Get your patient on Benzonatate 100 Mg
Benzonatate 100 Mg prescribing information
INDICATIONS & USAGE
Benzonatate capsules are indicated for the symptomatic relief of cough.
DOSAGE & ADMINISTRATION
Adults and Children over 10 years of age: Usual dose is one 100 mg or 200 mg capsule three times a day as needed for cough. If necessary to control cough, up to 600 mg daily in three divided doses may be given. Benzonatate Capsules should be swallowed whole. Benzonatate Capsules are not to be broken, chewed, dissolved, cut or crushed.
CONTRAINDICATIONS
Hypersensitivity to benzonatate or related compounds.
ADVERSE REACTIONS
Potential Adverse Reactions to benzonatate may include:
Hypersensitivity reactions including bronchospasm, laryngospasm, cardiovascular collapse possibly related to local anesthesia from chewing or sucking the capsule. CNS: sedation; headache; dizziness; mental confusion; visual hallucinations. GI: constipation; nausea; GI upset. Dermatologic: pruritus; skin eruptions. Other: nasal congestion; sensation of burning in the eyes; vague “chilly” sensation; numbness of the chest; hypersensitivity. Deliberate or accidental overdose has resulted in death, particularly in children.
DESCRIPTION
Benzonatate, a non-narcotic oral antitussive agent, is 2, 5, 8, 11, 14, 17, 20, 23, 26-nonaoxaoctacosan-28-yl p- (butylamino) benzoate; with a molecular weight of 603.7.
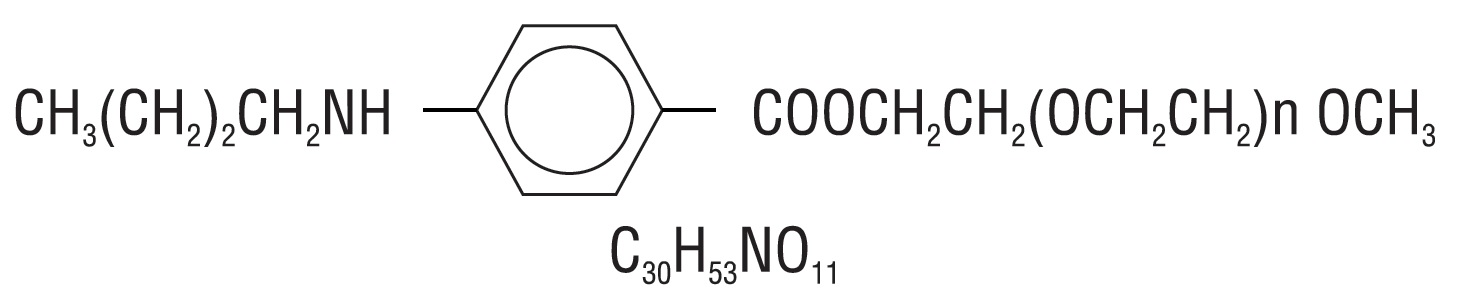
Each Benzonatate Capsule contains Benzonatate, USP 100 mg or 200 mg
Benzonatate Capsules USP also contain: gelatin, glycerin, purified water, sorbitol sorbitan solution. The imprinting ink contains propylene glycol, shellac and titanium dioxide.
FDA approved dissolution test specifications differ from USP.
CLINICAL PHARMACOLOGY
Benzonatate acts peripherally by anesthetizing the stretch receptors located in the respiratory passages, lungs, and pleura by dampening their activity and thereby reducing the cough reflex at its source. It begins to act within 15 to 20 minutes and its effect lasts for 3 to 8 hours. Benzonatate has no inhibitory effect on the respiratory center in recommended dosage.
HOW SUPPLIED
Benzonatate Capsules USP, 100 mg (Light yellow to yellow coloured, transparent to translucent, oval shaped capsule imprinted with '132' in white colour on capsule shell) Bottle of 30 NDC 25000-002-03
Benzonatate Capsules USP, 100 mg (Light yellow to yellow coloured, transparent to translucent, oval shaped capsule imprinted with '132' in white colour on capsule shell) Bottle of 100 NDC 25000-002-08
Benzonatate Capsules USP, 100 mg (Light yellow to yellow coloured, transparent to translucent, oval shaped capsule imprinted with '132' in white colour on capsule shell) Bottle of 500 NDC 25000-002-12 Benzonatate Capsules USP, 200 mg (Light yellow to yellow coloured, transparent to translucent, oval shaped capsule imprinted with '137' in white colour on capsule shell) Bottle of 30 NDC 25000-003-03 Benzonatate Capsules USP, 200 mg (Light yellow to yellow coloured, transparent to translucent, oval shaped capsule imprinted with '137' in white colour on capsule shell) Bottle of 100 NDC 25000-003-08 Benzonatate Capsules USP, 200 mg (Light yellow to yellow coloured, transparent to translucent, oval shaped capsule imprinted with '137' in white colour on capsule shell) Bottle of 500 NDC 25000-003-12
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Protect from light. To report SUSPECTED ADVERSE REACTIONS, contact Marksans Pharma Ltd. at 1-877-290-4008 and/or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. Manufactured for: Time-Cap Labs, Inc. 7 Michael Avenue, Farmingdale, NY 11735, USA Manufactured by: Marksans Pharma Ltd. Plot No. L-82, L-83, Verna Indl. Estate, Verna, Goa-403722, India Rev. 03/26