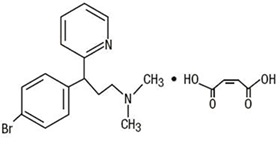
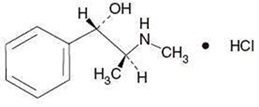
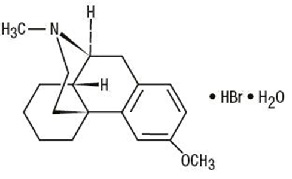
Get your patient on Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide - Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide syrup (Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide)
Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide - Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide syrup prescribing information
Check medication interactionsReview interactions as part of your prescribing workflow
Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide - Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide syrup PubMed™ news
No results.Review your filter criteria.
Show the latest PubMed™ articles for Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide - Brompheniramine Maleate, Pseudoephedrine Hydrochloride, And Dextromethorphan Hydrobromide syrup