Get your patient on Chlorzoxazone - Chlorzoxazone tablet (Chlorzoxazone)
Chlorzoxazone - Chlorzoxazone tablet prescribing information
INDICATIONS AND USAGE
Chlorzoxazone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions. The mode of action of this drug has not been clearly identified, but may be related to its sedative properties. Chlorzoxazone does not directly relax tense skeletal muscles in man.
DOSAGE AND ADMINISTRATION
Usual Adult Dosage: One tablet (250 mg) three or four times daily. Initial dosage for painful musculoskeletal conditions should be two tablets (500 mg) three or four times daily. If adequate response is not obtained with this dose, it may be increased to three tablets (750 mg) three or four times daily. As improvement occurs dosage can usually be reduced.
CONTRAINDICATIONS
Chlorzoxazone is contraindicated in patients with known intolerance to the drug.
ADVERSE REACTIONS
After extensive clinical use of chlorzoxazone-containing products, it is apparent that the product is well tolerated and seldom produces undesirable side effects. Occasional patients may develop gastrointestinal disturbances. It is possible in rare instances that chlorzoxazone may have been associated with gastrointestinal bleeding. Drowsiness, dizziness, lightheadedness, malaise, or overstimulation may be noted by an occasional patient. Rarely, allergic-type skin rashes, petechiae, or ecchymoses may develop during treatment. Angioneurotic edema or anaphylactic reactions are extremely rare. There is no evidence that the drug will cause renal damage. Rarely, a patient may note discoloration of the urine resulting from a phenolic metabolite of chlorzoxazone. This finding is of no known clinical significance.
To report SUSPECTED ADVERSE REACTIONS, contact Camber Pharmaceuticals, Inc. at 1-866-495-1995 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch for voluntary reporting of adverse reactions.
DESCRIPTION
Each tablet contains:
Chlorzoxazone, USP•…………… 250 mg
•5-Chloro-2-benzoxazolinone
Structural Formula:
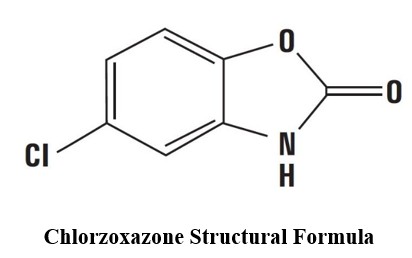
Molecular Formula: C 7 H 4 ClNO 2
Molecular Weight: 169.57
Chlorzoxazone, USP is a white to off-white crystalline powder. Chlorzoxazone is soluble in alkali hydroxides and sparingly soluble in methanol.
Inactive ingredients: anhydrous lactose, croscarmellose sodium, docusate sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, pregelatinized starch (maize) and sodium benzoate.
FDA approved dissolution test specifications differ from USP
CLINICAL PHARMACOLOGY
Chlorzoxazone is a centrally-acting agent for painful musculoskeletal conditions. Data available from animal experiments as well as human study indicate that chlorzoxazone acts primarily at the level of the spinal cord and subcortical areas of the brain where it inhibits multisynaptic reflex arcs involved in producing and maintaining skeletal muscle spasm of varied etiology. The clinical result is a reduction of the skeletal muscle spasm with relief of pain and increased mobility of the involved muscles. Blood levels of chlorzoxazone can be detected in people during the first 30 minutes and peak levels may be reached, in the majority of the subjects, in about 1 to 2 hours after oral administration of chlorzoxazone. Chlorzoxazone is rapidly metabolized and is excreted in the urine, primarily in a conjugated form as the glucuronide. Less than one percent of a dose of chlorzoxazone is excreted unchanged in the urine in 24 hours.
HOW SUPPLIED
Chlorzoxazone tablets, USP, 250 mg are supplied as white to off-white, capsule shaped, uncoated tablets debossed with “C15” on one side and plain on other side.
Bottles of 30 Tablets NDC 31722-974-30
Bottles of 60 Tablets NDC 31722-974-60
Bottles of 100 Tablets NDC 31722-974-01
Bottles of 500 Tablets NDC 31722-974-05
Bottles of 1000 Tablets NDC 31722-974-10
Dispense in tight container as defined in the USP.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Manufactured for:
 Camber Pharmaceuticals, Inc. Piscataway, NJ 08854.
Camber Pharmaceuticals, Inc. Piscataway, NJ 08854.
Manufactured by: CorePharma, LLC. 215 Wood Ave, Middlesex, NJ 08846
Revised: 02/2022 40033