Get your patient on Cetrotide - Cetrorelix Acetate (Cetrorelix Acetate)
Cetrotide - Cetrorelix Acetate prescribing information
INDICATIONS AND USAGE
Cetrotide ® (cetrorelix acetate for injection) is indicated for the inhibition of premature LH surges in women undergoing controlled ovarian stimulation.
DOSAGE AND ADMINISTRATION
Ovarian stimulation therapy with gonadotropins (FSH, hMG) is started on cycle Day 2 or 3. The dose of gonadotropins should be adjusted according to individual response. Cetrotide ® (cetrorelix acetate for injection) 0.25 mg may be administered subcutaneously once daily during the early- to mid-follicular phase.
Cetrotide ® 0.25 mg is administered on either stimulation day 5 (morning or evening) or day 6 (morning) and continued daily until the day of hCG administration.
When assessment by ultrasound shows a sufficient number of follicles of adequate size, hCG is administered to induce ovulation and final maturation of the oocytes. No hCG should be administered if the ovaries show an excessive response to the treatment with gonadotropins to reduce the chance of developing ovarian hyperstimulation syndrome (OHSS).
Administration
Cetrotide ® 0.25 mg can be administered by the patient herself after appropriate instructions by her doctor.
Directions for using Cetrotide ® 0.25 mg with the enclosed needles and pre-filled syringe:
- Wash hands thoroughly with soap and water.
- Flip off the plastic cover of the vial and wipe the aluminum ring and the rubber stopper with an alcohol swab.
- Twist the injection needle with the yellow mark (20 gauge) on the pre-filled syringe.
- Push the needle through the center of the rubber stopper of the vial and slowly inject the solvent into the vial.
- Leaving the syringe in the vial, gently swirl the vial until the solution is clear and without residues. Avoid forming bubbles.
- Draw the total contents of the vial into the syringe. If necessary, invert the vial and pull back the needle as far as needed to withdraw the entire contents of the vial.
- Replace the needle with the yellow mark by the injection needle with the grey mark (27 gauge).
- Invert the syringe and push the plunger until all air bubbles have been expelled.
- Choose an injection site in the lower abdominal area, preferably around, but staying at least one inch away from the navel. Choose a different injection site each day to minimize local irritation. Use a second alcohol swab to clean the skin at the injection site and allow alcohol to dry. Gently pinch up the skin surrounding the site of injection.
- Inject the prescribed dose as directed by your doctor, nurse or pharmacist.
- Use the syringe and needles only once. Dispose of the syringe and needles properly after use. If available, use a medical waste container for disposal.
CONTRAINDICATIONS
Cetrotide ® (cetrorelix acetate for injection) is contraindicated under the following conditions:
- Hypersensitivity to cetrorelix acetate, extrinsic peptide hormones or mannitol.
- Known hypersensitivity to GnRH or any other GnRH analogs.
- Known or suspected pregnancy, and lactation (see PRECAUTIONS ).
- Severe renal impairment
ADVERSE REACTIONS
The safety of Cetrotide ® (cetrorelix acetate for injection) in 949 patients undergoing controlled ovarian stimulation in clinical studies was evaluated. Women were between 19 and 40 years of age (mean: 32). 94.0% of them were Caucasian. Cetrotide ® was given in doses ranging from 0.1 mg to 5 mg as either a single or multiple dose.
Table 3 shows systemic adverse events, reported in clinical studies without regard to causality, from the beginning of Cetrotide ® treatment until confirmation of pregnancy by ultrasound at an incidence ≥ 1% in Cetrotide ® treated subjects undergoing COS.
| (WHO preferred term) | Cetrotide ® N=949 % (n) |
|---|---|
| Ovarian Hyperstimulation Syndrome Intensity moderate or severe, or WHO Grade II or III, respectively | 3.5 (33) |
| Nausea | 1.3 (12) |
| Headache | 1.1 (10) |
Local site reactions (e.g. redness, erythema, bruising, itching, swelling, and pruritus) were reported. Usually, they were of a transient nature, mild intensity and short duration. During post-marketing surveillance, cases of mild to moderate Ovarian Hyperstimulation syndrome and cases of hypersensitivity reactions including anaphylactoid reactions have been reported.
Two stillbirths were reported in Phase 3 studies of Cetrotide ® .
Congenital Anomalies
Clinical follow-up studies of 316 newborns of women administered Cetrotide ® were reviewed. One infant of a set of twin neonates was found to have anencephaly at birth and died after four days. The other twin was normal. Developmental findings from ongoing baby follow-up included a child with a ventricular septal defect and another child with bilateral congenital glaucoma.
Four pregnancies that resulted in therapeutic abortion in Phase 2 and Phase 3 controlled ovarian stimulation studies had major anomalies (diaphragmatic hernia, trisomy 21, Klinefelter syndrome, polymalformation, and trisomy 18). In three of these four cases, intracytoplasmic sperm injection (ICSI) was the fertilization method employed; in the fourth case, in vitro fertilization (IVF) was the method employed.
The minor congenital anomalies reported include: supernumerary nipple, bilateral strabismus, imperforate hymen, congenital nevi, hemangiomata, and QT syndrome.
The causal relationship between the reported anomalies and Cetrotide ® is unknown. Multiple factors, genetic and others (including, but not limited to ICSI, IVF, gonadotropins, and progesterone) make causal attribution difficult to study.
Drug Interactions
No formal drug interaction studies have been performed with Cetrotide ® .
DESCRIPTION
Cetrotide ® (cetrorelix acetate for injection) is a synthetic decapeptide with gonadotropin-releasing hormone (GnRH) antagonistic activity. Cetrorelix acetate is an analog of native GnRH with substitutions of amino acids at positions 1, 2, 3, 6, and 10. The molecular formula is Acetyl-D-3-(2´-naphthyl)-alanine-D-4-chlorophenylalanine-D-3-(3´-pyridyl)-alanine-L-serine-L-tyrosine-D-citrulline-L-leucine-L-arginine-L-proline-D-alanine-amide, and the molecular weight is 1431.06, calculated as the anhydrous free base. The structural formula is as follows:
Cetrorelix acetate
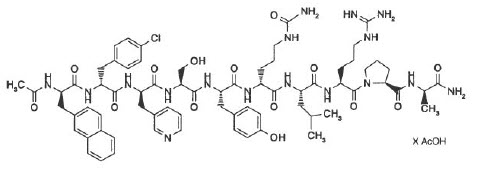 |
| (Ac-D-Nal 1 - D -Cpa 2 - D -Pal 3 -Ser 4 -Tyr 5 - D -Cit 6 -Leu 7 -Arg 8 -Pro 9 - D -Ala 10 -NH 2 ) |
Cetrotide ® (cetrorelix acetate for injection) 0.25 mg is a sterile lyophilized powder intended for subcutaneous injection after reconstitution with Sterile Water for Injection, that comes supplied in a 1.0 mL pre-filled syringe. Each vial of Cetrotide ® 0.25 mg contains 0.26-0.27 mg cetrorelix acetate, equivalent to 0.25 mg cetrorelix, and 54.80 mg mannitol.
CLINICAL PHARMACOLOGY
GnRH induces the production and release of luteinizing hormone (LH) and follicle stimulating hormone (FSH) from the gonadotrophic cells of the anterior pituitary. Due to a positive estradiol (E2) feedback at midcycle, GnRH liberation is enhanced resulting in an LH-surge. This LH-surge induces the ovulation of the dominant follicle, resumption of oocyte meiosis and subsequently luteinization as indicated by rising progesterone levels.
Cetrotide ® competes with natural GnRH for binding to membrane receptors on pituitary cells and thus controls the release of LH and FSH in a dose-dependent manner. The onset of LH suppression is approximately one hour with the 3 mg dose and two hours with the 0.25 mg dose. This suppression is maintained by continuous treatment and there is a more pronounced effect on LH than on FSH. An initial release of endogenous gonadotropins has not been detected with Cetrotide ® , which is consistent with an antagonist effect.
The effects of Cetrotide ® on LH and FSH are reversible after discontinuation of treatment. In women, Cetrotide ® delays the LH-surge, and consequently ovulation, in a dose-dependent fashion. FSH levels are not affected at the doses used during controlled ovarian stimulation. Following a single 3 mg dose of Cetrotide ® , duration of action of at least 4 days has been established. A dose of Cetrotide ® 0.25 mg every 24 hours has been shown to maintain the effect.
Pharmacokinetics
The pharmacokinetic parameters of single and multiple doses of Cetrotide ® (cetrorelix acetate for injection) in adult healthy female subjects are summarized in Table 1.
| Single dose 3 mg | Single dose 0.25 mg | Multiple dose 0.25 mg | |
|---|---|---|---|
| t max Time to reach observed maximum plasma concentration t 1/2 Elimination half-life C max Maximum plasma concentration; multiple dose C ss, max AUC Area under the curve; single dose AUC 0-inf , multiple dose AUCt CL Total plasma clearance Vz Volume of distribution Geometric mean (95% CI ln ), | |||
| No. of subjects | 12 | 12 | 12 |
| t max median (min-max) [h] | 1.5 (0.5-2) | 1.0 (0.5-1.5) | 1.0 (0.5-2) |
| t 1/2 [h] | 62.8 (38.2-108) | 5.0 (2.4-48.8) | 20.6 (4.1-179.3) |
| C max [ng/ml] | 28.5 (22.5-36.2) | 4.97 (4.17-5.92) | 6.42 (5.18-7.96) |
| AUC [ng∙h/ml] | 536 (451-636) | 31.4 (23.4-42.0) | 44.5 (36.7-54.2) |
| CL arithmetic mean, [ml/min∙kg] | 1.28 Based on iv administration (n=6, separate study 0013) | ||
| Vz [l/kg] | 1.16 | ||
Absorption
Cetrotide ® is rapidly absorbed following subcutaneous injection, maximal plasma concentrations being achieved approximately one to two hours after administration. The mean absolute bioavailability of Cetrotide ® following subcutaneous administration to healthy female subjects is 85%.
Distribution
The volume of distribution of Cetrotide ® following a single intravenous dose of 3 mg is about 1 l/kg. In vitro protein binding to human plasma is 86%.
Cetrotide ® concentrations in follicular fluid and plasma were similar on the day of oocyte pick-up in patients undergoing controlled ovarian stimulation. Following subcutaneous administration of Cetrotide ® 0.25 mg and 3 mg, plasma concentrations of cetrorelix were below or in the range of the lower limit of quantitation on the day of oocyte pick-up and embryo transfer.
Metabolism
After subcutaneous administration of 10 mg Cetrotide ® to females and males, Cetrotide ® and small amounts of (1-9), (1-7), (1-6), and (1-4) peptides were found in bile samples over 24 hours.
In in vitro studies, Cetrotide ® was stable against phase I- and phase II-metabolism. Cetrotide ® was transformed by peptidases, and the (1-4) peptide was the predominant metabolite.
Excretion
Following subcutaneous administration of 10 mg cetrorelix to males and females, only unchanged cetrorelix was detected in urine. In 24 hours, cetrorelix and small amounts of the (1-9), (1-7), (1-6), and (1-4) peptides were found in bile samples. 2-4% of the dose was eliminated in the urine as unchanged cetrorelix, while 5-10% was eliminated as cetrorelix and the four metabolites in bile. Therefore, only 7-14% of the total dose was recovered as unchanged cetrorelix and metabolites in urine and bile up to 24 hours. The remaining portion of the dose may not have been recovered since bile and urine were not collected for a longer period of time.
Special Populations
Pharmacokinetic investigations have not been performed either in subjects with impaired renal or liver function, or in the elderly, or in children (see PRECAUTIONS ).
Pharmacokinetic differences in different races have not been determined.
There is no evidence of differences in pharmacokinetic parameters for Cetrotide ® between healthy subjects and patients undergoing controlled ovarian stimulation.
Drug-Drug Interactions
No formal drug-drug interaction studies have been performed with Cetrotide ® (see PRECAUTIONS ).
Clinical Studies
Seven hundred thirty two (732) patients were treated with Cetrotide ® (cetrorelix acetate for injection) in five (two Phase 2 dose-finding and three Phase 3) clinical trials. The clinical trial population consisted of Caucasians (95.5%) and Black, Asian, Arabian and others (4.5%). Women were between 19 and 40 years of age (mean: 32). The studies excluded subjects with polycystic ovary syndrome (PCOS), subjects with low or no ovarian reserve, and subjects with stage III-IV endometriosis.
Two dose regimens were investigated in these clinical trials, either a single dose per treatment cycle or multiple dosing. In the Phase 2 studies, a single dose of 3 mg was established as the minimal effective dose for the inhibition of premature LH surges with a protection period of at least 4 days. When Cetrotide ® is administered in a multidose regimen, 0.25 mg was established as the minimal effective dose. The extent and duration of LH-suppression is dose dependent.
In the Phase 3 program, efficacy of the single 3 mg dose regimen of Cetrotide ® and the multiple 0.25 mg dose regimen of Cetrotide ® was established separately in two adequate and well controlled clinical studies utilizing active comparators. A third non-comparative clinical study evaluated only the multiple 0.25 mg dose regimen of Cetrotide ®. The ovarian stimulation treatment with recombinant FSH or human menopausal gonadotropin (hMG) was initiated on day 2 or 3 of a normal menstrual cycle. The dose of gonadotropins was administered according to the individual patient's disposition and response.
In the single dose regimen study, Cetrotide ® 3 mg was administered on the day of controlled ovarian stimulation when adequate estradiol levels (400 pg/mL) were obtained, usually on day 7 (range day 5-12). If hCG was not given within 4 days of the 3 mg dose of Cetrotide ® , then 0.25 mg of Cetrotide ® was administered daily beginning 96 hours after the 3 mg injection until and including the day of hCG administration.
In the two multiple dose regimen studies, Cetrotide ® 0.25 mg was started on day 5 or 6 of COS. Both gonadotropins and Cetrotide ® were continued daily (multiple dose regimen) until the injection of human chorionic gonadotropin (hCG).
Oocyte pick-up (OPU) followed by in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI) as well as embryo transfer (ET) were subsequently performed. The results for Cetrotide ® are summarized below in Table 2.
| Parameter | Cetrotide ® 3 mg (sd, active comparator study) | Cetrotide ® 0.25 mg (md, active comparator study) | Cetrotide ® 0.25 mg (md, non-comparative study) |
|---|---|---|---|
| No. of subjects | 115 | 159 | 303 |
| hCG administered [%] | 98.3 | 96.2 | 96.0 |
| Oocyte pick-up [%] | 98.3 | 94.3 | 93.1 |
| LH-surge [%] (LH ≥ 10 U/L and P Progesterone ≥ 1 ng/mL) Following initiation of Cetrotide ® therapy | 0.0 | 1.9 | 1.0 |
| Serum E 2 [pg/ml] at day hCG Morning values , Median with 5th – 95th percentiles | 1125 (470-2952) | 1064 (341-2531) | 1185 (311-3676) |
| Serum LH [U/L] at day hCG , | 1.0 (0.5-2.5) | 1.5 (0.5-7.6) | 1.1 (0.5-3.5) |
| No. of follicles ≥ 11 mm at day hCG Mean ± standard deviation | 11.2±5.5 | 10.8±5.2 | 10.4±4.5 |
| No. of oocytes: IVF ICSI | 9.2±5.2 10.0±4.2 | 7.6±4.3 10.1±5.6 | 8.5±5.1 9.3±5.9 |
| Fertilization rate: IVF ICSI | 0.48±0.33 0.66±0.29 | 0.62±0.26 0.63±0.29 | 0.60±0.26 0.61±0.25 |
| No. of embryos transferred | 2.6±0.9 | 2.1±0.6 | 2.7±1.0 |
| Clinical pregnancy rate [%] | |||
| per attempt | 22.6 | 20.8 | 19.8 |
| per subject with ET | 26.3 | 24.1 | 23.3 |
In addition to IVF and ICSI, one pregnancy was obtained after intrauterine insemination. In the five Phase 2 and Phase 3 clinical trials, 184 pregnancies have been reported out of a total of 732 patients (including 21 pregnancies following the replacement of frozen-thawed embryos).
In the 3 mg regimen, 9 patients received an additional dose of 0.25 mg of Cetrotide ® and two other patients received two additional doses of 0.25 mg Cetrotide ® . The median number of days of Cetrotide ® multiple dose treatment was 5 (range 1-15) in both studies.
No drug related allergic reactions were reported from these clinical studies.
HOW SUPPLIED
Cetrotide ® (cetrorelix acetate for injection) 0.25 mg is available in a carton of one packaged tray (NDC 44087-1225-1).
Each packaged tray contains: one glass vial containing 0.26 - 0.27 mg cetrorelix acetate (corresponding to 0.25 mg cetrorelix), one pre-filled glass syringe with 1 mL of Sterile Water for Injection, one 20 gauge needle (yellow) and one 27 gauge needle (grey).
Storage
Store Cetrotide ® 0.25 mg refrigerated, 2ºC - 8ºC (36ºF - 46ºF). Store the packaged tray in the outer carton in order to protect from light.