Get your patient on Carbocaine - Mepivacaine Hydrochloride injection, Solution (Mepivacaine Hydrochloride)
Carbocaine - Mepivacaine Hydrochloride injection, Solution prescribing information
INDICATIONS AND USAGE
CARBOCAINE is indicated for production of local anesthesia for dental procedures by infiltration or nerve block in adults and pediatric patients.
DOSAGE AND ADMINISTRATION
As with all local anesthetics, the dose varies and depends upon the area to be anesthetized, the vascularity of the tissues, individual tolerance and the technique of anesthesia. The lowest dose needed to provide effective anesthesia should be administered. For specific techniques and procedures refer to standard dental manuals and textbooks.
For infiltration and block injections in the upper or lower jaw, the average dose of 1 cartridge will usually suffice.
Each cartridge contains 1.7 mL (34 mg of 2% or 51 mg of 3%).
5.3 cartridges (180 mg of the 2% solution or 270 mg of the 3% solution) are usually adequate to effect anesthesia of the entire oral cavity. Whenever a larger dose seems to be necessary for an extensive procedure, the maximum dose should be calculated according to the patient's weight. A dose of up to 3 mg per pound of body weight may be administered. At any single dental sitting the total dose for all injected sites should not exceed 400 mg in adults.
The maximum pediatric dose should be carefully calculated.
| Maximum dose for pediatric population = | ||
| Child's Weight (lbs.) | × | Maximum Recommended Dose for Adults (400 mg) |
| 150 | ||
The following table, approximating these calculations, may also be used as a guide. This table is based upon a recommended maximum for larger pediatric population of 5.3 cartridges (the maximum recommended adult dose) during any single dental sitting, regardless of the pediatric patient's weight or (for 2% mepivacaine) calculated maximum amount of drug:
| 3% Mepivacaine (Plain) | 2% Mepivacaine 1:20,000 Levonordefrin | |||
|---|---|---|---|---|
| 3 mg/lb (270 mg max.) | 3mg/lb (180 mg max.) | |||
| Weight (lb.) | mg | Number of Cartridges | mg | Number of Cartridges |
| 20 | 60 | 1.2 | 60 | 1.8 |
| 30 | 90 | 1.8 | 90 | 2.6 |
| 40 | 120 | 2.3 | 120 | 3.5 |
| 50 | 150 | 2.9 | 150 | 4.4 |
| 60 | 180 | 3.5 | 180 | 5.3 |
| 80 | 240 | 4.7 | 180 | 5.3 |
| 100 | 270 | 5.3 | 180 | 5.3 |
| 120 | 270 | 5.3 | 180 | 5.3 |
When using CARBOCAINE for infiltration or regional block anesthesia, injection should always be made slowly and with frequent aspiration.
Any unused portion of a cartridge should be discarded.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
CARBOCAINE is contraindicated in patients with a known hypersensitivity to amide-type local anesthetics.
ADVERSE REACTIONS
Reactions to CARBOCAINE are characteristic of those associated with other amide-type local anesthetics. Systemic adverse reactions involving the central nervous system and the cardiovascular system usually result from high plasma levels (which may be due to excessive dosage, rapid absorption, inadvertent intravascular injection, or slow metabolic degradation), injection technique, or volume of injection.
A small number of reactions may result from hypersensitivity, idiosyncrasy or diminished tolerance to normal dosage on the part of the patient.
Persistent paresthesias of the lips, tongue, and oral tissues have been reported with the use of mepivacaine, with slow, incomplete, or no recovery. These post-markeing events have been reported chiefly following nerve blocks in the mandible and have involved the trigeminal nerve and its branches.
Reactions involving the central nervous system are characterized by excitation and/or depression. Nervousness, dizziness, blurred vision, or tremors may occur followed by drowsiness, convulsions, unconsciousness, and possible respiratory arrest. Since excitement may be transient or absent, the first manifestations may be drowsiness merging into unconsciousness and respiratory arrest.
Cardiovascular reactions are depressant. They may be the result of direct drug effect or more commonly in dental practice, the result of vasovagal reaction, particularly if the patient is in the sitting position. Failure to recognize premonitory signs such as sweating, feeling of faintness, changes in pulse or sensorium may result in progressive cerebral hypoxia and seizure or serious cardiovascular catastrophe. Management consists of placing the patient in the recumbent position and administration of oxygen. Vasoactive drugs such as Ephedrine or Methoxamine may be administered intravenously.
Allergic reactions are rare and may occur as a result of sensitivity to the local anesthetic and are characterized by cutaneous lesions of delayed onset or urticaria, edema and other manifestations of allergy. The detection of sensitivity by skin testing is of limited value. As with other local anesthetics, anaphylactoid reactions to Mepivacaine have occurred rarely. The reaction may be abrupt and severe and is not usually dose related. Localized puffiness and swelling may occur.
Clinically Significant Drug Interactions
The administration of local anesthetic solutions containing vasopressors, such as Neo-Cobefrin, Epinephrine or Norepinephrine, to patients receiving tricyclic antidepressants or monoamine oxidase inhibitors may produce severe, prolonged hypertension. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful patient monitoring is essential.
Concurrent administration of vasopressor drugs and of ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents.
Phenothiazines and butyrophenones may reduce or reverse the pressor effect of Epinephrine.
Solutions containing a vasoconstrictor should be used cautiously in the presence of disease which may adversely affect the patient's cardiovascular system. Serious cardiac arrhythmias may occur if preparations containing a vasoconstrictor are employed in patients during or following the administration of potent inhalation anesthetics.
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
| Class | Examples |
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprus-side, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic Agents | cyclophosphamide, flutamide, hy-droxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
MEPIVACAINE SHOULD BE USED WITH CAUTION IN PATIENTS WITH KNOWN DRUG ALLERGIES AND SENSITIVITIES. A thorough history of the patient's prior experience with Mepivacaine or other local anesthetics as well as concomitant or recent drug use should be taken (see CONTRAINDICATIONS ). Patients allergic to methylparaben or para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross-sensitivity to agents of the amide type such as Mepivacaine. Since Mepivacaine is metabolized in the liver and excreted by the kidneys, it should be used cautiously in patients with liver and renal disease.
DESCRIPTION
CARBOCAINE (mepivacaine hydrochloride), a tertiary amine used as a local anesthetic, is 1-methyl-2', 6' - pipecoloxylidide monohydrochloride with the following structural formula:
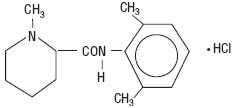 | C 15 H 22 N 2 0.HCl M.W. 282.81 |
It is a white, crystalline, odorless powder soluble in water, but very resistant to both acid and alkaline hydrolysis.
NEO-COBEFRIN, a sympathomimetic amine used as a vasoconstrictor in local anesthetic solution, is (-)-  -(1-Aminoethyl)-3, 4-dihydroxybenzyl alcohol with the following structural formula:
-(1-Aminoethyl)-3, 4-dihydroxybenzyl alcohol with the following structural formula:
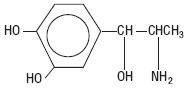 | C 9 H 13 NO 3 M.W. 183.21 |
It is a white or buff-colored crystalline solid, freely soluble in aqueous solutions of mineral acids, but practically insoluble in water.
DENTAL CARTRIDGES MAY NOT BE AUTOCLAVED.
CARBOCAINE 3% (30 mg/mL) injection and CARBOCAINE 2% (20 mg/mL) with NEO-COBEFRIN 1:20,000 injection are sterile solutions for injection.
| COMPOSITION: | CARTRIDGE | |
|---|---|---|
| Each mL contains: | 2% | 3% |
| Mepivacaine hydrochloride | 20 mg | 30 mg |
| Levonordefrin | 0.05mg | - |
| Sodium chloride | 4 mg | 6 mg |
| Potassium metabisulfite | 1.2 mg | - |
| Edetate disodium | 0.25mg | - |
| Sodium hydroxide q.s. ad pH | ||
| Hydrochloric acid | 0.5 mg | - |
| Water for injections qs. ad. | 1 mL | 1 mL |
| The pH of the 2% cartridge solution is adjusted between 3.3 and 5.5 with NaOH. | ||
| The pH of the 3% cartridge solution is adjusted between 4.5 and 6.8 with NaOH. | ||
CLINICAL PHARMACOLOGY
CARBOCAINE stabilizes the neuronal membrane and prevents the initiation and transmission of nerve impulses, thereby effecting local anesthesia.
CARBOCAINE is rapidly metabolized, with only a small percentage of the anesthetic (5 to 10 percent) being excreted unchanged in the urine. CARBOCAINE because of its amide structure, is not detoxified by the circulating plasma esterases. The liver is the principal site of metabolism, with over 50 percent of the administered dose being excreted into the bile as metabolites. Most of the metabolized mepivacaine is probably resorbed in the intestine and then excreted into the urine since only a small percentage is found in the feces. The principal route of excretion is via the kidney. Most of the anesthetic and its metabolites are eliminated within 30 hours. It has been shown that hydroxylation and N-demethylation, which are detoxification reactions, play important roles in the metabolism of the anesthetic. Three metabolites of mepivacaine have been identified from adult humans: two phenols, which are excreted almost exclusively as their glucuronide conjugates, and the N-demethylated compound (2', 6' - pipecoloxylidide).
The onset of action is rapid (30 to 120 seconds in the upper jaw; 1 to 4 minutes in the lower jaw) and CARBOCAINE 3% (30 mg/mL) injection will ordinarily provide operating anesthesia of 20 minutes in the upper jaw and 40 minutes in the lower jaw .
CARBOCAINE 2% (20 mg/mL) with Neo-Cobefrin 1:20,000 injection provides anesthesia of longer duration for more prolonged procedures, 1 hour to 2.5 hours in the upper jaw and 2.5 hours to 5.5 hours in the lower jaw.
CARBOCAINE does not ordinarily produce irritation or tissue damage.
Neo-Cobefrin is a sympathomimetic amine used as a vasoconstrictor in local anesthetic solutions. It has pharmacologic activity similar to that of Epinephrine but it is more stable than Epinephrine. In equal concentrations, Neo-Cobefrin is less potent than Epinephrine in raising blood pressure, and as a vasoconstrictor.
HOW SUPPLIED
CARBOCAINE 3% (30 mg/mL) injection (mepivacaine hydrocholoride injection, USP) is available in cartons containing 5 blisters of 10 × 1.7 mL single-dose dental cartridges, 50 per carton (NDC 0362-0753-05). CARBOCAINE 2% (20 mg/mL) with NEO-COBEFRIN 1:20,000 injection, USP (mepivacaine hydrochloride and levonordefrin injection, USP) is available in cartons containing 5 blisters of 10 × 1.7 mL single-dose dental cartridges, 50 per carton (NDC 0362-0931-05).
Both solutions should be stored at controlled room temperature, below 25° C (77° F). Protect from light. Do not permit to freeze. For protection from light, retain in box until time of use. Once opened, the box should be reclosed by closing the flap. The mepivacaine 2% (20 mg/mL) solution should not be used if its color is pinkish or darker than slightly yellow or it contains a precipitate. Cartridge warmers should not be used with CARBOCAINE products.