Get your patient on Ampicillin - Ampicillin Sodium injection, Powder, For Solution (Ampicillin Sodium)
Ampicillin - Ampicillin Sodium injection, Powder, For Solution prescribing information
INDICATIONS AND USAGE
Ampicillin for Injection, USP is indicated in the treatment of infections caused by susceptible strains of the designated organisms in the following conditions:
Respiratory Tract Infections caused by Streptococcus pneumoniae, Staphylococcus aureus (penicillinase and nonpenicillinase-producing), H. influenzae , and Group A beta-hemolytic streptococci .
Bacterial Meningitis caused by E. coli , Group B streptococci , and other Gram-negative bacteria ( Listeria monocytogenes, N. meningitidis ). The addition of an aminoglycoside with ampicillin may increase its effectiveness against Gram-negative bacteria.
Septicemia and Endocarditis caused by susceptible Gram-positive organisms including Streptococcus spp., penicillin G-susceptible staphylococci , and enterococci . Gram-negative sepsis caused by E. coli, Proteus mirabilis and Salmonella spp. responds to ampicillin. Endocarditis due to enterococcal strains usually respond to intravenous therapy. The addition of an aminoglycoside may enhance the effectiveness of ampicillin when treating streptococcal endocarditis.
Urinary Tract Infections caused by sensitive strains of E. coli and Proteus mirabilis.
Gastrointestinal Infections caused by Salmonella typhi (typhoid fever), other Salmonella spp., and Shigella spp. (dysentery) usually respond to oral or intravenous therapy.
Bacteriology studies to determine the causative organisms and their susceptibility to ampicillin should be performed. Therapy may be instituted prior to obtaining results of susceptibility testing. It is advisable to reserve the parenteral form of this drug for moderately severe and severe infections and for patients who are unable to take the oral forms. A change to oral ampicillin may be made as soon as appropriate.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ampicillin for Injection, USP and other antibacterial drugs, Ampicillin for Injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Indicated surgical procedures should be performed.
DOSAGE AND ADMINISTRATION
This insert is for a Pharmacy Bulk Package and is intended for preparing intravenous admixtures only. Dosage recommendations for intravenous injection are for informational purposes only.
Infections of the respiratory tract and soft tissues.
Patients weighing 40 kg (88 lbs) or more: 250 mg to 500 mg every 6 hours.
Patients weighing less than 40 kg (88 lbs): 25 to 50 mg/kg/day in equally divided doses at 6- to 8-hour intervals.
Infections of the gastrointestinal and genitourinary tracts (including those caused by Neisseria gonorrhoeae in females).
Patients weighing 40 kg (88 lbs) or more: 500 mg every 6 hours.
Patients weighing less than 40 kg (88 lbs): 50 mg/kg/day in equally divided doses at 6- to 8-hour intervals.
In the treatment of chronic urinary tract and intestinal infections, frequent bacteriological and clinical appraisal is necessary. Smaller doses than those recommended above should not be used. Higher doses should be used for stubborn or severe infections. In stubborn infections, therapy may be required for several weeks. It may be necessary to continue clinical and/or bacteriological follow-up for several months after cessation of therapy.
Urethritis in males due to N. gonorrhoeae .
Adults - Two doses of 500 mg each at an interval of 8 to 12 hours. Treatment may be repeated if necessary or extended if required.
In the treatment of complications of gonorrheal urethritis, such as prostatitis and epididymitis, prolonged and intensive therapy is recommended. Cases of gonorrhea with a suspected primary lesion of syphilis should have darkfield examinations before receiving treatment. In all other cases where concomitant syphilis is suspected, monthly serological tests should be made for a minimum of four months.
The doses for the preceding infections may be given by either the intramuscular or intravenous route. A change to oral ampicillin may be made when appropriate.
Bacterial Meningitis
Adults and children - 150 to 200 mg/kg/day in equally divided doses every 3 to 4 hours. (Treatment may be initiated with intravenous drip therapy and continued with intramuscular injections.) The doses for other infections may be given by either the intravenous or intramuscular route.
Neonates (less than or equal to 28 days of postnatal age) - Dosage should be based on Gestational age and Postnatal age according to Table 1 .
| Gestational age (weeks) | Postnatal age (days) | Dosage |
| less than or equal to 34 | less than or equal to 7 | 100 mg/kg/day in equally divided doses every 12 hours |
| less than or equal to 34 | greater than or equal to 8 and less than 28 | 150 mg/kg/day in equally divided doses every 12 hours |
| greater than 34 | less than or equal to 28 | 150 mg/kg/day in equally divided doses every 8 hours |
Septicemia
Adults and children: 150 to 200 mg/kg/day. Start with intravenous administration for at least three days and continue with the intramuscular route every 3 to 4 hours.
Neonates (less than or equal to 28 days of postnatal age) - Dosage should be based on Gestational age and Postnatal age according to Table 1 (above).
Treatment of all infections should be continued for a minimum of 48 to 72 hours beyond the time that the patient becomes asymptomatic or evidence of bacterial eradication has been obtained. A minimum of 10-days treatment is recommended for any infection caused by Group A beta-hemolytic streptococci to help prevent the occurrence of acute rheumatic fever or acute glomerulonephritis.
CONTRAINDICATIONS
A history of a previous hypersensitivity reaction to any of the penicillins is a contraindication.
ADVERSE REACTIONS
As with other penicillins, it may be expected that untoward reactions will be essentially limited to sensitivity phenomena. They are more likely to occur in individuals who have previously demonstrated hypersensitivity to penicillins and in those with a history of allergy, asthma, hay fever, or urticaria.
The following adverse reactions have been reported as associated with the use of ampicillin:
Gastrointestinal
Glossitis, stomatitis, black “hairy” tongue, nausea, vomiting, enterocolitis, pseudomembranous colitis, and diarrhea. (These reactions are usually associated with oral dosage forms.)
Hypersensitivity Reactions
Skin rashes and urticaria have been reported frequently. A few cases of exfoliative dermatitis and erythema multiforme have been reported. Linear IgA bullous dermatosis has been reported. Anaphylaxis is the most serious reaction experienced and has usually been associated with the parenteral dosage form.
Note: Urticaria, other skin rashes, and serum sickness-like reactions may be controlled with antihistamines and, if necessary, systemic corticosteroids. Whenever such reactions occur, ampicillin should be discontinued, unless, in the opinion of the physician, the condition being treated is life-threatening and amenable only to ampicillin therapy. Serious anaphylactic reactions require the immediate use of epinephrine, oxygen, and intravenous steroids.
Liver - A moderate rise in serum glutamic oxaloacetic transaminase (SGOT) has been noted, particularly in infants, but the significance of this finding is unknown. Mild transitory SGOT elevations have been observed in individuals receiving larger (two to four times) than usual and oft-repeated intramuscular injections. Evidence indicates that glutamic oxaloacetic transaminase (GOT) is released at the site of intramuscular injection of ampicillin sodium and that the presence of increased amounts of this enzyme in the blood does not necessarily indicate liver involvement.
Hemic and Lymphatic Systems - Anemia, thrombocytopenia, thrombocytopenic purpura, eosinophilia, leukopenia, and agranulocytosis have been reported during therapy with the penicillins. These reactions are usually reversible on discontinuation of therapy and are believed to be hypersensitivity phenomena.
Central Nervous System - Seizures
To report SUSPECTED ADVERSE REACTIONS, contact Avenacy Inc. at 1-855-283-6229 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
The concurrent administration of allopurinol and ampicillin increases substantially the incidence of skin rashes in patients receiving both drugs as compared to patients receiving ampicillin alone. It is not known whether this potentiation of ampicillin rashes is due to allopurinol or the hyperuricemia present in these patients.
DESCRIPTION
Ampicillin for Injection, USP the monosodium salt of [2S-[2α,5α,6β(S•)]]-6-[(aminophenylacetyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, is a synthetic penicillin for intravenous use.
The pharmacy bulk package contains sterile ampicillin sodium equivalent to 10 grams ampicillin. It is an antibacterial agent with a broad spectrum of bactericidal activity against both penicillin-susceptible Gram-positive organisms and many common Gram-negative pathogens.
Ampicillin for Injection, USP is a dry, white to off-white powder. The reconstituted solution is clear, colorless and free from visible particulates.
Each Pharmacy Bulk Package bottle contains ampicillin sodium equivalent to 10 grams of ampicillin. The sodium content is 65.8 mg (2.9 mEq) per one gram ampicillin.
A Pharmacy Bulk Package is a container of a sterile preparation for parenteral use that contains many single doses. The contents of this pharmacy bulk package are intended for use by a pharmacy admixture service for addition to suitable parenteral fluids in the preparation of admixtures for intravenous infusion (see DOSAGE AND ADMINISTRATION, Directions for Proper Use of Pharmacy Bulk Package ).
It has the following molecular structure:
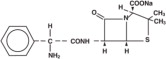
The molecular formula is C 16 H 18 N 3 NaO 4 S, and the molecular weight is 371.39. The pH range of the reconstituted solution is 8 to 10.
CLINICAL PHARMACOLOGY
Ampicillin for Injection, USP diffuses readily into most body tissues and fluids. However, penetration into the cerebrospinal fluid and brain occurs only when the meninges are inflamed. Ampicillin is excreted largely unchanged in the urine and its excretion can be delayed by concurrent administration of probenecid. Due to maturational changes in renal function, ampicillin half-life decreases as postmenstrual age (a sum of gestational age and postnatal age) increases for infants with postnatal age of less than 28 days. The active form appears in the bile in higher concentrations than those found in serum. Ampicillin is the least serum-bound of all the penicillins, averaging about 20% compared to approximately 60 to 90% for other penicillins. Ampicillin for Injection, USP is well tolerated by most patients and has been given in doses of 2 grams daily for many weeks without adverse reactions.
Microbiology
While in vitro studies have demonstrated the susceptibility of most strains of the following organisms, clinical efficacy for infections other than those included in the INDICATIONS AND USAGE section has not been demonstrated.
Antibacterial Activity
The following bacteria have been shown in in vitro studies to be susceptible to Ampicillin for Injection, USP:
Gram-positive Bacteria
Hemolytic and nonhemolytic streptococci Streptococcus pneumoniae Nonpenicillinase-producing staphylococci Clostridium spp. B. anthracis Listeria monocytogenes Most strains of enterococci.
Gram-negative Bacteria
H. influenzae N. gonorrhoeae N. meningitidis Proteus mirabilis Many strains of Salmonella , Shigella , and E. coli .
AMPICILLIN does not resist destruction by penicillinase.
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC .
HOW SUPPLIED
Ampicillin for Injection, USP is supplied as follows:
| NDC | Ampicillin for Injection, USP | Package Factor |
| 83634-100-99 | 10 gram Pharmacy Bulk Bottle | 1 bottle per carton |
Each Ampicillin for Injection, USP Pharmacy Bulk Package bottle contains ampicillin sodium equivalent to 10 grams of ampicillin.
Storage Conditions
Store dry powder at 20º to 25ºC (68º to 77ºF). [See USP Controlled Room Temperature.]
Protect the constituted solution from freezing.
Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
Clinitest is a registered trademark of Miles, Inc. Clinistix is a registered trademark of Bayer Corporation. Tes-Tape is a registered trademark of Eli Lilly Company.
AVENACY Mfd. for Avenacy Schaumburg, IL 60173 (USA)
Made in Italy
©2024 Avenacy Inc.
September 2024