Get your patient on Amicar - Aminocaproic Acid solution (Aminocaproic Acid)
Amicar - Aminocaproic Acid solution prescribing information
INDICATIONS AND USAGE
AMICAR is useful in enhancing hemostasis when fibrinolysis contributes to bleeding. In life-threatening situations, transfusion of appropriate blood products and other emergency measures may be required.
Fibrinolytic bleeding may frequently be associated with surgical complications following heart surgery (with or without cardiac bypass procedures) and portacaval shunt; hematological disorders such as amegakaryocytic thrombocytopenia (accompanying aplastic anemia); acute and life-threatening abruptio placentae; hepatic cirrhosis; and neoplastic disease such as carcinoma of the prostate, lung, stomach, and cervix.
Urinary fibrinolysis, usually a normal physiological phenomenon, may contribute to excessive urinary tract fibrinolytic bleeding associated with surgical hematuria (following prostatectomy and nephrectomy) or nonsurgical hematuria (accompanying polycystic or neoplastic diseases of the genitourinary system). (See WARNINGS .)
DOSAGE AND ADMINISTRATION
An identical dosage regimen may be followed by administering AMICAR Tablets or AMICAR Oral Solution as follows:
For the treatment of acute bleeding syndromes due to elevated fibrinolytic activity, it is suggested that 5 AMICAR 1000 mg Tablets or 10 AMICAR 500 mg Tablets (5 g) or 20 milliliter of AMICAR Oral Solution (5 g) be administered during the first hour of treatment, followed by a continuing rate of 1 AMICAR 1000 mg Tablet or 2 AMICAR 500 mg Tablets (1 g) or 5 milliliter of AMICAR Oral Solution (1.25 g) per hour. This method of treatment would ordinarily be continued for about 8 hours or until the bleeding situation has been controlled.
CONTRAINDICATIONS
AMICAR should not be used when there is evidence of an active intravascular clotting process.
When there is uncertainty as to whether the cause of bleeding is primary fibrinolysis or disseminated intravascular coagulation (DIC), this distinction must be made before administering AMICAR.
The following tests can be applied to differentiate the two conditions:
Platelet count is usually decreased in DIC but normal in primary fibrinolysis.
Protamine paracoagulation test is positive in DIC; a precipitate forms when protamine sulfate is dropped into citrated plasma. The test is negative in the presence of primary fibrinolysis.
The euglobulin clot lysis test is abnormal in primary fibrinolysis but normal in DIC.
AMICAR must not be used in the presence of DIC without concomitant heparin.
ADVERSE REACTIONS
AMICAR is generally well tolerated. The following adverse experiences have been reported:
General: Edema, headache, malaise.
Hypersensitivity Reactions: Allergic and anaphylactoid reactions, anaphylaxis.
Cardiovascular: Bradycardia, hypotension, peripheral ischemia, thrombosis.
Gastrointestinal: Abdominal pain, diarrhea, nausea, vomiting.
Hematologic: Agranulocytosis, coagulation disorder, leukopenia, thrombocytopenia.
Musculoskeletal: CPK increased, muscle weakness, myalgia, myopathy (see WARNINGS ), myositis, rhabdomyolysis.
Neurologic: Confusion, convulsions, delirium, dizziness, hallucinations, intracranial hypertension, stroke, syncope.
Respiratory: Dyspnea, nasal congestion, pulmonary embolism.
Skin: Pruritis, rash.
Special Senses: Tinnitus, vision decreased, watery eyes.
Urogenital: BUN increased, renal failure. There have been some reports of dry ejaculation during the period of AMICAR treatment. These have been reported to date only in hemophilia patients who received the drug after undergoing dental surgical procedures. However, this symptom resolved in all patients within 24 to 48 hours of completion of therapy.
DESCRIPTION
AMICAR (aminocaproic acid) is 6-aminohexanoic acid, which acts as an inhibitor of fibrinolysis.
Its chemical structure is:
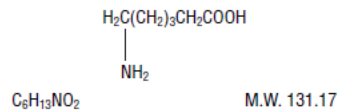
AMICAR is soluble in water, acid, and alkaline solutions; it is sparingly soluble in methanol and practically insoluble in chloroform.
AMICAR (aminocaproic acid) Oral Solution for oral administration, contains 0.25 g/mL of aminocaproic acid with methylparaben 0.20%, propylparaben 0.05%, edetate disodium 0.30% as preservatives and the following inactive ingredients: sodium saccharin, sorbitol solution, citric acid anhydrous, natural and artificial raspberry flavor and an artificial bitterness modifier.
Each AMICAR (aminocaproic acid) Tablet, for oral administration contains 500 mg or 1000 mg of aminocaproic acid and the following inactive ingredients: povidone, crospovidone, stearic acid, and magnesium stearate.
CLINICAL PHARMACOLOGY
The fibrinolysis-inhibitory effects of AMICAR appear to be exerted principally via inhibition of plasminogen activators and to a lesser degree through antiplasmin activity.
In adults, oral absorption appears to be a zero-order process with an absorption rate of 5.2 g/hr. The mean lag time in absorption is 10 minutes. After a single oral dose of 5 g, absorption was complete (F=1). Mean ± SD peak plasma concentrations (164 ± 28 mcg/mL) were reached within 1.2 ± 0.45 hours.
After oral administration, the apparent volume of distribution was estimated to be 23.1 ± 6.6 L (mean ± SD). Correspondingly, the volume of distribution after intravenous administration has been reported to be 30.0 ± 8.2 L. After prolonged administration, AMICAR has been found to distribute throughout extravascular and intravascular compartments of the body, penetrating human red blood cells as well as other tissue cells.
Renal excretion is the primary route of elimination. Sixty-five percent of the dose is recovered in the urine as unchanged drug and 11% of the dose appears as the metabolite adipic acid. Renal clearance (116 mL/min) approximates endogenous creatinine clearance. The total body clearance is 169 mL/min. The terminal elimination half-life for AMICAR is approximately 2 hours.
HOW SUPPLIED
AMICAR ® (aminocaproic acid)
AMICAR Oral Solution, 0.25 g/mL
Each mL of raspberry-flavored oral solution contains 0.25 g/mL of aminocaproic acid.
8 Fl. Oz. (236.5 mL) Bottle – NDC 49411-052-08
Store at 20°-25°C (68°-77°F)[see USP Controlled Room Temperature];Dispense in Tight Containers; Do Not Freeze.
AMICAR 500 mg Tablets
Each round, white tablet, engraved with XP on one side and scored on the other with A to the left of the score and 10 on the right, contains 500 mg of aminocaproic acid.
Bottle of 30 – NDC 49411-050-30
Store at 20°-25°C (68°-77°F)[see USP Controlled Room Temperature];Dispense in Tight Containers; Do Not Freeze.
AMICAR 1000 mg Tablets
Each oblong, white tablet, engraved with XP on one side and scored on the other with A to the left of the score and 20 on the right, contains 1000 mg of aminocaproic acid.
Bottle of 30 – NDC 49411-051-30
Store at 20°-25°C (68°-77°F)[see USP Controlled Room Temperature];Dispense in Tight Containers; Do Not Freeze.