Get your patient on Amphetamine Sulfate - Amphetamine Sulfate tablet (Amphetamine Sulfate)
Amphetamine Sulfate - Amphetamine Sulfate tablet prescribing information
WARNING: ABUSE, MISUSE, AND ADDICTION
Amphetamine sulfate tablets have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including amphetamine sulfate, can result in overdose and death (see OVERDOSAGE ), and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing amphetamine sulfate tablets, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout amphetamine sulfate tablets treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction (see WARNINGS and DRUG ABUSE AND DEPENDENCE ).
INDICATIONS AND USAGE
Amphetamine sulfate tablets, USP, are indicated for:
- Narcolepsy
- Attention Deficit Disorder with Hyperactivity as an integral part of a total treatment program which typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in children with behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: moderate to severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity. The diagnosis of the syndrome should not be made with finality when these symptoms are only of comparatively recent origin. Nonlocalizing (soft) neurological signs, learning disability, and abnormal EEG may or may not be present, and a diagnosis of central nervous system dysfunction may or not be warranted.
- Exogenous Obesity as a short term (a few weeks) adjunct in a regimen of weight reduction based on caloric restriction for patients refractory to alternative therapy, e.g., repeated diets, group programs, and other drugs. The limited usefulness of amphetamines (see CLINICAL PHARMACOLOGY ) should be weighed against possible risks inherent in use of the drug, such as those described below.
OVERDOSAGE
Clinical Effects of Overdose
Overdose of CNS stimulants is characterized by the following sympathomimetic effects:
- Cardiovascular effects including tachyarrhythmias, and hypertension or hypotension. Vasospasm, myocardial infarction, or aortic dissection may precipitate sudden cardiac death. Takotsubo cardiomyopathy may develop.
- CNS effects including psychomotor agitation, confusion, and hallucinations. Serotonin syndrome, seizures, cerebral vascular accidents, and coma may occur.
- Life-threatening hyperthermia (temperatures greater than 104°F) and rhabdomyolysis may develop.
Overdose Management
Consider the possibility of multiple drug ingestion. D-amphetamine is not dialyzable. Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
CONTRAINDICATIONS
- Known hypersensitivity to amphetamine products.
- During or within 14 days following the administration of monoamine oxidase inhibitors (hypertensive crises may result) (see WARNINGS ).
ADVERSE REACTIONS
Cardiovascular
Palpitations, tachycardia, elevation of blood pressure. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use.
Central Nervous System
Psychotic episodes at recommended doses (rare), overstimulation, restlessness, dizziness, insomnia, euphoria, dyskinesia, dysphoria, tremor, headache, exacerbation of motor and verbal tics and Tourette's syndrome.
Gastrointestinal
Dryness of the mouth, unpleasant taste, diarrhea, constipation, intestinal ischemia and other gastrointestinal disturbances. Anorexia and weight loss may occur as undesirable effects when amphetamines are used for other than the anorectic effect.
Allergic
Urticaria
Endocrine
Impotence, changes in libido, and frequent or prolonged erections.
Musculoskeletal
Rhabdomyolysis.
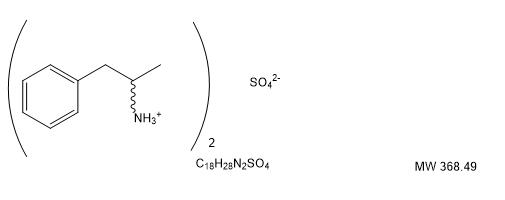
DESCRIPTION
Amphetamine Sulfate is a sympathomimetic amino of the amphetamine group. It is a white, odorless crystalline powder. It has a slightly bitter taste. Its solutions are acid to litmus, having a pH of 5.0 to 6.0. It is freely soluble in water and slightly soluble in alcohol.
Each tablet, for oral administration contains 5 mg or 10 mg of amphetamine sulfate, USP. Each tablet also contains the following inactive ingredients: crospovidone, magnesium stearate, silicified microcrystalline cellulose, and stearic acid.
Structural Formula:
CLINICAL PHARMACOLOGY
Amphetamines are non-catecholamine, sympathomimetic amines with CNS stimulant activity. Peripheral actions include elevations of systolic and diastolic blood pressures, and weak bronchodilator, and respiratory stimulant action.
Amphetamine, as the racemic form, differs from dextroamphetamine in a number of ways. The l-isomer is more potent than the d-isomer in cardiovascular activity, but much less potent in causing CNS excitatory effects. The racemic mixture also is less effective as an appetite suppressant when compared to dextroamphetamine. There is neither specific evidence which clearly establishes the mechanism whereby amphetamines produce mental and behavioral effects in children, nor conclusive evidence regarding how those effects relate to the condition of the central nervous system.
Drugs in this class used in obesity are commonly known as "anorectics" or "anorexigenics." It has not been established, however, that the action of such drugs in treating obesity is primarily one of appetite suppression. Other central nervous system actions or metabolic effects, may be involved, for example. Adult obese subjects instructed in dietary management and treated with "anorectic" drugs lose more weight on the average than these treated with placebo and diet, as determined in relatively short-term clinical trials.
The magnitude of increased weight loss of drug-treated patients over placebo-treated patients is only a fraction of a pound a week. The rate of weight loss is greatest in the first weeks of therapy for both drug and placebo subjects and tends to decrease in succeeding weeks. The origins of the increased weight loss due to the various possible drug effects are not established. The amount of weight loss associated with the use of an "anorectic" drug varies from trial to trial, and the increased weight loss appears to be related in part to variables other than the drug prescribed, such as the physician-investigator, the population treated, and the diet prescribed. Studies do not permit conclusions as to the relative importance of the drug and nondrug factors on weight loss.
The natural history of obesity is measured in years, whereas the studies cited are restricted to few weeks duration; thus, the total impact of drug-induced weight loss over that of diet alone must be considered clinically limited.
HOW SUPPLIED
Amphetamine sulfate tablets, USP, are supplied as follows:
5 mg: White, round, convex tablet, uppers scored and debossed "5" on the left side, lowers debossed "S" in bottles of 100 tablets with child-resistant closure, NDC 43547-457-10.
10 mg: White, round, convex tablet, uppers are double crossed scored and debossed "1" on the lower left corner and "0" on the lower right corner, lowers debossed "S" in bottles of 100 tablets with child-resistant closure, NDC 43547-458-10.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15 o C and 30 o C (59 o F and 86 o F). [see USP Controlled Room Temperature.] Dispense in a well-closed container, as defined in the USP.
Dispense with Medication Guide available at:
www.solcohealthcare.com/medguide/amphetamine-tablets.pdf
Manufactured by:
Prinston Laboratories
Charlotte, NC 28206, USA
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Revised: 10/2023
9040380-06