated with concomitant use of all strong CYP3A inhibitors, except ritonavir [see Contraindications (4) , Warnings and Precautions (5.5) , Drug Interactions (7.1) ] .
Switching Patients from Alprazolam Tablets to Alprazolam XR Tablets
Patients who are currently being treated with divided doses of alprazolam may be switched to alprazolam XR at the same total daily dose taken once daily. If the clinical response after switching is inadequate, titrate the dosage as outlined above.
DOSAGE FORMS AND STRENGTHS
Alprazolam XR Extended-Release Tablets are available as:
- 0.5 mg: white, pentagonal shaped tablets debossed with an "G" on one side and "0.5" on the other side
- 1 mg: yellow, square shaped tablets debossed with an "G" on one side and "1" on the other side
- 2 mg: blue, round shaped tablets debossed with an "G" on one side and "2" on the other side
- 3 mg: green, triangular shaped tablets debossed with an "G" on one side and "3" on the other side
USE IN SPECIFIC POPULATIONS
Lactation: Breastfeeding not recommended. (8.2 )
Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to psychiatric medications, including alprazolam XR, during pregnancy. Healthcare providers are encouraged to register patients by calling the National Pregnancy Registry for Psychiatric Medications at 1-866-961-2388 or visiting online at https://womensmentalhealth.org/pregnancyregistry/.
Risk Summary
Neonates born to mothers using benzodiazepines late in pregnancy have been reported to experience symptoms of sedation and/or neonatal withdrawal [see Warnings and Precautions (5.8) , and Clinical Considerations ] . Available data from published observational studies of pregnant women exposed to benzodiazepines do not report a clear association with benzodiazepines and major birth defects (see Data ) .
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and of miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Benzodiazepines cross the placenta and may produce respiratory depression, hypotonia, and sedation in neonates. Monitor neonates exposed to alprazolam XR during pregnancy or labor for signs of sedation, respiratory depression, hypotonia, and feeding problems. Monitor neonates exposed to alprazolam XR during pregnancy for signs of withdrawal. Manage these neonates accordingly [see Warnings and Precautions (5.8) ] .
Data
Human Data
Published data from observational studies on the use of benzodiazepines during pregnancy do not report a clear association with benzodiazepines and major birth defects. Although early studies reported an increased risk of congenital malformations with diazepam and chlordiazepoxide, there was no consistent pattern noted. In addition, the majority of recent case-control and cohort studies of benzodiazepine use during pregnancy, which were adjusted for confounding exposures to alcohol, tobacco, and other medications, have not confirmed these findings.
Lactation
Risk Summary
Limited data from published literature reports the presence of alprazolam in human breast milk. There are reports of sedation, poor feeding and poor weight gain in infants exposed to benzodiazepines through breast milk. The effects of alprazolam on lactation are unknown.
Because of the potential for serious adverse reactions, including sedation and withdrawal symptoms in breastfed infants, advise patients that breastfeeding is not recommended during treatment with alprazolam XR.
Pediatric Use
Safety and effectiveness of alprazolam XR have not been established in pediatric patients.
Geriatric Use
Alprazolam XR‑treated geriatric patients had higher plasma concentrations of alprazolam (due to reduced clearance) compared to younger adults receiving the same doses. Therefore, dosage reduction of alprazolam XR is recommended in geriatric patients [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3) ] .
Hepatic Impairment
Patients with alcoholic liver disease exhibit a longer elimination half-life (19.7 hours), compared to healthy subjects (11.4 hours). This may be caused by decreased clearance of alprazolam in patients with alcoholic liver disease. Dosage reduction of alprazolam XR is recommended in patients with hepatic impairment [see Dosage and Administration (2.4) , Clinical Pharmacology (12.3) ] .
CONTRAINDICATIONS
Alprazolam XR is contraindicated in patients:
- with known hypersensitivity to alprazolam or other benzodiazepines. Angioedema has been reported [see Adverse Reactions (6.2) ] .
- taking strong cytochrome P450 3A (CYP3A) inhibitors (e.g., ketoconazole, itraconazole), except ritonavir [see Dosage and Administration (2.5) , Warnings and Precautions (5.5) , Drug Interactions (7.1) ] .
WARNINGS AND PRECAUTIONS
- Effects on Driving and Operating Machinery: Patients receiving
- alprazolam XR should be cautioned against operating machinery or driving a motor vehicle, as well as avoiding concomitant use of alcohol and other central nervous system (CNS) depressant drugs. (5.4 )
- Patients with Depression: Exercise caution in patients with signs or symptoms of depression. Prescribe the least number of tablets feasible to avoid intentional overdosage. (5.6 )
- Neonatal Sedation and Withdrawal Syndrome: Alprazolam XR use during pregnancy can result in neonatal sedation and/or neonatal withdrawal. (5.8 , 8.1 )
Risks from Concomitant Use with Opioids
Concomitant use of benzodiazepines, including alprazolam XR, and opioids may result in profound sedation, respiratory depression, coma, and death. Because of these risks, reserve concomitant prescribing of these drugs in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioids alone. If a decision is made to prescribe alprazolam XR concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. In patients already receiving an opioid analgesic, prescribe a lower initial dose of alprazolam XR than indicated in the absence of an opioid and titrate based on clinical response. If an opioid is initiated in a patient already taking alprazolam XR, prescribe a lower initial dose of the opioid and titrate based upon clinical response.
Advise both patients and caregivers about the risks of respiratory depression and sedation when alprazolam XR is used with opioids. Advise patients not to drive or operate heavy machinery until the effects of concomitant use with the opioid have been determined [see Drug Interactions (7.1) ] .
Abuse, Misuse, and Addiction
The use of benzodiazepines, including alprazolam XR, exposes users to the risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines often (but not always) involve the use of doses greater than the maximum recommended dosage and commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes, including respiratory depression, overdose, or death [see Drug Abuse and Dependence (9.2) ] .
Before prescribing alprazolam XR and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (e.g., using a standardized screening tool). Use of alprazolam XR, particularly in patients at elevated risk, necessitates counseling about the risks and proper use of alprazolam XR along with monitoring for signs and symptoms of abuse, misuse, and addiction. Prescribe the lowest effective dosage; avoid or minimize concomitant use of CNS depressants and other substances associated with abuse, misuse, and addiction (e.g., opioid analgesics, stimulants); and advise patients on the proper disposal of unused drug. If a substance use disorder is suspected, evaluate the patient and institute (or refer them for) early treatment, as appropriate.
Dependence and Withdrawal Reactions
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue alprazolam XR or reduce the dosage (a patient-specific plan should be used to taper the dose) [see Dosage and Administration (2.3) ] .
Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages, and those who have had longer durations of use.
Acute Withdrawal Reactions
The continued use of benzodiazepines, including alprazolam XR, may lead to clinically significant physical dependence. Abrupt discontinuation or rapid dosage reduction of alprazolam XR after continued use, or administration of flumazenil (a benzodiazepine antagonist) may precipitate acute withdrawal reactions, which can be life-threatening (e.g., seizures) [see Drug Abuse and Dependence (9.3) ] .
Protracted Withdrawal Syndrome
In some cases, benzodiazepine users have developed a protracted withdrawal syndrome with withdrawal symptoms lasting weeks to more than 12 months [see Drug Abuse and Dependence (9.3) ] .
Certain adverse clinical events, some life-threatening, are a direct consequence of physical dependence to alprazolam XR. These include a spectrum of withdrawal symptoms; the most important is seizure [see Drug Abuse and Dependence (9.3) ] . Even after relatively short-term use at doses of ≤ 4 mg/day, there is some risk of dependence. Spontaneous reporting system data suggest that the risk of dependence and its severity appear to be greater in patients treated with doses greater than 4 mg/day and for long periods (more than 12 weeks). However, in a controlled postmarketing discontinuation study of panic disorder patients who received alprazolam, the duration of treatment (3 months compared to 6 months) had no effect on the ability of patients to taper to zero dose. In contrast, patients treated with doses of alprazolam greater than 4 mg/day had more difficulty tapering to zero dose than those treated with less than 4 mg/day.
In a controlled clinical trial in which 63 patients were randomized to alprazolam and where withdrawal symptoms were specifically sought, the following were identified as symptoms of withdrawal: heightened sensory perception, impaired concentration, dysosmia, clouded sensorium, paresthesias, muscle cramps, muscle twitch, diarrhea, blurred vision, appetite decrease, and weight loss. Other symptoms, such as anxiety and insomnia, were frequently seen during discontinuation, but it could not be determined if they were due to return of illness, rebound, or withdrawal.
Interdose Symptoms
Early morning anxiety and emergence of anxiety symptoms between doses of alprazolam have been reported in patients with panic disorder taking prescribed maintenance doses. These symptoms may reflect the development of tolerance or a time interval between doses which is longer than the duration of clinical action of the administered dose. In either case, it is presumed that the prescribed dose is not sufficient to maintain plasma levels above those needed to prevent relapse, rebound, or withdrawal symptoms over the entire course of the interdosing interval.
Effects on Driving and Operating Machinery
Because of its CNS depressant effects, patients receiving alprazolam XR should be cautioned against engaging in hazardous occupations or activities requiring complete mental alertness such as operating machinery or driving a motor vehicle. For the same reason, patients should be cautioned about the concomitant use of alcohol and other CNS depressant drugs during treatment with alprazolam XR [see Drug Interactions (7.1) ] .
Interaction with Drugs that Inhibit Metabolism via Cytochrome P450 3A
The initial step in alprazolam metabolism is hydroxylation catalyzed by cytochrome P450 3A (CYP3A). Drugs that inhibit this metabolic pathway may have a profound effect on the clearance of alprazolam.
Strong CYP3A Inhibitors
Alprazolam XR is contraindicated in patients receiving strong inhibitors of CYP3A such as azole antifungal agents [see Contraindications (4) ] . Ketoconazole and itraconazole have been shown in vivo to increase plasma alprazolam concentrations 3.98 fold and 2.70 fold, respectively.
Dosage adjustment is necessary when alprazolam XR and ritonavir are initiated concomitantly or when ritonavir is added to a stable dosage of alprazolam XR [see Dosage and Administration (2.5) , Drug Interactions (7.1) ].
Drugs demonstrated to be CYP3A inhibitors on the basis of clinical studies involving alprazolam: nefazodone, fluvoxamine, and cimetidine [see Drug Interaction (7.1) , Clinical Pharmacology (12.3) ] . Use caution and consider dose reduction of alprazolam XR, as appropriate, during co-administration with these drugs.
Patients with Depression
Benzodiazepines may worsen depression. Panic disorder has been associated with primary and secondary major depressive disorders and increased reports of suicide among untreated patients. Consequently, appropriate precautions (e.g., limiting the total prescription size and increased monitoring for suicidal ideation) should be considered in patients with depression.
Mania
Episodes of hypomania and mania have been reported in association with the use of alprazolam XR in patients with depression [see Adverse Reactions (6.1) ] .
Ne onatal Sedation and Withdrawal Syndrome
Use of alprazolam XR late in pregnancy can result in sedation (respiratory depression, lethargy, hypotonia) and/or withdrawal symptoms (hyperreflexia, irritability, restlessness, tremors, inconsolable crying, and feeding difficulties) in the neonate [see Use in Specific Populations (8.1) ] . Monitor neonates exposed to alprazolam XR during pregnancy or labor for signs of sedation and monitor neonates exposed to alprazolam XR during pregnancy for signs of withdrawal; manage these neonates accordingly.
Risks in Patients with Impaired Respiratory Function
There have been reports of death in patients with severe pulmonary disease shortly after the initiation of treatment with alprazolam. Closely monitor patients with impaired respiratory function. If signs and symptoms of respiratory depression, hypoventilation, or apnea occur, discontinue alprazolam XR.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks from Concomitant Use with Opioids [see Warnings and Precautions (5.1) ]
- Abuse, Misuse, and Addiction [see Warnings and Precautions (5.2) ]
- Dependence and Withdrawal Reactions [see Warnings and Precautions (5.3) ]
- Effects on Driving and Operating Machinery [see Warnings and Precautions (5.4) ]
- Patients with Depression [see Warnings and Precautions (5.7) ]
- Neonatal Sedation and Withdrawal Syndrome [see Warnings and Precautions (5.8) ]
- Risks in Patients with Impaired Respiratory Function [see Warnings and Precautions (5.9) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The information included in the section on Adverse Reactions Observed in Short-Term, Placebo-Controlled Trials with Alprazolam XR is based on pooled data of five 6- and 8-week placebo-controlled clinical studies in panic disorder.
Adverse Reactions Observed in Short-Term, Placebo-Controlled Trials of Alprazolam XR
Adverse Reactions Reported as Reasons for Discontinuation of Treatment in Placebo-Controlled Trials
Approximately 17% of the 531 patients who received alprazolam XR in placebo-controlled clinical trials for panic disorder had at least 1 adverse event that led to discontinuation compared to 8% of 349 placebo-treated patients. The most common events leading to discontinuation and considered to be drug-related (i.e., leading to discontinuation in at least 1% of the patients treated with alprazolam XR at a rate at least twice that of placebo) are shown in Table 1.
| n=number of patients | ||
Percentage of Patients Discontinuing Due to Adverse Reactions | ||
Alprazolam XR (n=531) | Placebo (n=349) | |
Nervous system disorders | ||
Sedation | 7.5 | 0.6 |
Somnolence | 3.2 | 0.3 |
Dysarthria | 2.1 | 0 |
Coordination abnormal | 1.9 | 0.3 |
Memory impairment | 1.5 | 0.3 |
General disorders/administration site conditions | ||
Fatigue | 1.7 | 0.6 |
Psychiatric disorders | ||
Depression | 2.5 | 1.2 |
Adverse Reactions Occurring at an Incidence of 1% or More Among Patients Treated with Alprazolam XR
Table 2 shows the incidence of adverse reactions that occurred during 6- and 8-week placebo-controlled trials in 1% or more of patients treated with alprazolam XR where the incidence in patients treated with alprazolam XR was greater than the incidence in placebo-treated patients. The most commonly observed adverse reactions in panic disorder patients treated with alprazolam XR (incidence of 5% or greater and at least twice the incidence in placebo patients) were: sedation, somnolence, memory impairment, dysarthria, coordination abnormal, ataxia, libido decreased.
| Alprazolam XR (n=531) | Placebo (n=349) | |
|---|---|---|
Nervous system disorders | ||
Sedation | 45% | 23% |
Somnolence | 23% | 6% |
Memory impairment | 15% | 7% |
Dysarthria | 11% | 3% |
Coordination abnormal | 9% | 1% |
Mental impairment | 7% | 6% |
Ataxia | 7% | 3% |
Disturbance in attention | 3% | 1% |
Balance impaired | 3% | 1% |
Dyskinesia | 2% | 1% |
Hypoesthesia | 1% | <1% |
Hypersomnia | 1% | 0% |
General disorders/administration site conditions | ||
Fatigue | 14% | 9% |
Lethargy | 2% | 1% |
Psychiatric disorders | ||
Depression | 12% | 9% |
Libido decreased | 6% | 2% |
Disorientation | 2% | 0% |
Confusion | 2% | 1% |
Depressed mood | 1% | <1% |
Metabolism and nutrition disorders | ||
Appetite increased | 7% | 6% |
Anorexia | 2% | 0% |
Gastrointestinal disorders | ||
Constipation | 8% | 4% |
Nausea | 6% | 3% |
Investigations | ||
Weight increased | 5 | 4 |
Injury, poisoning, and procedural complications | ||
Road traffic accident | 2% | 0% |
Reproductive system and breast disorders | ||
Dysmenorrhea | 4% | 3% |
Sexual dysfunction | 2% | 1% |
Musculoskeletal and connective tissue disorder | ||
Arthralgia Myalgia Pain in limb | 2% 2% 1% | 1% 1% 0% |
Respiratory, thoracic, and mediastinal disorders | ||
Dyspnea | 2% | 0% |
Other Adverse Reactions Observed During the Premarketing Evaluation of Alprazolam XR
Following is a list of other adverse reaction reported by 531 patients with panic disorder treated with alprazolam XR. Adverse reactions are further categorized by body system and listed in order of decreasing frequency according to the following definitions: those occurring in at least 1/100 patients (frequent); those occurring in less than 1/100 patients but at least 1/1000 patients (infrequent); those occurring in fewer than 1/1000 patients (rare).
Cardiac disorders: Frequent: palpitation; Infrequent: sinus tachycardia
Ear and labyrinth disorders: Frequent: vertigo; Infrequent : tinnitus, ear pain
Eye disorders: Frequent: blurred vision; Infrequent: mydriasis, photophobia
Gastrointestinal disorders: Frequent: diarrhea, vomiting, dyspepsia, abdominal pain; Infrequent: dysphagia, salivary hypersecretion
General disorders and administration site conditions: Frequent: malaise, weakness, chest pains; Infrequent: fall, pyrexia, thirst, feeling hot and cold, edema, feeling jittery, sluggishness, asthenia, feeling drunk, chest tightness, increased energy, feeling of relaxation, hangover, loss of control of legs, rigors
Musculoskeletal and connective tissue disorders: Frequent: back pain, muscle cramps, muscle twitching
Nervous system disorders: Frequent: headache, dizziness, tremor; Infrequent: amnesia, clumsiness, syncope, hypotonia, seizures, depressed level of consciousness, sleep apnea syndrome, sleep talking, stupor
Psychiatric system disorders: Frequent: irritability, insomnia, nervousness, derealization, libido increased, restlessness, agitation, depersonalization, nightmare; Infrequent: abnormal dreams, apathy, aggression, anger, bradyphrenia, euphoric mood, logorrhea, mood swings, dysphonia, hallucination, homicidal ideation, mania, hypomania, impulse control, psychomotor retardation, suicidal ideation
Renal and urinary disorders: Frequent: difficulty in micturition; Infrequent: urinary frequency, urinary incontinence
Respiratory, thoracic, and mediastinal disorders: Frequent: nasal congestion, hyperventilation; Infrequent: choking sensation, epistaxis, rhinorrhea
Skin and subcutaneous tissue disorders: Frequent: sweating increased; Infrequent: clamminess, rash, urticaria
Vascular disorders: Infrequent: hypotension
Discontinuation-Emergent Adverse Reactions Occurring at an Incidence of 5% or More Among Patients Treated with Alprazolam XR
Table 3 shows the incidence of discontinuation-emergent adverse reactions that occurred during short-term, placebo-controlled trials in 5% or more of patients treated with alprazolam XR where the incidence in patients treated with alprazolam XR was 2 times greater than the incidence in placebo-treated patients.
Alprazolam XR n=422 (%) | Placebo n=261(%) | |
Nervous system disorders | ||
Tremor | 28.2 | 10.7 |
Headache | 26.5 | 12.6 |
Hypoesthesia | 7.8 | 2.3 |
Paresthesia | 7.1 | 2.7 |
Psychiatric disorders | ||
Insomnia | 24.2 | 9.6 |
Nervousness | 21.8 | 8.8 |
Depression | 10.9 | 5.0 |
Derealization | 8.0 | 3.8 |
Anxiety | 7.8 | 2.7 |
Depersonalization | 5.7 | 1.9 |
Gastrointestinal disorders | ||
Diarrhea | 12.1 | 3.1 |
Respiratory, thoracic and mediastinal disorders | ||
Hyperventilation | 8.5 | 2.7 |
Metabolism and nutrition disorders | ||
Appetite decreased | 9.5 | 3.8 |
Musculoskeletal and connective tissue disorders | ||
Muscle twitching | 7.4 | 2.7 |
Vascular disorders | ||
Hot flushes | 5.9 | 2.7 |
There have also been reports of withdrawal seizures upon rapid decrease or abrupt discontinuation of alprazolam [see Warning and Precautions (5.2), Drug Abuse and Dependence (9.3)] .
Paradoxical reactions such as stimulation, increased muscle spasticity, sleep disturbances, hallucinations, and other adverse behavioral effects such as agitation, rage, irritability, and aggressive or hostile behavior have been reported rarely. In many of the spontaneous case reports of adverse behavioral effects, patients were receiving other CNS drugs concomitantly and/or were described as having underlying psychiatric conditions. Should any of the above events occur, alprazolam should be discontinued. Isolated published reports involving small numbers of patients have suggested that patients who have borderline personality disorder, a prior history of violent or aggressive behavior, or alcohol or substance abuse may be at risk for such events. Instances of irritability, hostility, and intrusive thoughts have been reported during discontinuation of alprazolam in patients with posttraumatic stress disorder.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of alprazolam and/or alprazolam XR. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Endocrine disorders: Hyperprolactinemia
General disorders and administration site conditions: Edema peripheral
Hepatobiliary disorders: Hepatitis, hepatic failure, jaundice
Investigations: Liver enzyme elevations
Psychiatric disorders: Hypomania, mania
Reproductive system and breast disorders: Gynecomastia, galactorrhea, menstruation irregular
Skin and subcutaneous tissue disorders: Photosensitivity reaction, angioedema, Stevens-Johnson syndrome
DRUG INTERACTIONS
- Use with Opioids: Increase the risk of respiratory depression. (7.1 )
- Use with Other CNS Depressants: Produces additive CNS depressant effects. (7.1 )
- Use with Digoxin: Increase the risk of digoxin toxicity. (7.1 )
- Use with CYP3A Inhibitors (except ritonavir): Increase the risk of adverse reactions of alprazolam. (4 , 5.5 , 7.1 )
- Use with CYP3A Inducers: Increase the risk of reduced efficacy of alprazolam. (7.1 )
Drugs Having Clinically Important Interactions with Alprazolam XR
Table 4 includes clinically significant drug interactions with alprazolam XR [see Clinical Pharmacology (12.3) ] .
Opioids | |
Clinical implication | The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at gamma-aminobutyric acid (GABA A ) sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid‑related respiratory depression exists. |
Prevention or management | Limit dosage and duration of concomitant use of alprazolam XR and opioids, and monitor patients closely for respiratory depression and sedation [see Warnings and Precautions (5.1) ] . |
Examples | Morphine, buprenorphine, hydromorphone, oxymorphone, oxycodone, fentanyl, methadone, alfentanil, butorphanol, codeine, dihydrocodeine, meperidine, pentazocine, remifentanil, sufentanil, tapentadol, tramadol. |
CNS Depressants | |
Clinical implication | The benzodiazepines, including alprazolam, produce additive CNS depressant effects when coadministered with other CNS depressants. |
Prevention or management | Limit dosage and duration of alprazolam XR during concomitant use with CNS depressants [see Warnings and Precautions (5.3) ] . |
Examples | Psychotropic medications, anticonvulsants, antihistaminics, ethanol, and other drugs which themselves produce CNS depression. |
Strong Inhibitors of CYP3A (except ritonavir) | |
Clinical implication | Concomitant use of alprazolam XR with strong CYP3A inhibitors has a profound effect on the clearance of alprazolam, resulting in increased concentrations of alprazolam and increased risk of adverse reactions [see Clinical Pharmacology (12.3) ] . |
Prevention or management | Concomitant use of alprazolam XR with a strong CYP3A4 inhibitor (except ritonavir) is contraindicated [see Contraindications (4) , Warnings and Precautions (5.5) ] . |
Examples | Ketoconazole, itraconazole, clarithromycin |
Moderate or Weak Inhibitors of CYP3A | |
Clinical implication | Concomitant use of alprazolam XR with CYP3A inhibitors may increase the concentrations of alprazolam XR, resulting in increased risk of adverse reactions [see Clinical Pharmacology (12.3) ] . |
Prevention or management | Avoid use and consider appropriate dose reduction when alprazolam XR is coadministered with a moderate or weak CYP3A inhibitor [see Warnings and Precautions (5.5) ] . |
Examples | Nefazodone, fluvoxamine, cimetidine, erythromycin |
CYP3A Inducers | |
Clinical implication | Concomitant use of CYP3A inducers can increase alprazolam metabolism and therefore can decease plasma levels of alprazolam [see Clinical Pharmacology (12.3) ] . |
Prevention or management | Caution is recommended during coadministration with alprazolam. |
Examples | Carbamazepine, phenytoin |
Ritonavir | |
Clinical implication | Interactions involving ritonavir and alprazolam are complex and time dependent. Short term administration of ritonavir increased alprazolam exposure due to CYP3A4 inhibition. Following long term treatment of ritonavir (> 10-14 days), CYP3A4 induction offsets this inhibition. Alprazolam exposure was not meaningfully affected in the presence of ritonavir. |
Prevention or management | Reduce alprazolam XR dose when a patient is initiated with ritonavir and alprazolam XR concomitantly, or when ritonavir is added to a regimen where alprazolam XR is stabilized. Increase alprazolam XR dosage to the target dosage after 10 to 14 days of dosing ritonavir and alprazolam XR concomitantly. No dosage adjustment of alprazolam XR is necessary in patients receiving ritonavir for more than 10 to 14 days [see Dosage and Administration (2.5) ] . Concomitant use of alprazolam XR with a strong CYP3A inhibitor, except ritonavir, is contraindicated [see Contraindications (4) , Warnings and Precautions (5.5) ] . |
Digoxin | |
Clinical implication | Increased digoxin concentrations have been reported when alprazolam was given, especially in geriatric patients (>65 years of age). |
Prevention or management | In patients on digoxin therapy, measure serum digoxin concentrations before initiating alprazolam XR. Continue monitoring digoxin serum concentration and toxicity frequently. Reduce the digoxin dose if necessary. |
Drug/Laboratory Test Interactions
Although interactions between benzodiazepines and commonly employed clinical laboratory tests have occasionally been reported, there is no consistent pattern for a specific drug or specific test.
DESCRIPTION
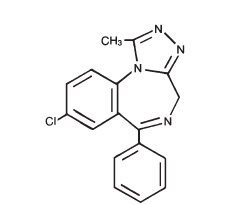
Alprazolam XR contains alprazolam which is a triazolo analog of the 1,4 benzodiazepine class of central nervous system-active compounds.
The chemical name of alprazolam is 8-chloro-1-methyl-6-phenyl-4 H - s -triazolo [4,3-α] [1,4] benzodiazepine. The molecular formula is C 17 H 13 ClN 4 which corresponds to a molecular weight of 308.76.
The structural formula is represented below:
Alprazolam is a white crystalline powder, which is soluble in methanol or ethanol but which has no appreciable solubility in water at physiological pH.
Each alprazolam XR extended-release tablet, for oral administration, contains 0.5 mg, 1 mg, 2 mg, or 3 mg of alprazolam. The inactive ingredients are colloidal silicon dioxide, hypromellose, lactose, and magnesium stearate. In addition, the 1 mg and 3 mg tablets contain D & C yellow No. 10 and the 2 mg and 3 mg tablets contain FD&C blue No. 2.
CLINICAL PHARMACOLOGY
Mechanism of Action
Alprazolam is a 1,4 benzodiazepine. Alprazolam exerts its effect for the treatment of panic disorder through binding to the benzodiazepine site of gamma‑aminobutyric acid-A (GABA A ) receptors in the brain and enhances GABA-mediated synaptic inhibition.
Pharmacokinetics
The pharmacokinetics of alprazolam and two of its major active metabolites (4-hydroxyalprazolam and α-hydroxyalprazolam) are linear, and concentrations are proportional up to 10 mg alprazolam XR given once daily.
Absorption
Following oral administration of alprazolam XR in the morning, peak plasma concentration of alprazolam (C max ) occurs in about 10 hours postdose. Compared to morning dosing, alprazolam C max increased by 30% and the T max decreased by an hour following dosing at night.
The mean absolute bioavailability of alprazolam following administration of alprazolam XR is approximately 90%, and the relative bioavailability compared to alprazolam is about 100%. The bioavailability and pharmacokinetics of alprazolam following administration of alprazolam XR are similar to that for alprazolam, with the exception of a slower rate of absorption.
Effect of Food
A high-fat meal given up to 2 hours before dosing with alprazolam XR increased the mean C max by about 25%. The effect of this meal on T max depended on the timing of the meal, with a reduction in T max by about 1/3 for subjects eating immediately before dosing and an increase in T max by about 1/3 for subjects eating 1 hour or more after dosing. The extent of exposure (AUC) and elimination half-life (t ½ ) were not affected by eating.
Distribution
The apparent volume of distribution of alprazolam is similar for alprazolam XR and alprazolam. Alprazolam is 80% bound to human serum protein, and albumin accounts for the majority of the binding.
Elimination
The mean plasma elimination half-life of alprazolam following administration of alprazolam XR ranges from 10.7 to 15.8 hours in healthy adults.
Metabolism
Alprazolam is extensively metabolized in humans, primarily by cytochrome P450 3A4 (CYP3A4), to two major active metabolites in the plasma: 4-hydroxyalprazolam and α-hydroxyalprazolam. The plasma circulation levels of the two active metabolites after both alprazolam XR and alprazolam are less than 10% and 4% of the parent, respectively. The reported relative potencies in benzodiazepine receptor binding experiments and in animal models of induced seizure inhibition are 0.20 and 0.66, respectively, for 4-hydroxyalprazolam and α-hydroxyalprazolam. The low concentrations and low potencies of 4-hydroxyalprazolam and α-hydroxyalprazolam indicate that they unlikely contribute much to the effects of alprazolam. A benzophenone derived from alprazolam is also found in humans. Their half-lives appear to be similar to that of alprazolam. The pharmacokinetic parameters at steady-state for the two hydroxylated metabolites of alprazolam (4-hydroxyalprazolam and α‑hydroxyalprazolam) were similar for alprazolam and alprazolam XR, indicating that the metabolism of alprazolam is not affected by absorption rate.
Excretion
Alprazolam and its metabolites are excreted primarily in the urine.
Specific Populations
Geriatric Patients
The mean T 1/2 of alprazolam was 16.3 hours (range: 9.0 to 26.9 hours) in healthy elderly subjects compared to 11.0 hours (range: 6.3 to 15.8 hours, n=16) in healthy adult subjects.
Obese Patients
The mean T 1/2 of alprazolam was 21.8 hours (range: 9.9 to 40.4 hours) in a group of obese subjects.
Patients with Hepatic Impairment
The mean T 1/2 of alprazolam was 19.7 hours (range: 5.8 to 65.3 hours) in patients with alcoholic liver disease.
Racial or Ethnic Groups
Maximal concentrations and T 1/2 of alprazolam are approximately 15% and 25% higher in Asians compared to Caucasians.
Smoking
Alprazolam concentrations may be reduced by up to 50% in smokers compared to non-smokers.
Drug Interaction Studies
In Vivo Studies
Most of the interactions that have been documented with alprazolam are with drugs that modulate CYP3A4 activity.
Compounds that are inhibitors or inducers of CYP3A would be expected to increase or decrease plasma alprazolam concentrations, respectively. Drug products that have been studied in vivo, along with their effect on increasing alprazolam AUC, are as follows: ketoconazole, 3.98 fold; itraconazole, 2.66 fold; nefazodone, 1.98 fold; fluvoxamine, 1.96 fold; and erythromycin, 1.61 fold [see Contraindications (4), Warnings and Precautions (5.5), Drug Interactions (7.2)] . Other studied drugs include:
Cimetidine: Coadministration of cimetidine increased the maximum plasma concentration of alprazolam by 82%, decreased clearance by 42%, and increased T 1/2 by 16%.
Fluoxetine: Coadministration of fluoxetine with alprazolam increased the maximum plasma concentration of alprazolam by 46%, decreased clearance by 21%, increased T 1/2 by 17%, and decreased measured psychomotor performance.
Oral Contraceptives: Coadministration of oral contraceptives increased the maximum plasma concentration of alprazolam by 18%, decreased clearance by 22%, and increased T 1/2 by 29%.
Carbamazepine: The oral clearance of alprazolam (given in a 0.8 mg single dose) was increased from 0.90±0.21 mL/min/kg to 2.13±0.54 mL/min/kg and the elimination T 1/2 was shortened (from 17.1±4.9 to 7.7 ±1.7 hour) following administration of 300 mg per day carbamazepine for 10 days [see Drug Interactions (7.2)] . However, the carbamazepine dose used in this study was fairly low compared to the recommended doses (1000‑1200 mg per day); the effect at usual carbamazepine doses is unknown.
Ritonavir: Interactions involving HIV protease inhibitors (e.g., ritonavir) and alprazolam are complex and time dependent. Short-term low doses of ritonavir (4 doses of 200 mg) increased mean AUC of alprazolam by about 2.5-fold, and did not significantly affect C max of alprazolam. The elimination T 1/2 was prolonged (30 hours versus 13 hours). However, upon extended exposure to ritonavir (500 mg, twice daily for 10 days), CYP3A induction offset this inhibition. Alprazolam AUC and C max was reduced by 12% and 16%, respectively, in the presence of ritonavir. The elimination T 1/2 of alprazolam was not significantly changed [see Warnings and Precautions (5.5) ] .
Sertraline: A single dose of alprazolam 1 mg and steady state dose of sertraline (50 to 150 mg per day) did not reveal any clinically significant changes in the pharmacokinetics of alprazolam.
Imipramine and Desipramine: The steady state plasma concentrations of imipramine and desipramine have been reported to be increased an average of 31% and 20%, respectively, by the concomitant administration of alprazolam in doses up to 4 mg per day.
Warfarin: Alprazolam did not affect the prothrombin or plasma warfarin levels in male volunteers administered sodium warfarin orally.
In Vitro Studies
Data from in vitro studies of alprazolam suggest a possible drug interaction of alprazolam with paroxetine. The ability of alprazolam to induce human hepatic enzyme systems has not been determined.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No evidence of carcinogenic potential was observed in rats or mice administered alprazolam for 2 years at doses up to 30 and 10 mg/kg/day, respectively. These doses are 29 times and 4.8 times the maximum recommended human dose of 10 mg/day based on mg/m 2 body surface area, respectively.
Mutagenesis
Alprazolam was negative in the in vitro Ames bacterial reverse mutation assay and DNA Damage/Alkaline Elution Assay and in vivo rat micronucleus genetic toxicology assays.
Impairment of Fertility
Alprazolam produced no impairment of fertility in rats at doses up to 5 mg/kg per day, which is approximately 5 times the maximum recommended human dose of 10 mg per day based on mg/m 2 body surface area.
Animal Toxicology and/or Pharmacology
When rats were treated with alprazolam at oral doses of 3 mg/kg, 10 mg/kg, and 30 mg/kg per day (3 to 29 times the maximum recommended human dose based on mg/m 2 body surface area) for 2 years, a tendency for a dose related increase in the number of cataracts was observed in females and a tendency for a dose related increase in corneal vascularization was observed in males. These lesions did not appear until after 11 months of treatment.
CLINICAL STUDIES
The efficacy of alprazolam XR in the treatment of panic disorder in adults was established in two 6-week, flexible-dose, placebo-controlled studies in adult patients meeting DSM-III criteria for panic disorder. In these studies, patients were treated with alprazolam XR in a dose range of 1 mg to 10 mg once per day. The effectiveness of alprazolam XR was assessed on the basis of changes in various measures of panic attack frequency, on various measures of the Clinical Global Impression, and on the Overall Phobia Scale. In all, there were 7 primary efficacy measures in these studies, and alprazolam XR was superior to placebo on all 7 outcomes in both studies. The mean dose of alprazolam XR at the last treatment visit was 4.2 mg per day in the first study and 4.6 mg per day in the second.
In addition, there were two 8-week, fixed-dose, placebo-controlled studies of alprazolam XR in adult patients with panic disorder, involving fixed alprazolam XR doses of 4 mg and 6 mg once per day that did not show a benefit for either dose of alprazolam XR.
Analyses of the relationship between treatment outcome and gender did not suggest any differential responsiveness on the basis of gender.
HOW SUPPLIED/STORAGE AND HANDLING
Alprazolam XR is supplied in the following strengths and package configurations:
| Alprazolam XR Tablets | |||
|---|---|---|---|
| Package Configuration | Tablet Strength (mg) | NDC | |
Bottles of 60 | 0.5 mg | NDC 59762-0057-1 | white, pentagonal shaped tablets debossed with an "G" on one side and "0.5" on the other side |
Bottles of 60 | 1 mg | NDC 59762-0059-1 | yellow, square shaped tablets debossed with an "G" on one side and "1" on the other side |
Bottles of 60 | 2 mg | NDC 59762-0066-1 | blue, round shaped tablets debossed with an "G" on one side and "2" on the other side |
Bottles of 60 | 3 mg | NDC 59762-0068-1 | green, triangular shaped tablets debossed with an "G" on one side and "3" on the other side |
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Alprazolam is a 1,4 benzodiazepine. Alprazolam exerts its effect for the treatment of panic disorder through binding to the benzodiazepine site of gamma‑aminobutyric acid-A (GABA A ) receptors in the brain and enhances GABA-mediated synaptic inhibition.
Alprazolam xr - Alprazolam tablet, Extended Release PubMed™ news
- Journal Article • 2026 AprInsights from a six-year hair drug analysis compendium in drug-facilitated crimes involving vulnerable population cases.
- Journal Article • 2026 AprPharmacotherapeutic interventions to improve postoperative sleep quality in older adult patients: a systematic review and meta-analysis.
- Journal Article • 2026 AprInsights from a 6-year hair drug analysis compendium in drug-facilitated sexual assault cases.
- Journal Article • 2026 AprIn vitro metabolomics for the characterization of drugs of abuse and new psychoactive substances in SH-SY5Y cells.
- Journal Article • 2026 AprSignificant Anxiolytic Effect and Enhanced Recovery Benefits of Perioperative Low-Dose Olanzapine in Patients with Anxiety Undergoing THA: A Randomized Controlled Trial.