Get your patient on Bisoprolol Fumarate - Bisoprolol Fumarate tablet (Bisoprolol Fumarate)
Bisoprolol Fumarate - Bisoprolol Fumarate tablet prescribing information
INDICATIONS AND USAGE
Bisoprolol fumarate tablets are indicated in the management of hypertension. It may be used alone or in combination with other antihypertensive agents.
DOSAGE AND ADMINISTRATION
The dose of bisoprolol fumarate tablets must be individualized to the needs of the patient. The usual starting dose is 5 mg once daily. In some patients, 2.5 mg may be an appropriate starting dose (see Bronchospastic Disease in WARNINGS ). If the antihypertensive effect of 5 mg is inadequate, the dose may be increased to 10 mg and then, if necessary, to 20 mg once daily.
Patients with Renal or Hepatic Impairment
In patients with hepatic impairment (hepatitis or cirrhosis) or renal dysfunction (creatinine clearance less than 40 mL/min), the initial daily dose should be 2.5 mg and caution should be used in dose-titration. Since limited data suggest that bisoprolol fumarate is not dialyzable, drug replacement is not necessary in patients undergoing dialysis.
Geriatric Patients
It is not necessary to adjust the dose in the elderly, unless there is also significant renal or hepatic dysfunction (see above and Geriatric Use in PRECAUTIONS ).
Pediatric Patients
There is no pediatric experience with bisoprolol fumarate tablets.
CONTRAINDICATIONS
Bisoprolol fumarate tablets are contraindicated in patients with cardiogenic shock, overt cardiac failure, second or third degree AV block, and marked sinus bradycardia.
ADVERSE REACTIONS
Safety data are available in more than 30,000 patients or volunteers. Frequency estimates and rates of withdrawal of therapy for adverse events were derived from two U.S. placebo-controlled studies.
In Study A, doses of 5, 10, and 20 mg bisoprolol fumarate were administered for 4 weeks. In Study B, doses of 2.5, 10, and 40 mg of bisoprolol fumarate were administered for 12 weeks. A total of 273 patients were treated with 5-20 mg of bisoprolol fumarate; 132 received placebo.
Withdrawal of therapy for adverse events was 3.3% for patients receiving bisoprolol fumarate and 6.8% for patients on placebo. Withdrawals were less than 1% for either bradycardia or fatigue/lack of energy.
The following table presents adverse experiences, whether or not considered drug related, reported in at least 1% of patients in these studies, for all patients studied in placebo-controlled clinical trials (2.5-40 mg), as well as for a subgroup that was treated with doses within the recommended dosage range (5-20 mg). Of the adverse events listed in the table, bradycardia, diarrhea, asthenia, fatigue, and sinusitis appear to be dose related.
| Body System/Adverse Experience | All Adverse Experiences (% a ) Bisoprolol Fumarate | ||
|---|---|---|---|
| Placebo (n=132) % | 5-20 mg (n=273) % | 2.5-40 mg (n=404) % | |
Skin | |||
| 1.5 | 0.7 | 1.0 |
Musculoskeletal | |||
| 2.3 | 2.2 | 2.7 |
Central Nervous System | |||
| 3.8 | 2.9 | 3.5 |
| 11.4 | 8.8 | 10.9 |
| 0.8 | 1.1 | 1.5 |
Autonomic Nervous System | |||
| 1.5 | 0.7 | 1.3 |
Heart Rate/Rhythm | |||
| 0 | 0.4 | 0.5 |
Psychiatric | |||
| 0 | 0 | 0 |
| 2.3 | 1.5 | 2.5 |
| 0.8 | 0 | 0.2 |
Gastrointestinal | |||
| 1.5 | 2.6 | 3.5 |
| 1.5 | 1.5 | 2.2 |
| 0 | 1.1 | 1.5 |
Respiratory | |||
| 0 | 0 | 0 |
| 4.5 | 2.6 | 2.5 |
| 0.8 | 1.1 | 1.5 |
| 2.3 | 2.2 | 2.2 |
| 3.0 | 2.9 | 4.0 |
| 1.5 | 2.2 | 2.2 |
| 3.8 | 4.8 | 5.0 |
Body as a Whole | |||
| 0 | 0.4 | 1.5 |
| 0.8 | 1.1 | 1.5 |
| 1.5 | 6.6 | 8.2 |
| 3.8 | 3.7 | 3.0 |
a percentage of patients with event | |||
The following is a comprehensive list of adverse experiences reported with bisoprolol fumarate in worldwide studies, or in postmarketing experience (in italics):
Central Nervous System
Dizziness, unsteadiness, vertigo, syncope, headache, paresthesia, hypoesthesia, hyperesthesia, somnolence, sleep disturbances, anxiety/restlessness, decreased concentration/memory.
Autonomic Nervous System
Dry mouth.
Cardiovascular
Bradycardia, palpitations and other rhythm disturbances, cold extremities, claudication, hypotension, orthostatic hypotension, chest pain, congestive heart failure, dyspnea on exertion.
Psychiatric
Vivid dreams, insomnia, depression.
Gastrointestinal
Gastric/epigastric/abdominal pain, gastritis, dyspepsia, nausea, vomiting, diarrhea, constipation, peptic ulcer.
Musculoskeletal
Muscle/joint pain, arthralgia , back/neck pain, muscle cramps, twitching/tremor.
Skin
Rash, acne, eczema, psoriasis , skin irritation, pruritus, flushing, sweating, alopecia, dermatitis, angioedema, exfoliative dermatitis , cutaneous vasculitis.
Special Senses
Visual disturbances, ocular pain/pressure, abnormal lacrimation, tinnitus, decreased hearing , earache, taste abnormalities.
Metabolic
Gout.
Respiratory
Asthma/bronchospasm, bronchitis, coughing, dyspnea, pharyngitis, rhinitis, sinusitis, URI.
Genitourinary
Decreased libido/impotence, Peyronie’s disease , cystitis, renal colic, polyuria.
Hematologic
Purpura.
General
Fatigue, asthenia, chest pain, malaise, edema, weight gain, angioedema.
In addition, a variety of adverse effects have been reported with other beta-adrenergic blocking agents and should be considered potential adverse effects of bisoprolol fumarate tablets:
Central Nervous System
Reversible mental depression progressing to catatonia, hallucinations, an acute reversible syndrome characterized by disorientation to time and place, emotional lability, slightly clouded sensorium.
Allergic
Fever, combined with aching and sore throat, laryngospasm, respiratory distress.
Hematologic
Agranulocytosis, thrombocytopenia, thrombocytopenic purpura.
Gastrointestinal
Mesenteric arterial thrombosis, ischemic colitis.
Miscellaneous
The oculomucocutaneous syndrome associated with the beta-blocker practolol has not been reported with bisoprolol fumarate tablets during investigational use or extensive foreign marketing experience.
DESCRIPTION
Bisoprolol fumarate tablets are a synthetic, beta 1 -selective (cardioselective) adrenoceptor blocking agent. The chemical name for bisoprolol fumarate is (±)-1-[4-[[2-(1-Methylethoxy)ethoxy]methyl]phenoxy]-3‑ [(1-methylethyl)amino]-2-propanol( E )-2-butenedioate (2:1) (salt). It possesses an asymmetric carbon atom in its structure and is provided as a racemic mixture. The S(-) enantiomer is responsible for most of the beta-blocking activity. Its empirical formula is (C 18 H 31 NO 4 ) 2 •C 4 H 4 O 4 and its structure is:
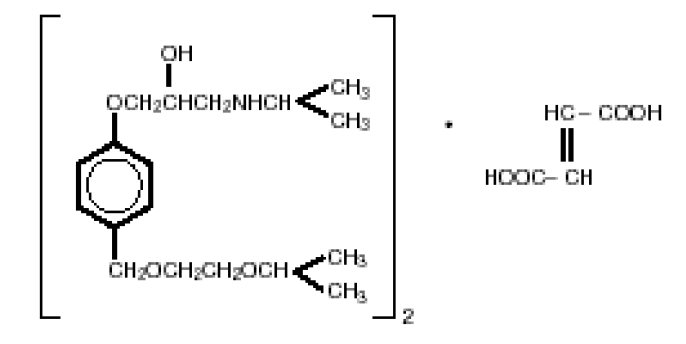
Bisoprolol fumarate, USP, has a molecular weight of 766.97. It is a white crystalline powder which is approximately equally hydrophilic and lipophilic, and is readily soluble in water, methanol, ethanol, and chloroform.
Bisoprolol fumarate tablets are available as 5 and 10 mg tablets for oral administration.
Inactive ingredients include Colloidal Silicon Dioxide, Crospovidone, Dicalcium Phosphate Anhydrous, Magnesium Stearate, Mannitol, Microcrystalline Cellulose, Pregelatinized Starch. The coating materials for all strengths contain Macrogol/PEG, Polyvinyl Alcohol-part hydrolyzed, Talc, Titanium Dioxide. The 5 mg tablets also contain Red Iron Oxide.
FDA approved dissolution test specifications differ from USP.
CLINICAL PHARMACOLOGY
Bisoprolol fumarate tablets are a beta 1 -selective (cardioselective) adrenoceptor blocking agent without significant membrane stabilizing activity or intrinsic sympathomimetic activity in its therapeutic dosage range.
Cardioselectivity is not absolute, however, and at higher doses (≥ 20 mg) bisoprolol fumarate also inhibits beta 2 -adrenoceptors, chiefly located in the bronchial and vascular musculature; to retain selectivity it is therefore important to use the lowest effective dose.
Pharmacokinetics and Metabolism
The absolute bioavailability after a 10 mg oral dose of bisoprolol fumarate is about 80%. Absorption is not affected by the presence of food. The first pass metabolism of bisoprolol fumarate is about 20%.
Binding to serum proteins is approximately 30%. Peak plasma concentrations occur within 2-4 hours of dosing with 5 to 20 mg, and mean peak values range from 16 ng/mL at 5 mg to 70 ng/mL at 20 mg. Once daily dosing with bisoprolol fumarate results in less than twofold intersubject variation in peak plasma levels. The plasma elimination half-life is 9-12 hours and is slightly longer in elderly patients, in part because of decreased renal function in that population. Steady state is attained within 5 days of once daily dosing. In both young and elderly populations, plasma accumulation is low; the accumulation factor ranges from 1.1 to 1.3, and is what would be expected from the first order kinetics and once daily dosing. Plasma concentrations are proportional to the administered dose in the range of 5 to 20 mg. Pharmacokinetic characteristics of the two enantiomers are similar.
Bisoprolol fumarate is eliminated equally by renal and non-renal pathways with about 50% of the dose appearing unchanged in the urine and the remainder appearing in the form of inactive metabolites. In humans, the known metabolites are labile or have no known pharmacologic activity. Less than 2% of the dose is excreted in the feces. Bisoprolol fumarate is not metabolized by cytochrome P450 II D6 (debrisoquin hydroxylase).
In subjects with creatinine clearance less than 40 mL/min, the plasma half-life is increased approximately threefold compared to healthy subjects.
In patients with cirrhosis of the liver, the elimination of bisoprolol fumarate tablets is more variable in rate and significantly slower than that in healthy subjects, with plasma half-life ranging from 8.3 to 21.7 hours.
Pharmacodynamics
The most prominent effect of bisoprolol fumarate tablets is the negative chronotropic effect, resulting in a reduction in resting and exercise heart rate. There is a fall in resting and exercise cardiac output with little observed change in stroke volume, and only a small increase in right atrial pressure, or pulmonary capillary wedge pressure at rest or during exercise.
Findings in short-term clinical hemodynamics studies with bisoprolol fumarate tablets are similar to those observed with other beta-blocking agents.
The mechanism of action of its antihypertensive effects has not been completely established. Factors which may be involved include:
1) Decreased cardiac output,
2) Inhibition of renin release by the kidneys,
3) Diminution of tonic sympathetic outflow from the vasomotor centers in the brain.
In normal volunteers, bisoprolol fumarate tablets therapy resulted in a reduction of exercise- and isoproterenol-induced tachycardia. The maximal effect occurred within 1-4 hours post-dosing. Effects persisted for 24 hours at doses equal to or greater than 5 mg.
Electrophysiology studies in man have demonstrated that bisoprolol fumarate tablets significantly decreases heart rate, increases sinus node recovery time, prolongs AV node refractory periods, and, with rapid atrial stimulation, prolongs AV nodal conduction.
Beta 1 -selectivity of bisoprolol fumarate tablets has been demonstrated in both animal and human studies. No effects at therapeutic doses on beta 2 -adrenoceptor density have been observed. Pulmonary function studies have been conducted in healthy volunteers, asthmatics, and patients with chronic obstructive pulmonary disease (COPD). Doses of bisoprolol fumarate tablets ranged from 5 to 60 mg, atenolol from 50 to 200 mg, metoprolol from 100 to 200 mg, and propranolol from 40 to 80 mg. In some studies, slight, asymptomatic increases in airways resistance (AWR) and decreases in forced expiratory volume (FEV 1 ) were observed with doses of bisoprolol fumarate 20 mg and higher, similar to the small increases in AWR also noted with the other cardioselective beta-blockers. The changes induced by beta- blockade with all agents were reversed by bronchodilator therapy.
Bisoprolol fumarate tablets had minimal effect on serum lipids during antihypertensive studies. In U.S. placebo-controlled trials, changes in total cholesterol averaged +0.8% for bisoprolol fumarate-treated patients, and +0.7% for placebo. Changes in triglycerides averaged +19% for bisoprolol fumarate-treated patients, and +17% for placebo.
Bisoprolol fumarate tablets have also been given concomitantly with thiazide diuretics. Even very low doses of hydrochlorothiazide (6.25 mg) were found to be additive with bisoprolol fumarate in lowering blood pressure in patients with mild-to-moderate hypertension.
CLINICAL STUDIES
In two randomized double-blind placebo-controlled trials conducted in the U.S., reductions in systolic and diastolic blood pressure and heart rate 24 hours after dosing in patients with mild-to-moderate hypertension are shown below. In both studies, mean systolic/diastolic blood pressures at baseline were approximately 150/100 mm Hg, and mean heart rate was 76 bpm. Drug effect is calculated by subtracting the placebo effect from the overall change in blood pressure and heart rate.
Sitting Systolic/Diastolic Pressure (BP) and Heart Rate (HR) Mean Decrease (D) After 3 to 4 Weeks | ||||
Study A | Bisoprolol Fumarate | |||
Placebo | 5 mg | 10 mg | 20 mg | |
n= | 61 | 61 | 61 | 61 |
Total ΔBP (mm Hg) | 5.4/3.2 | 10.4/8.0 | 11.2/10.9 | 12.8/11.9 |
Drug Effect a | 5.0/4.8 | 5.8/7.7 | 7.4/8.7 | |
Total ΔHR (bpm) | 0.5 | 7.2 | 8.7 | 11.3 |
Drug Effect a | 6.7 | 8.2 | 10.8 | |
Study B | Bisoprolol Fumarate | |||
Placebo | 2.5 mg | 10 mg | ||
n= | 56 | 59 | 62 | |
Total ΔBP (mm Hg) | 3.0/3.7 | 7.6/8.1 | 13.5/11.2 | |
Drug Effect a | 4.6/4.4 | 10.5/7.5 | ||
Total ΔHR (bpm) | 1.6 | 3.8 | 10.7 | |
Drug Effect a | 2.2 | 9.1 | ||
a Observed total change from baseline minus placebo. | ||||
Blood pressure responses were seen within one week of treatment and changed little thereafter. They were sustained for 12 weeks and for over a year in studies of longer duration. Blood pressure returned to baseline when bisoprolol fumarate was tapered over two weeks in a long-term study.
Overall, significantly greater blood pressure reductions were observed on bisoprolol fumarate than on placebo regardless of race, age, or gender. There were no significant differences in response between black and nonblack patients.
HOW SUPPLIED
Bisoprolol fumarate tablets, USP, are supplied as 5 mg and 10 mg tablets.
The 5 mg tablet is pink color coated tablet, capsule shaped, convex scored tablets debossed with “6|6” on one side and “S” on the other side, supplied as follows:
bottles of 30 (Unit-of-Use) NDC 43547-616-03
bottles of 100 NDC 43547-616-10
bottles of 500 NDC 43547-616-50
The 10 mg tablet is white color coated tablet, round shaped, convex tablets debossed with “6 7” on one side and “S” on the other side, supplied as follows:
bottles of 30 (Unit-of-Use) NDC 43547-617-03
bottles of 100 NDC 43547-617-10
bottles of 500 NDC 43547-617-50
Store at 20 o C to 25 o C (68 o Fto 77 o F); excursions permitted to 15 o C to 30 o C (59 o Fto 86 o F) [see USP Controlled Room Temperature].
Protect from moisture.
Dispense in tight, light resistant containers.
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Manufactured by:
Prinston Laboratories
Charlotte, NC 28206, USA
Revised: 06/2023
9040560-01