Get your patient on Chlorhexidine Gluconate - Chlorhexidine Gluconate rinse (Chlorhexidine Gluconate)
Chlorhexidine Gluconate - Chlorhexidine Gluconate rinse prescribing information
INDICATION
Chlorhexidine gluconate oral rinse, 0.12% is indicated for use between dental visits as part of a professional program for the treatment of gingivitis as characterized by redness and swelling of the gingivae, including gingival bleeding upon probing. Chlorhexidine gluconate oral rinse, 0.12% has not been tested among patients with acute necrotizing ulcerative gingivitis (ANUG). For patients having coexisting gingivitis and periodontitis, see PRECAUTIONS .
DOSAGE AND ADMINISTRATION
Chlorhexidine gluconate oral rinse, 0.12% therapy should be initiated directly following a dental prophylaxis. Patients using chlorhexidine gluconate oral rinse, 0.12% should be reevaluated and given a thorough prophylaxis at intervals no longer than six months. Recommended use is twice daily oral rinsing for 30 seconds, morning and evening after toothbrushing. Usual dosage is 15 mL of undiluted chlorhexidine gluconate oral rinse, 0.12%. Patients should be instructed to not rinse with water or other mouthwashes, brush teeth or eat immediately after using chlorhexidine gluconate oral rinse, 0.12%. Chlorhexidine gluconate oral rinse, 0.12% is not intended for ingestion and should be expectorated after rinsing.
CONTRAINDICATIONS
Chlorhexidine gluconate oral rinse, 0.12% should not be used by persons who are known to be hypersensitive to chlorhexidine gluconate or other formula ingredients.
ADVERSE REACTIONS
The most common side effects associated with chlorhexidine gluconate oral rinses are: 1) an increase in staining of teeth and other oral surfaces; 2) an increase in calculus formation; and 3) an alteration in taste perception, see WARNINGS and PRECAUTIONS . Oral irritation and local allergy-type symptoms have been spontaneously reported as side effects associated with the use of chlorhexidine gluconate oral rinse. The following oral mucosal side effects were reported during placebo-controlled adult clinical trials: aphthous ulcer, grossly obvious gingivitis, trauma, ulceration, erythema, desquamation, coated tongue, keratinization, geographic tongue, mucocele, and short frenum. Each occurred at a frequency of less than 1%. Among postmarketing reports, the most frequently reported oral mucosal symptoms associated with chlorhexidine gluconate oral rinse are stomatitis, gingivitis, glossitis, ulcer, dry mouth, hypesthesia, glossal edema, and paresthesia. Minor irritation and superficial desquamation of the oral mucosa have been noted in patients using chlorhexidine gluconate oral rinse. There have been cases of parotid gland swelling and inflammation of the salivary glands (sialadenitis) reported in patients using chlorhexidine gluconate oral rinse.
To report SUSPECTED ADVERSE REACTIONS, contact Pharmaceutical Associates, Inc. at 1-800-845-8210 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Chlorhexidine gluconate oral rinse USP, 0.12% is an oral rinse containing 0.12% chlorhexidine gluconate (1,1’-hexamethylene bis [5-(p-chlorophenyl) biguanide]di-D-gluconate) in a base containing 11.6% alcohol, FD&C Blue No. 1, glycerin, peppermint flavor, polysorbate 80, purified water, and saccharin sodium. Chlorhexidine Gluconate Oral Rinse, 0.12% is a near-neutral solution (pH range 5-7). Chlorhexidine gluconate is a salt of chlorhexidine and gluconic acid. Its chemical structure is:
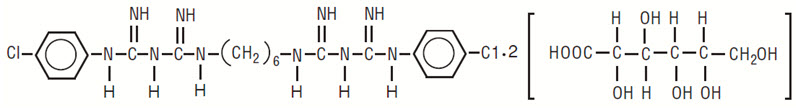
C 22 H 30 Cl 2 N 10 •2C 6 H 12 O 7 M.W. 897.72
CLINICAL PHARMACOLOGY
Chlorhexidine gluconate oral rinse, 0.12% provides antimicrobial activity during oral rinsing. The clinical significance of chlorhexidine gluconate oral rinse, 0.12% antimicrobial activities is not clear. Microbiological sampling of plaque has shown a general reduction of counts of certain assayed bacteria, both aerobic and anaerobic, ranging from 54-97% through six months use.
Use of chlorhexidine gluconate oral rinse in a six month clinical study did not result in any significant changes in bacteria resistance, overgrowth of potentially opportunistic organisms or other adverse changes in the oral microbial ecosystem. Three months after chlorhexidine gluconate oral rinse use was discontinued, the number of bacteria in plaque had returned to baseline levels and resistance of plaque bacteria to chlorhexidine gluconate was equal to that at baseline.
Pharmacokinetics
Pharmacokinetic studies with chlorhexidine gluconate oral rinse indicate approximately 30% of the active ingredient, chlorhexidine gluconate, is retained in the oral cavity following rinsing. This retained drug is slowly released into the oral fluids. Studies conducted on human subjects and animals demonstrate chlorhexidine gluconate is poorly absorbed from the gastrointestinal tract. The mean plasma level of chlorhexidine gluconate reached a peak of 0.206 µg/g in humans 30 minutes after they ingested a 300 mg dose of the drug. Detectable levels of chlorhexidine gluconate were not present in the plasma of these subjects 12 hours after the compound was administered. Excretion of chlorhexidine gluconate occurred primarily through the feces (~90%). Less than 1% of the chlorhexidine gluconate ingested by these subjects was excreted in the urine.
HOW SUPPLIED
Chlorhexidine gluconate oral rinse USP, 0.12% is a clear, light blue, peppermint flavored liquid and is supplied as:
NDC 0121-0893-16: 16 fl oz (473 mL)
NDC 0121-0893-15: 15 mL unit dose cup. Case contains 40 unit dose cups of 15 mL (NDC 0121-0893-40), packaged in 4 trays of 10 unit dose cups each and 100 unit dose cups of 15 mL (NDC 0121-0893-00) packaged in 10 trays of 10 unit dose cups each.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Rx only
Keep out of reach of children
Dispense in bottle as provided with a child-resistant closure.