Get your patient on Cardamyst (Etripamil)
Cardamyst prescribing information
INDICATIONS AND USAGE
CARDAMYST is indicated for the conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults.
DOSAGE AND ADMINISTRATION
- For intranasal use only (2.1 ).
- Initial dosage: A dose of 70 mg is administered as two nasal sprays, one spray into each nostril. Each nasal spray device delivers two sprays. The two sprays together contain a total of 70 mg etripamil (2.1 ).
- Repeat dosage (if needed): Should symptoms persist for 10 minutes after administration of CARDAMYST, take a second dose of 70 mg administered as two nasal sprays, one spray into each nostril. Do not exceed 140 mg in a 24-hour period (2.1 ).
Recommended Dosage
Administer as soon as possible after PSVT symptom onset.
Administer CARDAMYST by the nasal route only.
Each CARDAMYST device delivers two sprays for a total of 70 mg.
Recommended Dosage:
Using one nasal spray device, administer one spray into each nostril for a total initial dose of 70 mg. If symptoms persist after 10 minutes, use the second nasal spray device to administer a second dose of one spray into each nostril (70 mg total). Patients and caregivers should call their healthcare provider or seek emergency medical help if symptoms do not improve within 20 minutes after a second dose. Do not exceed 140 mg in a 24-hour period. See Instructions for Use for proper nasal spray technique.
If a full initial dose (i.e., 2 sprays, one in each nostril) is not administered due to device malfunction or misuse, the patient should wait at least 10 minutes before self-administering a second dose, if needed.
DOSAGE FORMS AND STRENGTHS
Nasal Spray: Clear colorless to yellow solution. 70 mg of etripamil per device.
USE IN SPECIFIC POPULATIONS
- Lactation: A lactating woman should pump and discard breastmilk for 12 hours after CARDAMYST administration (8.2 ).
Pregnancy
Risk Summary
There are no available data on the use of CARDAMYST during pregnancy to inform a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Reproductive studies conducted with intravenous administration of etripamil in pregnant rats and rabbits during organogenesis did not show any evidence of fetal harm or malformations in rats at exposures up to approximately 3x the maximum concentration (C max ) and 0.4x the AUC at the maximum recommended human dose (MRHD) and in rabbits at exposures approximately equivalent to the C max and 10x the AUC at the MRHD, at which maternal toxicities were observed (see Data ).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%-4% and 15%-20%, respectively.
Data
Animal Data
In pregnant rats, intravenous administration of etripamil throughout the period of organogenesis did not result in any adverse effects on embryofetal development at doses up to 0.375 mg/kg/day, approximately 3x the C max and 0.4x the AUC at the MRHD.
In pregnant rabbits, intravenous administration of etripamil throughout the period of organogenesis did not result in embryofetal abnormalities at doses up to 0.1 mg/kg/day, approximately equivalent to the C max and 10x the AUC at the MRHD. Abortion in one animal was noted at the high dose of 0.1 mg/kg/day, a dose that caused maternal toxicity.
In the pre- and post-natal toxicity study in rats, intravenous administration of etripamil from gestation day 7 through the lactation period (post-partum day 20), did not show any adverse effects on pre- and postnatal development at doses up to 0.374 mg/kg/day, approximately 3x the C max and 0.4x the AUC at the MRHD. Post-implantation loss was noted at 0.374 mg/kg/day, a dose that also caused significant maternal toxicity, including mortality, transient adverse clinical signs, and body weight reduction.
Lactation
Risk Summary
There are no data on the presence of etripamil in human milk or animal milk. However, the structurally related compound, verapamil, is known to be present in human milk. There are no data on the effects of etripamil on the breastfed infant or on milk production. Because the presence of etripamil in breastmilk has not been characterized, and there is a potential for adverse reactions in the breastfed infant including hypotension and bradycardia, lactating women should interrupt breastfeeding and pump and discard milk for 12 hours (approximately 5 terminal half-lives) after treatment with CARDAMYST.
Pediatric Use
The safety and effectiveness of CARDAMYST have not been established in the pediatric population.
Etripamil is structurally similar to another drug in the same pharmacologic class that has been associated with a high risk of potentially non-reversible electromechanical dissociation or cardiovascular collapse in pediatric patients less than 1 year of age, including neonates.
Geriatric Use
Of the total number of patients in clinical trials of CARDAMYST, 41% (681/1662) were 60 years of age and over, and 14% (238/1662) were 70 years of age and over. No meaningful differences in safety or effectiveness were observed between these patients and younger groups.
CONTRAINDICATIONS
CARDAMYST is contraindicated in patients with:
- Hypersensitivity to CARDAMYST or any of its components.
- Heart failure – New York Heart Association (NYHA) Class II to IV.
- Wolff-Parkinson-White (WPW), Lown-Ganong-Levine (LGL) syndromes, or manifest pre-excitation (delta wave) on a 12-lead electrocardiogram (ECG).
- Sick sinus syndrome without a permanent pacemaker.
- Second degree atrioventricular (AV) Mobitz 2 block or higher degree of AV block.
WARNINGS AND PRECAUTIONS
- Syncope: May cause dizziness and/or syncope, especially in patients with a history of syncope. Administer in a sitting position (5.1 ).
Syncope Related to Hemodynamic Effects
Because of effects on blood pressure, heart rate, and cardiac conduction, CARDAMYST may cause dizziness and/or syncope, especially in patients with a history of syncope and high-grade AV block or sinus node dysfunction, or those with a history of syncope during an episode of PSVT. In clinical trials, a small percentage of patients (0.4%) experienced clinically significant hypotension during test dosing prior to randomization, which precluded further participation in the study. Patients with a history of hypotensive episodes or those at increased risk for hemodynamic instability should be monitored appropriately when initiating CARDAMYST.
If syncope occurs, patients should be placed in the recumbent position and treated supportively.
Patients should be cautioned about these possible adverse effects and advised to administer CARDAMYST in a sitting position, and in a location where the risk of fall is minimal.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risk of syncope [see Warnings and Precautions (5.1 )]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of CARDAMYST was evaluated using pooled data from double-blind, randomized, placebo-controlled trials including NODE-1, NODE-301 Part 1, RAPID, and RAPID Extension. A total of 321 patients were treated with CARDAMYST in randomized controlled studies.
In the RAPID and RAPID Extension studies, in which patients had the option of self-administering a second dose of CARDAMYST for a perceived episode of PSVT, the majority of patients (65%) self-administered a second dose of CARDAMYST (2x70mg).
In NODE-301 Part 1, RAPID, and RAPID Extension, to assess tolerability, a test dose(s) was given prior to randomization. A small percentage of patients failed the test dose due to hypotension (0.4%) [see Warnings and Precautions (5.1 )].
The majority of treatment-related adverse reactions reported in clinical studies with CARDAMYST have been related to local reactions to, at, or near the nasal administration site, including the nose, throat, and eyes. These local reactions included nasal discomfort, nasal congestion, throat irritation, oropharyngeal pain, lacrimation, rhinorrhea, bleeding from the nose, upper-airway cough syndrome, and sneezing.
1) Adverse reactions that occurred within 24 hours of study drug administration (TEAE24h) for perceived PSVT in the double-blind, placebo-controlled studies, NODE-1, NODE-301 Part 1, RAPID and RAPID Extension that had an overall incidence of 5% or greater and where the incidence is at least 1% greater than the placebo group. | |||
2) 2x70 mg: first administration of etripamil 70 mg followed by a second dose of etripamil 70 mg 10 minutes later if symptoms persisted. | |||
| Placebo N=223 % | CARDAMYST 70 mg N=235 % | CARDAMYST 2x70 mg 2 N=86 % | |
| Nasal Discomfort | 6 | 28 | 23 |
| Nasal Congestion | 1 | 14 | 12 |
| Rhinorrhea | 2 | 12 | 10 |
| Throat Irritation | 1 | 7 | 6 |
| Epistaxis | 1 | 6 | 7 |
DESCRIPTION
Etripamil, the active ingredient of CARDAMYST is a calcium channel blocker.
The chemical name of etripamil is benzoic acid, 3-[2-[[(4S)-4-cyano-4-(3,4-dimethoxyphenyl)-5-methylhexyl]methylamino]ethyl]-, methyl ester.
Its molecular weight is 452.59 and its molecular formula is C 27 H 36 N 2 O 4 .
The structural formula is:
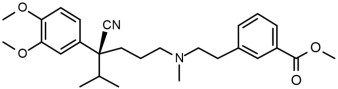
Etripamil is a colorless to slightly yellow oil. Etripamil has a pKa of 8.57 and is very soluble in methyl tert-butyl ether, freely soluble in methanol, dichloromethane and acetone, sparingly soluble in ethanol and hexane, and insoluble in water.
CARDAMYST is a spray intended for nasal administration. Each device of CARDAMYST delivers two metered sprays of etripamil with a total of 70 mg. CARDAMYST contains 350 mg/mL of etripamil and the following inactive ingredients: acetic acid, edetate disodium, sulfuric acid for pH adjustment, and water for injection.
CLINICAL PHARMACOLOGY
Mechanism of Action
Etripamil is an L-type calcium influx inhibitor (slow channel blocker or calcium ion antagonist). Etripamil exerts its pharmacologic effect by modulating the influx of ionic calcium across the cell membrane of the AV nodal cells as well as arterial smooth muscles and contractile myocardial cells. By interrupting reentry at the AV node, etripamil can restore sinus rhythm in patients with PSVT.
Pharmacodynamics
Cardiac Electrophysiology
Etripamil prolonged the baseline PR interval by 8% to 10% approximately 5 minutes following an intranasal dose of 70 mg. This PR interval prolongation begins to decline shortly after the etripamil maximum concentration (C max ) and falls below the 8 to 10% threshold between 25 and 50 minutes. Etripamil mean maximum change from baseline PR interval was approximately 13% following a single 70 mg dose and 20% following two 70 mg doses in the NODE-103 trial. At a dose of 1 to 2 times the maximum recommended therapeutic dose, etripamil does not prolong the QTc interval.
Hemodynamics
In patients with induced supraventricular tachycardia in an electrophysiology laboratory, a single intranasal etripamil administration was associated with a maximal systolic blood pressure reduction of approximately 21 mm Hg with 140 mg (2 x maximum approved dose), 17 mm Hg with 105 mg (1.5 x maximum approved dose), and 3 mm Hg with 70 mg dose. No reduction in blood pressure was observed with etripamil 35 mg dose.
Pharmacokinetics
After one 70 mg dose of etripamil, mean (%CV) area under the concentration-time curve (AUC) is approximately 5461 (51.6%) ng•min/mL and the C max is approximately 99 (64.6%) ng/mL. After a second 70 mg dose of etripamil administered 10 minutes after the first dose, mean (%CV) AUC is approximately 7721 (50.3%) ng•min/mL and the C max is approximately 132 (59.1%) ng/mL.
Absorption
Etripamil median (range) time to C max (T max ) is 7 minutes (3 to 20 minutes) following a single intranasal administration of 70 mg. Median T max is 13 minutes (3 to 35 minutes) following a second intranasal administration of 70 mg.
Distribution
Etripamil mean apparent volume of distribution ranges from approximately 2200 to 3500 L. Etripamil plasma protein binding is approximately 50%.
Elimination
Average etripamil concentration fell by approximately 60% of its peak value (C max ) at 25 minutes and 80% of the C max by 60 minutes after dosing. Subsequently, concentrations decrease at a slower rate, and this decline is associated with a half-life of approximately 2.5 hours.
Metabolism: Etripamil metabolic pathways include hydrolysis, demethylation, N-dealkylation, and secondary oxidation, glucuronidation, and taurine conjugation. Etripamil is primarily metabolized by blood esterases and hepatic metabolism, primarily via CYP3A4, and CYP3A5. Etripamil contains a methyl ester which renders it metabolically sensitive to bloodborne esterases.
Excretion: After a single dose of radiolabeled intranasal etripamil 70 mg to healthy subjects, approximately 29% of the dose was recovered in urine (<0.05% unchanged), 26% was recovered in feces (<0.05% unchanged), and the remainder was recovered on nose and face tissues. Approximately 71% of the total administered dose was recovered in 7-10 days.
Specific Populations
No clinically significant differences in the pharmacokinetics of etripamil were observed based on age (19 to 56 years old), body weight (49 to 91 kg), height (157 to 194 cm), sex, or race (Caucasian, Asian, or African American). The effect of renal impairment (eGFR < 90 mL/min) or hepatic impairment (Child Pugh A, B, or C) on etripamil pharmacokinetics is unknown. It is unknown whether etripamil is dialyzable.
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Negative chronotropic and inotropic agents: CARDAMYST was safely administered to patients taking beta blockers or calcium channel blockers. In RAPID, 107 (42%) patients were on beta blockers and 81 (32%) patients were on calcium channel blockers.
In Vitro Studies
Cytochrome P450 (CYP) Enzymes : Etripamil is a CYP3A4 and CYP3A5 substrate. Etripamil inhibits CYP2D6, CYP3A4, and CYP2C9 but does not inhibit CYP2B6 or CYP2C8. Etripamil does not induce CYP1A2, CYP2B6, or CYP3A4.
Transporter Systems : Etripamil is a substrate of P-gp and OATP1B1, but not BCRP, OATP1B3, OCT1, OCT2, OAT1, OAT3, MATE1, and MATE2-K. Etripamil inhibits P-gp and MATE1, but not BCRP, BSEP, MATE2-K, OAT1, OAT3, OATP1B1, OATP1B3, OCT1, and OCT2.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long term carcinogenicity studies in rodents have not been conducted with etripamil.
Mutagenesis
Etripamil was not genotoxic in a bacterial reverse mutation assay, an in vitro chromosome aberration assay in human peripheral lymphocytes, and an in vivo micronucleus study in rats (IV administration).
Impairment of Fertility
In a male and female fertility study, rats were dosed once daily via intravenous administration at etripamil doses of 0.05 to 0.374 mg/kg/day. Male rats were dosed for 28 days prior to mating, and female rats were dosed for 14 days prior to mating and continuing up to day 7 of pregnancy. There was no effect on fertility of male and female rats at doses up to 0.374 mg/kg/day, approximately 3× the C max and 0.4× the AUC at the MRHD.
CLINICAL STUDIES
The RAPID study (NCT #03464019) was a randomized, double-blind, placebo-controlled, multicenter, event-driven, phase 3 study designed to evaluate the efficacy and safety of CARDAMYST in patients with a history of symptomatic PSVT. Six hundred ninety-two (692) patients were randomized 1:1 to CARDAMYST 70 mg or placebo. Patients with an episode of perceived PSVT were to self-administer the study drug intranasally in a medically unsupervised setting and self-administer a second dose of the study drug if symptoms persisted at 10 minutes after the first dose. Continuously obtained electrocardiographic data during the episode of PSVT were blindly adjudicated. The primary endpoint was time-to-conversion of confirmed PSVT to sinus rhythm for at least 30 seconds within 30 minutes of the first dose.
Of the 692 randomized patients, 255 patients perceived an episode of PSVT and self-administered the study drug; 184 (72%) episodes were confirmed by blinded adjudication to be PSVT. Patients had a median age of 54 years (range 19 to 78 years) and were 71% female, 93% Caucasian, 3% Black and 4% other. Sixty-three percent (63%) of patients with confirmed PSVT episode were taking concomitant beta blocker or calcium channel blocker.
In the study's primary analysis, patients with confirmed episodes of PSVT, Kaplan-Meier Estimates of those who converted to sinus rhythm within 30 minutes were 64% and 31% for CARDAMYST and placebo, respectively, with a hazard ratio of 2.6 [95% CI: (1.7, 4.2)], p-value <0.001. Median time-to-conversion was 17.2 minutes (95% CI: 13.4, 26.5) with CARDAMYST versus 53.5 minutes (95% CI: 38.7, 87.3) with placebo. Kaplan-Meier estimates of conversion remained in favor of CARDAMYST at 300 minutes; hazard ratio 1.7 [95% CI: (1.2, 2.4)].
Kaplan-Meier plot of estimated probabilities of achieving sinus rhythm following treatment with CARDAMYST are shown in Figure 1 .
Figure 1: RAPID Primary Efficacy Outcome Kaplan Meier Curve: Conversion to Sinus Rhythm in the Patient Population with Confirmed PSVT 1
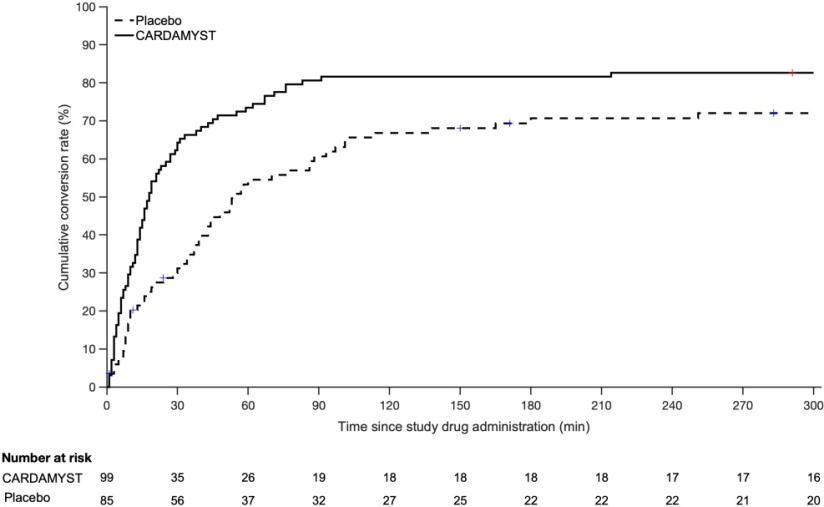
1) 71/255 (28%) patients who self-administered CARDAMYST for a perceived episode of PSVT did not have confirmed PSVT and are excluded.
Seventy-one (71) of 255 (28%) patients who self-administered CARDAMYST for a perceived episode of PSVT did not have PSVT confirmed due to missing ECG data (4%), resolution of PSVT prior to dosing (5%), or other rhythm diagnoses (19%). In an analysis assuming all 71 of these patients did not convert, the Kaplan-Meier estimates for conversion to sinus rhythm within 30 minutes were 50% and 23% for the CARDAMYST and placebo groups, respectively, with a hazard ratio of 2.6 [95% CI: (1.6, 4.1)].
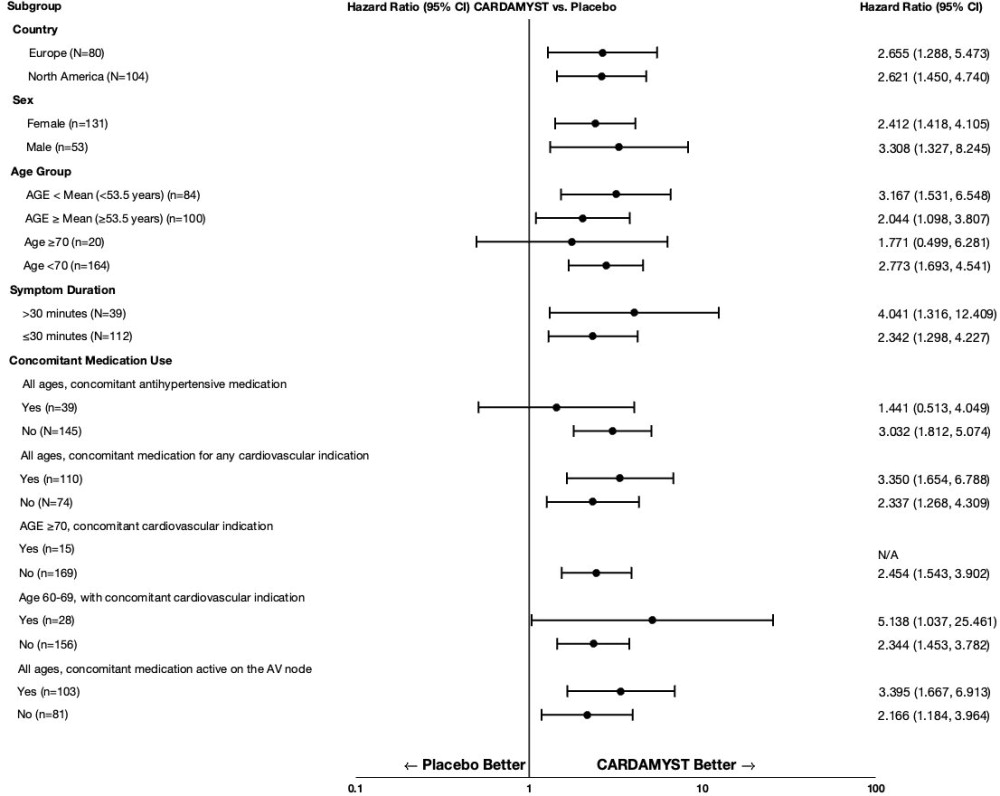
In RAPID, the results for the primary efficacy endpoint were generally consistent across major subgroups including geographic region, sex, age group, concomitant medications, and duration from onset of symptoms to first dose (Figure 2 ).
Figure 2: Conversion to Sinus Rhythm at 30 Minutes Hazard Ratios by Baseline Characteristics - RAPID Study
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
CARDAMYST is supplied in cartons (NDC 83468-070-03) of 2 disposable nasal spray devices contained in a plastic carrying case.
Each nasal spray device delivers two sprays containing a total of 70 mg etripamil.
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F), with excursions permitted from 15°C to 30°C (59°F to 86°F) (see USP Controlled Room Temperature).
Do not test spray, prime or press the plunger before use. Discard the CARDAMYST device after use.
INSTRUCTIONS FOR USE Cardamyst™ [kar da mist] (etripamil) nasal spray |
This Instructions for Use contains information on how to use CARDAMYST nasal spray .
Important Information You Need to Know Before Using CARDAMYST
| CARDAMYST Device 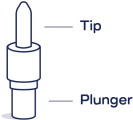 |
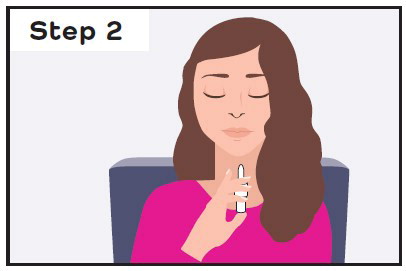
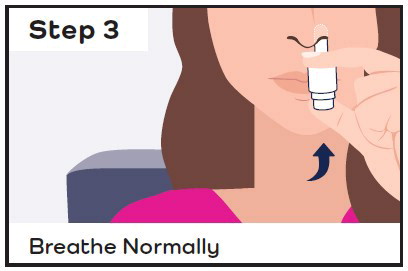
| Preparing the Dose | Using CARDAMYST Nasal Spray | |
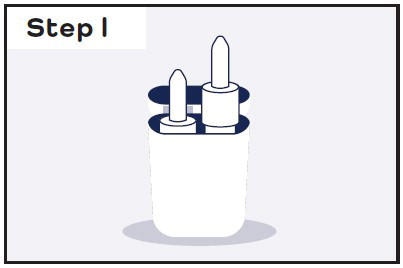 |  |  |
Remove the CARDAMYST device from the carrying case:
| Sit down and hold the nasal spray device in your hand:
| Insert the nasal spray device into the first nostril and spray:
|
| Using CARDAMYST Nasal Spray (continued) | ||
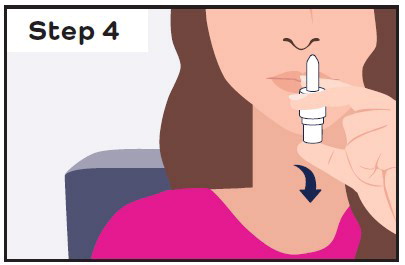 |  |  |
Release the plunger:
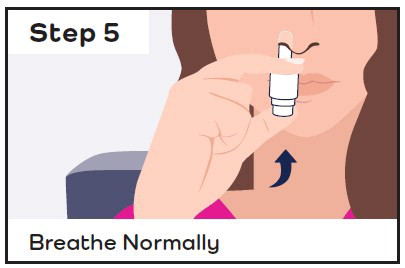
| Insert the same nasal spray device into the second nostril and spray right away:
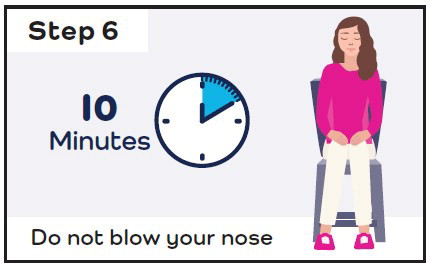
| Stay seated and keep your head straight for 10 minutes:
|
 | If you continue to have symptoms after 10 minutes:
| |
Storing CARDAMYST
Store CARDAMYST at room temperature between 20°C to 25°C (68°F to 77°F). | Keep CARDAMYST and all medicines out of the reach of children. |
For more information about CARDAMYST, visit cardamyst.com or call 1-844-805-5810 .
Manufactured for: Milestone Pharmaceuticals USA, Inc., 6210 Ardrey Kell Road, Suite 650, Charlotte, NC 28277
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Issued: December 2025
Mechanism of Action
Etripamil is an L-type calcium influx inhibitor (slow channel blocker or calcium ion antagonist). Etripamil exerts its pharmacologic effect by modulating the influx of ionic calcium across the cell membrane of the AV nodal cells as well as arterial smooth muscles and contractile myocardial cells. By interrupting reentry at the AV node, etripamil can restore sinus rhythm in patients with PSVT.