Get your patient on Barrier For Dogs (Imidacloprid, Moxidectin)
Barrier For Dogs prescribing information
WARNING
- DO NOT ADMINISTER THIS PRODUCT ORALLY
- For the first 30 minutes after application ensure that dogs cannot lick the product from application sites on themselves or other treated animals.
- Children should not come in contact with application sites for two (2) hours after application.
(See Contraindications , Warnings , Human Warnings , and Adverse Reactions , for more information)
DOSAGE AND ADMINISTRATION:
The recommended minimum dose is 4.5 mg/lb (10 mg/kg) imidacloprid and 1.1 mg/lb (2.5 mg/kg) moxidectin, once a month, by topical administration.
Do not apply to irritated skin.
- Remove one dose applicator tube from the package. As specified in the following table, administer the entire contents of the Barrier for Dogs (imidacloprid + moxidectin) tube that correctly corresponds with the body weight of the dog.
• Dogs over 110 lbs. should be treated with the appropriate combination of Barrier for Dogs tubes.
Dog (lbs.) Volume (mL) Imidacloprid (mg) Moxidectin (mg) 3–9 0.4 40 10 9.1–20 1.0 100 25 20.1–55 2.5 250 62.5 55.1–88 4.0 400 100 88.1–110• 5.0 500 125 
- While holding the tube in an upright position, twist the cap to break the seal and apply. The cap will remain on the tube.
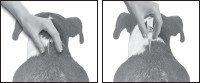
3. The dog should be standing for application. Part the hair on the back of the dog between the shoulder blades until the skin is visible. For dogs weighing 20 lbs. or less, place the tip of the tube on the skin and apply the entire contents directly on the exposed skin at one spot between the shoulder blades. For dogs weighing more than 20 lbs., place the tip of the tube on the skin and apply the entire contents directly on the exposed skin at 3 or 4 spots on the top of the backline from the base of the neck to the upper back in an area inaccessible to licking. Do not apply an amount of solution at any one location that could run off the side of the dog.
Do not let this product get in your dog's mouth or eyes. Do not allow the dog to lick any of the application sites for 30 minutes. In households with multiple pets, keep each treated dog separated from other treated dogs and other pets for 30 minutes after application to prevent licking the application sites. (See WARNINGS .)
Stiff hair, a damp appearance of the hair, pink skin, or a slight powdery residue may be observed at the application site on some animals. This is temporary and does not affect the safety and effectiveness of the product.
Shampooing 90 minutes after treatment does not reduce the effectiveness of Barrier for Dogs in the prevention of heartworm disease.
Shampooing or water immersion 4 days after treatment will not reduce the effectiveness of Barrier for Dogs in the treatment of flea infestations. However, shampooing as often as once weekly may reduce the effectiveness of the product against fleas.
Heartworm Prevention : For prevention of heartworm disease, Barrier for Dogs should be administered at one-month intervals. Barrier for Dogs may be administered year-round or at a minimum should start one month before the first expected exposure to mosquitoes and should continue at monthly intervals until one month after the last exposure to mosquitoes. If a dose is missed and a 30-day interval between doses is exceeded, administer Barrier for Dogs immediately and resume the monthly dosing schedule. When replacing another heartworm preventative product in a heartworm prevention program, the first treatment with Barrier for Dogs should be given within one month of the last dose of the former medication.
Treatment of Circulating Microfilaria: For the treatment of circulating D. immitis microfilaria in heartworm-positive dogs, Barrier for Dogs should be administered at one-month intervals. Treatment with an approved adulticide therapy is recommended because Barrier for Dogs is not effective for the treatment of adult D. immitis . (See PRECAUTIONS .)
Flea Treatment: For the treatment of flea infestations, Barrier for Dog s should be administered at one-month intervals. If the dog is already infested with fleas when the first dose of Barrier for Dogs is administered, adult fleas on the dog will be killed. However, reinfestation from the emergence of pre-existing pupae in the environment may continue to occur for six weeks or longer after treatment is initiated. Dogs treated with imidacloprid, including those with pre-existing flea allergy dermatitis have shown clinical improvement as a direct result of elimination of fleas from the dog.
Treatment and Control of Intestinal Nematode Infections:
For the treatment and control of intestinal hookworm infections caused by Ancylostoma caninum and Uncinaria stenocephala (adults, immature adults and fourth stage larvae) and roundworm infections caused by Toxocara canis (adults and fourth stage larvae), and Toxascaris leonina (adults), and whipworm infections caused by Trichuris vulpis (adults), Barrier for Dogs should be administered once as a single topical dose.
Treatment and Control of Sarcoptic Mange: For the treatment and control of sarcoptic mange caused by Sarcoptes scabiei var. canis, Barrier for Dogs should be administered as a single topical dose. A second monthly dose may be administered if necessary.
CONTRAINDICATIONS:
Do not administer this product orally. (See WARNINGS .)
Do not use this product (containing 2.5 % moxidectin) on cats.
ADVERSE REACTIONS:
Heartworm-Negative Dogs
Field Studies: Following treatment with imidacloprid and moxidectin or an active control, dog owners reported the following post-treatment reactions:
| OBSERVATION | Imidacloprid and Moxidectin Topical n = 128 | Active Control n = 68 |
| Pruritus | 19 dogs (14.8 %) | 7 dogs (10.3 %) |
| Residue | 9 dogs (7.0 %) | 5 dogs (7.4 %) |
| Medicinal Odor | 5 dogs (3.9 %) | None observed |
| Lethargy | 1 dog (0.8 %) | 1 dog (1.5 %) |
| Inappetence | 1 dog (0.8 %) | 1 dog (1.5 %) |
| Hyperactivity | 1 dog (0.8 %) | None observed |
During a field study using 61 dogs with pre-existing flea allergy dermatitis, one (1.6 %) dog experienced localized pruritus immediately after imidacloprid application, and one investigator noted hyperkeratosis at the application site of one dog (1.6 %).
In a field safety and effectiveness study, imidacloprid and moxidectin was administered to 92 client-owned dogs with sarcoptic mange. The dogs ranged in age from 2 months to 12.5 years and ranged in weight from 3 to 231.5 pounds. Adverse reactions in dogs treated with imidacloprid and moxidectin included hematochezia, diarrhea, vomiting, lethargy, inappetence, and pyoderma.
Laboratory Effectiveness Studies: One dog in a laboratory effectiveness study experienced weakness, depression, and unsteadiness between 6 and 9 days after application with imidacloprid and moxidectin. The signs resolved without intervention by day 10 post-application. The signs in this dog may have been related to peak serum levels of moxidectin, which vary between dogs, and occur between 1 and 21 days after application of imidacloprid and moxidectin.
The following clinical observations also occurred in laboratory effectiveness studies following application with imidacloprid and moxidectin and may be directly attributed to the drug or may be secondary to the intestinal parasite burden or other underlying conditions in the dogs: diarrhea, bloody stools, vomiting, anorexia, lethargy, coughing, ocular discharge and nasal discharge. Observations at the application sites included damp, stiff or greasy hair, the appearance of a white deposit on the hair, and mild erythema, which resolved without treatment within 2 to 48 hours.
Heartworm-Positive Dogs
Field Study: A 56-day field safety study was conducted in 214 D. immitis heartworm and microfilaria positive dogs with Class 1, 2 or 3 heartworm disease. All dogs received imidacloprid and moxidectin on Study Days 0 and 28; 108 dogs also received melarsomine dihydrochloride on Study Days – 14, 14, and 15. All dogs were hospitalized for a minimum of 12 hours following each treatment. Effectiveness against circulating D. immitis microfilariae was > 90 % at five of six sites; however, one site had an effectiveness of 73.3 %. The microfilaria count in some heartworm-positive dogs increased or remained unchanged following treatment with imidacloprid and moxidectin alone or in a dosing regimen with melarsomine dihydrochloride.
Following treatment with imidacloprid and moxidectin alone or in a dosing regimen with melarsomine dihydrochloride, the following adverse reactions were observed:
| Adverse Reaction | Dogs Treated with Imidacloprid and Moxidectin Only n = 106 | Dogs Treated with Imidacloprid and Moxidectin + Melarsomine n = 108 |
| Cough | 24 (22.6 %) | 25 (23.1 %) |
| Lethargy | 14 (13.2 %) | 42 (38.9 %) |
| Vomiting | 11 (10.4 %) | 18 (16.7 %) |
| Diarrhea, including hemorrhagic | 10 (9.4 %) | 22 (20.4 %) |
| Inappetence | 7 (6.6 %) | 19 (17.6 %) |
| Dyspnea | 6 (5.7 %) | 10 (9.3 %) |
| Tachypnea | 1 (< 1 %) | 7 (6.5 %) |
| Pulmonary Hemorrhage | 0 | 1 (< 1 %) |
| Death | 0 | 3 (2.8 %) |
Three dogs treated with imidacloprid and moxidectin in a dosing regimen with melarsomine dihydrochloride died of pulmonary embolism from dead and dying heartworms. One dog, treated with imidacloprid and moxidectin and melarsomine dihydrochloride, experienced pulmonary hemorrhage and responded to supportive medical treatment. Following the first treatment with imidacloprid and moxidectin alone, two dogs experienced adverse reactions (coughing, vomiting, and dyspnea) that required hospitalization. In both groups, there were more adverse reactions to imidacloprid and moxidectin following the first treatment than the second treatment.
To report a suspected adverse reaction, call Aurora Pharmaceutical at 1-888-215-1256.
Post-Approval Experience
The following adverse events are based on post-approval adverse drug experience reporting. Not all adverse reactions are reported to FDA CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using this data. The following adverse events reported in dogs are listed in decreasing order of reporting frequency: depression/lethargy, vomiting, pruritus, diarrhea, anorexia, hyperactivity, ataxia, trembling, hypersalivation, application site reactions (alopecia, pruritus, lesions, and erythema), seizures, and anaphylaxis/anaphylactic reactions (hives, urticaria, facial swelling, edema of the head).
Serious reactions, including neurologic signs and death have been reported when cats have been exposed (orally and topically) to this product.
In humans, nausea, numbness or tingling of the mouth/lips and throat, ocular and dermal irritation, pruritus, headache, vomiting, diarrhea, depression and dyspnea have been reported following exposure to this product.
DESCRIPTION:
Barrier for Dogs (10 % imidacloprid + 2.5 % moxidectin) is a colorless to yellow ready-to-use solution packaged in single dose applicator tubes for topical treatment of dogs. The formulation and dosage schedule are designed to provide a minimum of 4.5 mg/lb (10 mg/kg) imidacloprid and 1.1 mg/lb (2.5 mg/kg) moxidectin based on body weight.
Imidacloprid is a chloronicotinyl nitroguanidine insecticide. The chemical name for imidacloprid is 1-[(6-Chloro-3-pyridinyl)methyl]-N-nitro-2-imidazolidinimine. Moxidectin is a semisynthetic macrocyclic lactone endectocide derived from the actinomycete Streptomycetes cyaneogriseus noncyanogenus . The chemical name for moxidectin is [6R, 23E, 25S(E)]-5-0- Demethyl-28-deoxy-25-(1,3-dimethyl-1-butenyl)-6,28-epoxy-23-(methoxyimino) milbemycin B.
HOW SUPPLIED:
Applications Per Package
6 x 0.4 mL tubes
6 x 1.0 mL tubes
6 x 2.5 mL tubes
6 x 4.0 mL tubes
6 x 5.0 mL tubes
55-1554 06/2025
Approved by FDA under ANADA # 200-718
MANUFACTURED BY: AURORA PHARMACEUTICAL, INC. NORTHFIELD, MN 55057 www.aurorapharmaceutical.com
MANUFACTURED IN U.S.A.
Barrier is a registered trademark of Aurora Pharmaceutical, Inc.
Barrier For Dogs PubMed™ news
- Journal Article • 2026 MayProtective role of Algerian Myrtus communis L. against pesticide mixture-induced reproductive toxicity: a combined experimental and molecular dynamics insights targeting steroidogenesis pathway.
- Journal Article • 2026 MayPrioritizing emerging organic pollutants in rivers of Thailand using suspect screening analysis with high-resolution mass spectrometry.
- Journal Article • 2026 MayAmerican Association of Veterinary Parasitologists Hookworm Task Force review on best practices for diagnosis and treatment of multidrug-resistant hookworms.
- Journal Article • 2026 MayA cascade-functionalized ZnCdS/Ce-MOF composite gate coupled with a self-replicating catalytic hairpin assembly for OPECT sensing of imidacloprid.
- Journal Article • 2026 MayDevelopment and Validation of LC-MS/MS Methods for Quantification of Fluralaner and Moxidectin in Cat Plasma and Its Application in a Pharmacokinetic Study.