Get your patient on Bupivacaine Hydrochloride
Bupivacaine Hydrochloride prescribing information
WARNING: RISK OF CARDIAC ARREST WITH USE OF BUPIVACAINE HYDROCHLORIDE INJECTION IN OBSTETRICAL ANESTHESIA
WARNING: RISK OF CARDIAC ARREST WITH USE OF BUPIVACAINE HYDROCHLORIDE INJECTION IN OBSTETRICAL ANESTHESIA
There have been reports of cardiac arrest with difficult resuscitation or death during use of bupivacaine hydrochloride injection for epidural anesthesia in obstetrical patients. In most cases, this has followed use of the 0.75% (7.5 mg/mL) concentration. Resuscitation has been difficult or impossible despite apparently adequate preparation and appropriate management. Cardiac arrest has occurred after convulsions resulting from systemic toxicity, presumably following unintentional intravascular injection. The 0.75% (7.5 mg/mL) concentration of bupivacaine hydrochloride injection is not recommended for obstetrical anesthesia and should be reserved for surgical procedures where a high degree of muscle relaxation and prolonged effect are necessary [ see Warnings and Precautions (5.1) ].
INDICATIONS AND USAGE
Bupivacaine hydrochloride injection is indicated in adults for the production of local or regional anesthesia or analgesia for surgery and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of bupivacaine hydrochloride injection are recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration (2.2) ].
Limitations of Use
Not all blocks are indicated for use with bupivacaine hydrochloride injection given clinically significant risks associated with use [see Dosage and Administration (2.2) , Contraindications (4) , Warnings and Precautions (5.1 , 5.4 , 5.5 , 5.7 , 5.9 ) ].
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Bupivacaine hydrochloride injection is a clear, colorless solution available as:
• 0.25% (25 mg/10 mL) (2.5 mg/mL) in single-dose vials.
• 0.25% (75 mg/30 mL) (2.5 mg/mL) in single-dose vials.
• 0.5% (50 mg/10 mL) (5 mg/mL) in single-dose vials.
• 0.5% (150 mg/30 mL) (5 mg/mL) in single-dose vials.
• 0.75% (75 mg/10 mL) (7.5 mg/mL) in single-dose vials.
• 0.75% (225 mg/30 mL) (7.5 mg/mL) in single-dose vials.
USE IN SPECIFIC POPULATIONS
CONTRAINDICATIONS
Bupivacaine hydrochloride injection is contraindicated in:
• obstetrical paracervical block anesthesia. Its use in this technique has resulted in fetal bradycardia and death.
• intravenous regional anesthesia (Bier Block) [ see Warnings and Precautions (5.7) ].
• patients with a known hypersensitivity to bupivacaine or to any local anesthetic agent of the amide-type or to other components of bupivacaine hydrochloride injection.
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
The following clinically significant adverse reactions have been reported and described in the Warnings and Precautions section of the labeling:
• Cardiac Arrest in Obstetrical Anesthesia [see Warnings and Precautions (5.1)]
• Dose-Related Toxicity [see Warnings and Precautions (5.2)]
• Methemoglobinemia [see Warnings and Precautions (5.3)]
• Chondrolysis with Intra-Articular Infusion [see Warnings and Precautions (5.5)]
• Cardiac Arrest with Intravenous Regional Anesthesia Use [see Contraindications (4), Warnings and Precautions (5.7)]
• Systemic Toxicities with Unintended Intravascular or Intrathecal Injection [see Warnings and Precautions (5.9)]
• Respiratory Arrest Following Retrobulbar Block [see Warnings and Precautions (5.15)]
The following adverse reactions from voluntary reports or clinical studies have been reported with bupivacaine. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions to bupivacaine hydrochloride injection are characteristic of those associated with other amide-type local anesthetics. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage, unintentional intravascular injection, or slow metabolic degradation.
The most commonly encountered acute adverse reactions that demand immediate counter-measures were related to the CNS and the cardiovascular system. These adverse reactions were generally dose-related and due to high plasma levels which may have resulted from overdosage, rapid absorption from the injection site, diminished tolerance, or from unintentional intravascular injection of the local anesthetic solution. In addition to systemic dose-related toxicity, unintentional intrathecal injection of drug during the intended performance of caudal or lumbar epidural block or nerve blocks near the vertebral column (especially in the head and neck region) has resulted in underventilation or apnea (“Total or High Spinal”). Also, hypotension due to loss of sympathetic tone and respiratory paralysis or underventilation due to cephalad extension of the motor level of anesthesia have occurred. This has led to secondary cardiac arrest when untreated.
Nervous System Disorders : Adverse reactions were characterized by excitation and/or depression of the central nervous system and included restlessness, anxiety, dizziness, tinnitus, blurred vision, tremors, convulsions, drowsiness, unconsciousness, respiratory arrest, nausea, vomiting, chills, pupillary constriction.
In the practice of caudal or lumbar epidural block, unintentional penetration of the subarachnoid space by the catheter or needle has occurred. Subsequent adverse effects may have depended partially on the amount of drug administered intrathecally and the physiological and physical effects of a dural puncture. A high spinal has been characterized by paralysis of the legs, loss of consciousness, respiratory paralysis, and bradycardia.
Neurologic effects following epidural or caudal anesthesia have included spinal block of varying magnitude (including high or total spinal block); hypotension secondary to spinal block; urinary retention; fecal and urinary incontinence; loss of perineal sensation and sexual function; persistent anesthesia, paresthesia, weakness, paralysis of the lower extremities and loss of sphincter control, all of which had slow, incomplete, or no recovery; headache; backache; septic meningitis; meningismus; slowing of labor; increased incidence of forceps delivery; and cranial nerve palsies due to traction on nerves from loss of cerebrospinal fluid.
Neurologic effects following other procedures or routes of administration have included persistent anesthesia, paresthesia, weakness, paralysis, all with slow, incomplete, or no recovery.
Convulsions : Incidence varied with the procedure used and the total dose administered. In a survey of studies of epidural anesthesia, overt toxicity progressing to convulsions occurred in approximately 0.1% of local anesthetic administrations. The incidences of adverse neurologic reactions associated with the use of local anesthetics may be related to the total dose of local anesthetic administered and are also dependent upon the particular drug used, the route of administration, and the physical status of the patient.
Cardiac Disorders : High doses or unintentional intravascular injection have led to high plasma levels and related depression of the myocardium, decreased cardiac output, heartblock, hypotension, bradycardia, ventricular arrhythmias, including ventricular tachycardia and ventricular fibrillation, and cardiac arrest [ see Warnings and Precautions (5.9) ].
Immune System Disorders : Allergic-type reactions have occurred as a result of sensitivity to bupivacaine or to other formulation ingredients, such as the antimicrobial preservative methylparaben contained in multiple-dose vials. These reactions were characterized by signs such as urticaria, pruritus, erythema, angioneurotic edema (including laryngeal edema), tachycardia, sneezing, nausea, vomiting, dizziness, syncope, excessive sweating, elevated temperature, and severe hypotension. Cross sensitivity among members of the amide-type local anesthetic group has been reported [see Warnings and Precautions (5.8)].
DRUG INTERACTIONS
DESCRIPTION
Bupivacaine hydrochloride injection contains bupivacaine hydrochloride, an amide local anesthetic, as the active pharmaceutical ingredient. The route of administration for bupivacaine hydrochloride injection is by injection, for infiltration, perineural, caudal, epidural, or retrobulbar use.[ see Warnings and Precautions (5.4) ].
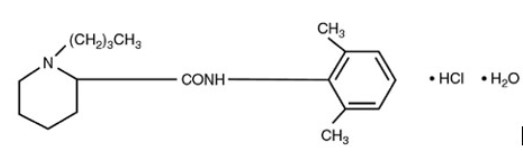
Bupivacaine hydrochloride is 2-piperidinecarboxamide, 1-butyl-N-(2,6-dimethylphenyl)-, monohydrochloride, monohydrate. It is a white crystalline powder that is freely soluble in 95 percent ethanol, soluble in water, and slightly soluble in chloroform or acetone. It has the following structural formula:
Bupivacaine hydrochloride injection is a clear and colorless sterile isotonic solution. Each mL of single-dose vial contains 2.5 mg, 5 mg or 7.5 mg of bupivacaine hydrochloride (equivalent to 2.22 mg, 4.44 mg or 6.66 mg of bupivacaine, respectively), sodium chloride for isotonicity, sodium hydroxide or hydrochloric acid to adjust the pH between 4 and 6.5, in water for injection.
For the Multiple-dose vials, each mL also contains 1 mg methylparaben as preservative.
CLINICAL PHARMACOLOGY 12.1 Mechanism of Action
Bupivacaine blocks the generation and the conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of affected nerve fibers. Clinically, the order of loss of nerve function is as follows: (1) pain, (2) temperature, (3) touch, (4) proprioception, and (5) skeletal muscle tone.
NONCLINICAL TOXICOLOGY
HOW SUPPLIED/STORAGE AND HANDLING
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 ° F to 86 °F). [See USP Controlled Room Temperature.]
Protect from light. This product is clear and colorless. Do not use the solution if it is discolored or if it contains a precipitate.
| Unit of Sale | Concentration | Each |
| 0.25% Contains 2.5 mg bupivacaine hydrochloride per mL. | ||
NDC 83270-162-10 Carton of 10 | 25 mg/10 mL (2.5 mg/mL) | NDC 83270-162-01 single-dose vial |
NDC 83270-165-01 Carton of 1 | 75 mg/30 mL (2.5 mg/mL) | NDC 83270-165-01 single-dose vial |
| 0.5% Contains 5 mg bupivacaine hydrochloride per mL. | ||
| NDC 83270-163-10 Carton of 10 | 50 mg/10 mL (5 mg/mL) | NDC 83270-163-01 single-dose vial |
| NDC 83270-166-01 Carton of 1 | 150 mg/30 mL (5 mg/mL) | NDC 83270-166-01 single-dose vial |
| 0.75% Contains 7.5 mg bupivacaine hydrochloride per mL. | ||
| NDC 83270-164-10 Carton of 10 | 75 mg/10 mL (7.5 mg/mL) | NDC 83270-164-01 single-dose vial |
| NDC 83270-167-01 Carton of 1 | 225 mg/30 mL (7.5 mg/mL) | NDC 83270-167-01 single-dose vial |
For single-dose vials: Discard unused portion.