Get your patient on Azurette - Desogestrel/ethinyl Estradiol And Ethinyl Estradiol (Desogestrel/Ethinyl Estradiol And Ethinyl Estradiol)
Azurette - Desogestrel/Ethinyl Estradiol And Ethinyl Estradiol prescribing information
INDICATIONS AND USAGE
Azurette (Desogestrel and Ethinyl Estradiol Tablets, USP and Ethinyl Estradiol Tablets, USP) is indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception.
Oral contraceptives are highly effective. Table II lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization, depends upon the reliability with which they are used. Correct and consistent use of these methods can result in lower failure rates.
| TABLE II : Percentage of women experiencing an unintended pregnancy during the first year of typical use and the first year of perfect use of contraception and the percentage continuing use at the end of the first year, United States. | |||
| % of Women Experiencing an Unintended Pregnancy within the First Year of Use | % of Women Continuing Use at One Year 3 | ||
| Method | Typical Use 1 | Perfect Use 2 | |
| ( 1 ) | ( 2 ) | ( 3 ) | ( 4 ) |
| Chance 4 | 85 | 85 | |
| Spermicides 5 | 26 | 6 | 40 |
| Periodic abstinence | 25 | 63 | |
| Calendar | 9 | ||
| Ovulation Method | 3 | ||
| Sympto-Thermal 6 | 2 | ||
| Post-Ovulation | 1 | ||
| Withdrawal | 19 | 4 | |
| Cap 7 | |||
| Parous Women | 40 | 26 | 42 |
| Nulliparous Women | 20 | 9 | 56 |
| Sponge | |||
| Parous Women | 40 | 20 | 42 |
| Nulliparous Women | 20 | 9 | 56 |
| Diaphragm 7 | 20 | 6 | 56 |
| Condom 8 | |||
| Female (Reality) | 21 | 5 | 56 |
| Male | 14 | 3 | 61 |
| Pill | 5 | 71 | |
| Progestin Only | 0.5 | ||
| Combined | 0.1 | ||
| IUD | |||
| Progesterone T | 2.0 | 1.5 | 81 |
| Copper T 380A | 0.8 | 0.6 | 78 |
| LNg 20 | 0.1 | 0.1 | 81 |
| Depo-Provera | 0.3 | 0.3 | 70 |
| Norplant and Norplant-2 | 0.05 | 0.05 | 88 |
| Female sterilization | 0.5 | 0.5 | 100 |
| Male sterilization | 0.15 | 0.10 | 100 |
Adapted from Hatcher et al., 1998, Ref#1.
1 Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
2 Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
3 Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year.
4 The percents becoming pregnant in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percent who would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether.
5 Foams, creams, gels, vaginal suppositories, and vaginal film.
6 Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the post-ovulatory phases.
7 With spermicidal cream or jelly.
8 Without spermicides.
DOSAGE AND ADMINISTRATION
To achieve maximum contraceptive effectiveness, Azurette (Desogestrel and Ethinyl Estradiol Tablets, USP and Ethinyl Estradiol Tablets, USP) must be taken exactly as directed and at intervals not exceeding 24 hours. Azurette may be initiated using either a Sunday start or a Day 1 start.
NOTE: Each cycle pack dispenser is preprinted with the days of the week, starting with Sunday, to facilitate a Sunday start regimen. Six different "day label stickers" are provided with each cycle pack dispenser in order to accommodate a Day 1 start regimen. In this case, the patient should place the self-adhesive "day label sticker" that corresponds to her starting day over the preprinted days.
IMPORTANT: The possibility of ovulation and conception prior to initiation of use of Azurette should be considered.
The use of Azurette for contraception may be initiated 4 weeks postpartum in women who elect not to breast feed. When the tablets are administered during the postpartum period, the increased risk of thromboembolic disease associated with the postpartum period must be considered (see CONTRAINDICATIONS and WARNINGS concerning thromboembolic disease. See also PRECAUTIONS for Nursing Mothers ).
If the patient starts on Azurette postpartum, and has not yet had a period, she should be instructed to use another method of contraception until a dark blue tablet has been taken daily for 7 days.
SUNDAY START
When initiating a Sunday start regimen, another method of contraception should be used until after the first 7 consecutive days of administration.
Using a Sunday start, tablets are taken daily without interruption as follows: The first dark blue tablet should be taken on the first Sunday after menstruation begins (if menstruation begins on Sunday, the first dark blue tablet is taken on that day). One dark blue tablet is taken daily for 21 days, followed by 1 white (inert) tablet daily for 2 days and 1 green (active) tablet daily for 5 days. For all subsequent cycles, the patient then begins a new 28-tablet regimen on the next day (Sunday) after taking the last green tablet. [If switching from a Sunday Start oral contraceptive, the first Azurette (desogestrel/ethinyl estradiol and ethinyl estradiol) tablet should be taken on the second Sunday after the last tablet of a 21 day regimen or should be taken on the first Sunday after the last inactive tablet of a 28 day regimen.]
If a patient misses 1 dark blue tablet, she should take the missed tablet as soon as she remembers. If the patient misses 2 consecutive dark blue tablets in Week 1 or Week 2, the patient should take 2 tablets the day she remembers and 2 tablets the next day; thereafter, the patient should resume taking 1 tablet daily until she finishes the cycle pack. The patient should be instructed to use a back-up method of birth control if she has intercourse in the 7 days after missing pills. If the patient misses 2 consecutive dark blue tablets in the third week or misses 3 or more dark blue tablets in a row at any time during the cycle, the patient should keep taking 1 dark blue tablet daily until the next Sunday. On Sunday the patient should throw out the rest of that cycle pack and start a new cycle pack that same day. The patient should be instructed to use a back-up method of birth control if she has intercourse in the 7 days after missing pills.
DAY 1 START
Counting the first day of menstruation as "Day 1", tablets are taken without interruption as follows: One dark blue tablet daily for 21 days, one white (inert) tablet daily for 2 days followed by 1 green (ethinyl estradiol) tablet daily for 5 days. For all subsequent cycles, the patient then begins a new 28-tablet regimen on the next day after taking the last green tablet. [If switching directly from another oral contraceptive, the first dark blue tablet should be taken on the first day of menstruation which begins after the last ACTIVE tablet of the previous product.]
If a patient misses 1 dark blue tablet, she should take the missed tablet as soon as she remembers. If the patient misses 2 consecutive dark blue tablets in Week 1 or Week 2, the patient should take 2 tablets the day she remembers and 2 tablets the next day; thereafter, the patient should resume taking 1 tablet daily until she finishes the cycle pack. The patient should be instructed to use a back-up method of birth control if she has intercourse in the 7 days after missing pills. If the patient misses 2 consecutive dark blue tablets in the third week or if the patient misses 3 or more dark blue tablets in a row at any time during the cycle, the patient should throw out the rest of that cycle pack and start a new cycle pack that same day. The patient should be instructed to use a back-up method of birth control if she has intercourse in the 7 days after missing pills.
ALL ORAL CONTRACEPTIVES
Breakthrough bleeding, spotting, and amenorrhea are frequent reasons for patients discontinuing oral contraceptives. In breakthrough bleeding, as in all cases of irregular bleeding from the vagina, non-functional causes should be borne in mind. In undiagnosed persistent or recurrent abnormal bleeding from the vagina, adequate diagnostic measures are indicated to rule out pregnancy or malignancy. If both pregnancy and pathology have been excluded, time or a change to another preparation may solve the problem. Changing to an oral contraceptive with a higher estrogen content, while potentially useful in minimizing menstrual irregularity, should be done only if necessary since this may increase the risk of thromboembolic disease.
Use of oral contraceptives in the event of a missed menstrual period:
1. If the patient has not adhered to the prescribed schedule, the possibility of pregnancy should be considered at the time of the first missed period and oral contraceptive use should be discontinued until pregnancy is ruled out.
2. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out before continuing oral contraceptive use.
CONTRAINDICATIONS
Oral contraceptives should not be used in women who currently have the following conditions:
• Thrombophlebitis or thromboembolic disorders
• A past history of deep vein thrombophlebitis or thromboembolic disorders
• Cerebral vascular or coronary artery disease
• Known or suspected carcinoma of the breast
• Carcinoma of the endometrium or other known or suspected estrogen-dependent neoplasia
• Undiagnosed abnormal genital bleeding
• Cholestatic jaundice of pregnancy or jaundice with prior pill use
• Hepatic adenomas or carcinomas
• Known or suspected pregnancy
• Are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see Warnings , RISK OF LIVER ENZYME ELEVATIONS WITH CONCOMITANT HEPATITIS C TREATMENT ).
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Mayne Pharma Toll-Free at 1-844-825-8500 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
An increased risk of the following serious adverse reactions has been associated with the use of oral contraceptives (see WARNINGS section):
• Thrombophlebitis and venous thrombosis with or without embolism
• Arterial thromboembolism
• Pulmonary embolism
• Myocardial infarction
• Cerebral hemorrhage
• Cerebral thrombosis
• Hypertension
• Gallbladder disease
• Hepatic adenomas or benign liver tumors
There is evidence of an association between the following conditions and the use of oral contraceptives:
• Mesenteric thrombosis
• Retinal thrombosis
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug-related:
• Nausea
• Vomiting
• Gastrointestinal symptoms (such as abdominal cramps and bloating)
• Breakthrough bleeding
• Spotting
• Change in menstrual flow
• Amenorrhea
• Temporary infertility after discontinuation of treatment
• Edema
• Melasma which may persist
• Breast changes: tenderness, enlargement, secretion
• Change in weight (increase or decrease)
• Change in cervical erosion and secretion
• Diminution in lactation when given immediately postpartum
• Cholestatic jaundice
• Migraine
• Rash (allergic)
• Mental depression
• Reduced tolerance to carbohydrates
• Vaginal candidiasis
• Change in corneal curvature (steepening)
• Intolerance to contact lenses
The following adverse reactions have been reported in users of oral contraceptives and the association has been neither confirmed nor refuted:
• Pre-menstrual syndrome
• Cataracts
• Changes in appetite
• Cystitis-like syndrome
• Headache
• Nervousness
• Dizziness
• Hirsutism
• Loss of scalp hair
• Erythema multiforme
• Erythema nodosum
• Hemorrhagic eruption
• Vaginitis
• Porphyria
• Impaired renal function
• Hemolytic uremic syndrome
• Acne
• Changes in libido
• Colitis
• Budd-Chiari Syndrome
DESCRIPTION
Azurette (Desogestrel and Ethinyl Estradiol tablets, USP and Ethinyl Estradiol tablets, USP) provide an oral contraceptive regimen of 21 dark blue tablets each containing 0.15 mg desogestrel (13-ethyl-11-methylene-18,19-dinor-17 alpha-pregn-4-en-20-yn-17-ol), 0.02 mg ethinyl estradiol (19-nor-17 alpha-pregna-1,3,5 (10)-trien-20-yne-3,17-diol), and inactive ingredients which include: titanium dioxide, macrogol/PEG 400 NF, hydroxypropyl-methylcellulose/hypromellose, FD&C Red #40, FD&C Yellow #6, FD&C Blue #1, vitamin E, lactose monohydrate, povidone, stearic acid and pregelatinized starch, followed by 2 white tablets with the following inactive ingredients: titanium dioxide, polydextrose, hypromellose, triacetin, macrogol/polyethylene glycol 8000, lactose, magnesium stearate and pregelatinized corn starch. Azurette also contains 5 green tablets containing 0.01 mg ethinyl estradiol (19-nor-17 alpha-pregna-1,3,5 (10)-trien-20-yne-3,17-diol) and inactive ingredients which include: polyvinyl alcohol, titanium dioxide, talc, macrogol/PEG, lecithin (soya), D&C Yellow No. 10, FD&C Blue No. 1, Iron Oxide Yellow, FD&C Red No. 40, lactose monohydrate, magnesium stearate and pregelatinized starch. The molecular weights for desogestrel and ethinyl estradiol are 310.48 and 296.21 respectively. The structural formulas are as follows:
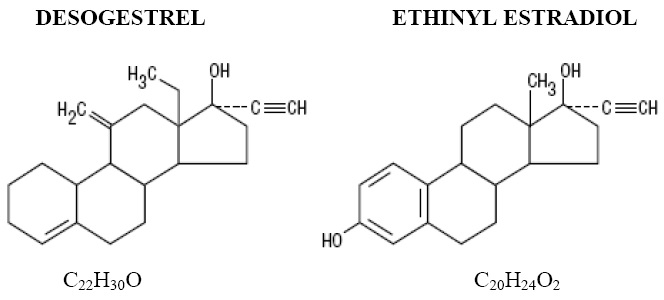
The 21 dark blue tablets meet USP Dissolution Test 2.
CLINICAL PHARMACOLOGY
Combination oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations include changes in the cervical mucus (which increase the difficulty of sperm entry into the uterus) and the endometrium (which reduce the likelihood of implantation).
Receptor binding studies, as well as studies in animals, have shown that etonogestrel, the biologically active metabolite of desogestrel, combines high progestational activity with minimal intrinsic androgenicity. 91,92 The relevance of this latter finding in humans is unknown.
Pharmacokinetics
Absorption
Desogestrel is rapidly and almost completely absorbed and converted into etonogestrel, its biologically active metabolite. Following oral administration, the relative bioavailability of desogestrel compared to a solution, as measured by serum levels of etonogestrel, is approximately 100%. Azurette (desogestrel and ethinyl estradiol tablets, USP and ethinyl estradiol tablets, USP) provide two different regimens of ethinyl estradiol; 0.02 mg in the combination tablet [dark blue] as well as 0.01 mg in the green tablet. Ethinyl estradiol is rapidly and almost completely absorbed. After a single dose of desogestrel/ethinyl estradiol combination tablet [dark blue], the relative bioavailability of ethinyl estradiol is approximately 93% while the relative bioavailability of the 0.01 mg tablet [green] is 99%. The effect of food on the bioavailability of Azurette tablets following oral administration has not been evaluated.
The pharmacokinetics of etonogestrel and ethinyl estradiol following multiple dose administration of desogestrel/ethinyl estradiol and ethinyl estradiol tablets were determined during the third cycle in 17 subjects. Plasma concentrations of etonogestrel and ethinyl estradiol reached steady-state by Day 21. The AUC (0–24) for etonogestrel at steady-state on Day 21 was approximately 2.2 times higher than AUC (0–24) on Day 1 of the third cycle. The pharmacokinetic parameters of etonogestrel and ethinyl estradiol during the third cycle following multiple dose administration of desogestrel/ethinyl estradiol and ethinyl estradiol tablets are summarized in Table I.
| TABLE I : MEAN (SD) PHARMACOKINETIC PARAMETERS OF DESOGESTREL/ETHINYL ESTRADIOL AND ETHINYL ESTRADIOL TABLETS OVER A 28-DAY DOSING PERIOD IN THE THIRD CYCLE (n=17). | ||||||
| Etonogestrel | ||||||
| Day | Dose• | C m a x | T m a x | t 1 / 2 | AUC 0 - 2 4 | CL/F |
| mg | pg/mL | h | h | pg/mL•hr | L/h | |
| 1 | 0.15 | 2503.6 (987.6) | 2.4 (1.0) | 29.8 (16.3) | 17,832 (5674) | 5.4 (2.5) |
| 21 | 0.15 | 4091.2 (1186.2) | 1.6 (0.7) | 27.8 (7.2) | 39,391 (12,134) | 4.4 (1.4) |
| •Desogestrel | ||||||
| EthinylEstradiol | ||||||
| Day | Dose | C m a x | T m a x | t 1 / 2 | AUC 0 - 2 4 | CL/F |
| mg | pg/mL | h | h | pg/mL•hr | L/h | |
| 1 | 0.02 | 51.9 (15.4) | 2.9 (1.2) | 16.5 (4.8) | 566 (173) a | 25.7 (9.1) |
| 21 | 0.02 | 62.2 (25.9) | 2.0 (0.8) | 23.9 (25.5) | 597 (127) a | 35.1 (8.2) |
| 24 | 0.01 | 24.6 (10.8) | 2.4 (1.0) | 18.8 (10.3) | 246 (65) | 43.6 (12.2) |
| 28 | 0.01 | 35.3 (27.5) | 2.1 (1.3) | 18.9 (8.3) | 312 (62) | 33.2 (6.6) |
| a n=16 | ||||||
| C m a x – measured peak concentration | ||||||
| T m a x – observed time of peak concentration | ||||||
| t 1 / 2 – elimination half-life, calculated by 0.693/K e l i m | ||||||
| AUC 0 – 2 4 – area under the concentration-time curve calculated by the linear trapezoidal rule (Time 0 to 24 hours) | ||||||
| CL/F – apparent clearance | ||||||
Distribution
Etonogestrel, the active metabolite of desogestrel, was found to be 99% protein bound, primarily to sex hormone-binding globulin (SHBG). Ethinyl estradiol is approximately 98.3% bound, mainly to plasma albumin. Ethinyl estradiol does not bind to SHBG, but induces SHBG synthesis. Desogestrel, in combination with ethinyl estradiol, does not counteract the estrogen-induced increase in SHBG, resulting in lower serum levels of free testosterone. 96–99
Metabolism
Desogestrel: Desogestrel is rapidly and completely metabolized by hydroxylation in the intestinal mucosa and on first pass through the liver to etonogestrel. Other metabolites (i.e., 3α-OH-desogestrel, 3β-OH-desogestrel, and 3α-OH-5α-H-desogestrel) with no pharmacologic actions also have been identified and these metabolites may undergo glucuronide and sulfate conjugation.
Ethinyl estradiol: Ethinyl estradiol is subject to a significant degree of presystemic conjugation (phase II metabolism). Ethinyl estradiol escaping gut wall conjugation undergoes phase I metabolism and hepatic conjugation (phase II metabolism). Major phase I metabolites are 2-OH-ethinyl estradiol and 2-methoxy-ethinyl estradiol. Sulfate and glucuronide conjugates of both ethinyl estradiol and phase I metabolites, which are excreted in bile, can undergo enterohepatic circulation.
Excretion
Etonogestrel and ethinyl estradiol are excreted in urine, bile, and feces. At steady state, on Day 21, the elimination half-life of etonogestrel is 27.8±7.2 hours and the elimination half-life of ethinyl estradiol for the combination tablet is 23.9±25.5 hours. For the 0.01 mg ethinyl estradiol tablet [green], the elimination half-life at steady state, Day 28, is 18.9±8.3 hours.
Special Populations
Race
There is no information to determine the effect of race on the pharmacokinetics of Azurette (desogestrel and ethinyl estradiol tablets, USP and ethinyl estradiol tablets, USP)
Hepatic Insufficiency
No formal studies were conducted to evaluate the effect of hepatic disease on the disposition of Azurette.
Renal Insufficiency
No formal studies were conducted to evaluate the effect of renal disease on the disposition of Azurette.
Drug-Drug Interactions
Interactions between desogestrel/ethinyl estradiol and other drugs have been reported in the literature. No formal drug-drug interaction studies were conducted (see PRECAUTIONS section).
HOW SUPPLIED
AZURETTE ® (Desogestrel and Ethinyl Estradiol Tablets, USP and Ethinyl Estradiol Tablets, USP) contain 21 round dark blue tablets, 2 round white tablets and 5 round green tablets in a blister card (NDC 51862-890-01) within a plastic dispenser. Each dark blue tablet (debossed with "M3" on one side) contains 0.15 mg desogestrel and 0.02 mg ethinyl estradiol. Each white tablet (debossed with "P" on one side and the " N " on the other side) contains inert ingredients.Each green tablet (debossed with "M4" on one side) contains 0.01 mg ethinyl estradiol.
AZURETTE Tablets are available in the following configurations:
Carton of 1 NDC 51862-890-02
Carton of 3 NDC 51862-890-03
Carton of 6 NDC 51862-890-06
Storage
Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature].
Rx Only