Get your patient on Carbzah - Carbinoxamine Maleate solution (Carbinoxamine Maleate)
Carbzah - Carbinoxamine Maleate solution prescribing information
INDICATIONS AND USAGE
Carbinoxamine maleate is effective for the symptomatic treatment of:
Seasonal and perennial allergic rhinitis.
Vasomotor rhinitis.
Allergic conjunctivitis due to inhalant allergens and foods.
Mild, uncomplicated allergic skin manifestations of urticaria and angioedema.
Dermatographism.
As therapy for anaphylactic reactions adjunctive to epinephrine and other standard measures after the acute manifestations have been controlled.
Amelioration of the severity of allergic reactions to blood or plasma.
DOSAGE AND ADMINISTRATION
Carbinoxamine maleate is contraindicated in children younger than 2 years of age (see CONTRAINDICATIONS ).
Carbinoxamine maleate should be taken on an empty stomach with water.
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND THE RESPONSE OF THE PATIENT.
Carbinoxamine maleate dosage should be based on the severity of the condition and the response of the patient. The drug is well tolerated in adults in doses as high as 24 mg daily, in divided doses, over prolonged periods. On the other hand, some patients respond to as little as 4 mg daily.
Clinical experience suggests the following dosage schedules:
Oral Solution
Usual Adult Dosage:
1 or 2 teaspoonfuls (4 to 8 mg) 3 to 4 times daily
Usual Child's Dosage (approximately 0.2 to 0.4 mg/kg/day, divided into 3 to 4 doses):
Six to eleven years– ½ to 1 teaspoonful (2 to 4 mg) 3 to 4 times daily.
Dosing for children 2 to 5 years of age should be based on weight whenever possible. The usual dosage for children 2 to 5 years of age is
approximately 0.2 to 0.4 mg/kg/day, divided into 3 to 4 daily doses. In general, this corresponds to a dose of ¼ to ½ teaspoonful (1 to 2 mg) 3
to 4 times daily.
CONTRAINDICATIONS
Carbinoxamine maleate is contraindicated in children younger than 2 years of age.
Carbinoxamine maleate is contraindicated in nursing mothers.
Carbinoxamine maleate is contraindicated in patients who are hypersensitive to the drug or on monoamine oxidase inhibitor therapy. (See Drug Interactions section).
ADVERSE REACTIONS
The most frequent adverse reactions are underlined:
Body as a Whole: Urticaria, drug rash, anaphylactic shock, photosensitivity, excessive perspiration, chills, dryness of mouth, nose and throat.
Cardiovascular: Hypotension, headache, palpitations, tachycardia, extrasystoles.
Hematologic: Hemolytic anemia, thrombocytopenia, agranulocytosis.
Central Nervous System: Sedation , sleepiness , dizziness , disturbed coordination , fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesia, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions.
Gastrointestinal: Epigastric distress , anorexia, nausea, vomiting, diarrhea, constipation.
Urogenital: Urinary frequency, difficult urination, urinary retention, early menses.
Respiratory: Thickening of bronchial secretions , tightness of chest and wheezing, nasal stuffiness.
To report SUSPECTED ADVERSE REACTIONS, contact IPG Pharmaceuticals, Inc. at 1-888-711-7116 or FDA at 1-800-FDA-1088 or www. fda.gov/medwatch.
Drug Interactions
Monoamine oxidase inhibitors prolong and intensify the anticholinergic (drying) effects of antihistamines.
Carbinoxamine maleate has additive effects with alcohol and other CNS depressants (hypnotics sedatives, tranquilizers, etc.).
DESCRIPTION
Carbinoxamine maleate is a histamine-H 1 receptor blocking agent.
Each 5 mL (teaspoonful) of oral solution contains 4 mg carbinoxamine maleate and the following inactive ingredients: artificial bubble gum flavor, citric acid (anhydrous), glycerin, methylparaben, propylene glycol, propylparaben, purified water, sodium citrate (hydrous) and sorbitol solution.
Carbinoxamine maleate is freely soluble in water. Its structure is:
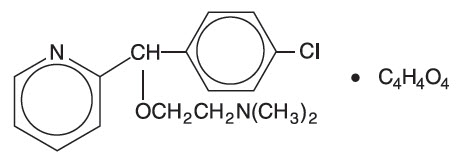
2-[(4-chlorophenyl)-2-pyridinylmethoxy]- N ,N- dimethylethanamine (Z)-2-butenedioate (1:1)
| C 16 H 19 CIN 2 O∙C 4 H 4 O 4 | MW = 406.86 |
CLINICAL PHARMACOLOGY
Mechanism of Actions
Carbinoxamine maleate, an ethanolamine derivative, is an antihistamine with anticholinergic (drying) and sedative properties. Carbinoxamine appears to compete with histamine (type H1) for receptor sites on effector cells in the gastrointestinal tract, blood vessels and respiratory tract.
Pharmacokinetics and Metabolism
Carbinoxamine is well absorbed from the GI tract and appears to be extensively metabolized by the liver, and excreted in the urine as inactive metabolites within 24 hours. Virtually no intact drug is extended in the urine.
In a study comparing a controlled-release suspension and a solution of carbinoxamine, healthy volunteers were administered a single dose of 8 mg carbinoxamine. A time to maximum concentration (Tmax) was between 1.5 hours to 5 hours, a peak plasma concentration (Cmax) of about 24 ng/mL was observed and extent of exposure (AUC) was about 286 ng hr/mL. The serum half-life is reported to be 10 to 20 hours.
Drug/Food Interactions
Carbinoxamine should not be used in patients with hypersensitivity to carbinoxamine. Carbinoxamine may increase the effects of other drugs such as barbiturates, TCAs, MAO inhibitors such as Phenelzine (Nardil), Tranylcypromine (Parnate), or Selegiline (Eldepryl), alcohol, other antihistamines, and CNS depressants. Carbinoxamine can be taken with or without food.
Cardiovascular Effects
Cardiac effects, including prolongation of QT interval have not been adequately studied. Unlike other newer antihistamines, severe adverse cardiovascular effects are uncommon, and usually limited to over dosage situations.
Special Populations
Pediatric Patients
Carbinoxamine should not be used in newborn or premature infants. Neonates have an increased susceptibility to anticholinergic side effects, such as CNS excitation, which may lead to convulsions.
Pregnancy and Lactation
Safe use of carbinoxamine during pregnancy has not been established. Therefore, carbinoxamine should not be used in women who are, or may become pregnant. Carbinoxamine is in the FDA pregnancy Category C.
Women who are breast-feeding should avoid use of carbinoxamine, since small amounts appear to be distributed into breast milk.
Geriatric Patients
Carbinoxamine is more likely to cause dizziness, sedation, and hypotension in elderly patients. The incidence of adverse reactions is higher in the elderly; therefore, a dosing adjustment may be necessary in this sub-population.
HOW SUPPLIED
Carbinoxamine Maleate Oral Solution, 4 mg/5 mL is supplied as clear, colorless liquid with a bubble gum aroma, and is supplied in 4 fl. oz. bottles, NDC 71085-080-07 and in 16 fl. oz. bottles, NDC 71085-080-07.
Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container with a child-resistant closure as defined in the official compendium.
Mechanism of Actions
Carbinoxamine maleate, an ethanolamine derivative, is an antihistamine with anticholinergic (drying) and sedative properties. Carbinoxamine appears to compete with histamine (type H1) for receptor sites on effector cells in the gastrointestinal tract, blood vessels and respiratory tract.