Get your patient on Cladribine - Cladribine injection, Solution (Cladribine)
Cladribine - Cladribine injection, Solution prescribing information
WARNING
Cladribine injection should be administered under the supervision of a qualified physician experienced in the use of antineoplastic therapy. Suppression of bone marrow function should be anticipated. This is usually reversible and appears to be dose dependent. Serious neurological toxicity (including irreversible paraparesis and quadraparesis) has been reported in patients who received cladribine injection by continuous infusion at high doses (4 to 9 times the recommended dose for Hairy Cell Leukemia). Neurologic toxicity appears to demonstrate a dose relationship; however, severe neurological toxicity has been reported rarely following treatment with standard cladribine dosing regimens.
Acute nephrotoxicity has been observed with high doses of cladribine (4 to 9 times the recommended dose for Hairy Cell Leukemia), especially when given concomitantly with other nephrotoxic agents/therapies.
INDICATIONS FOR USE
Cladribine Injection, USP is indicated for the treatment of active Hairy Cell Leukemia as defined by clinically significant anemia, neutropenia, thrombocytopenia or disease-related symptoms.
DOSAGE AND ADMINISTRATION
Usual Dose
The recommended dose and schedule of cladribine injection, USP for active Hairy Cell Leukemia is as a single course given by continuous infusion for 7 consecutive days at a dose of 0.09 mg/kg/day. Deviations from this dosage regimen are not advised. If the patient does not respond to the initial course of cladribine injection, USP for Hairy Cell Leukemia, it is unlikely that they will benefit from additional courses. Physicians should consider delaying or discontinuing the drug if neurotoxicity or renal toxicity occurs (see WARNINGS ).
Specific risk factors predisposing to increased toxicity from cladribine have not been defined. In view of the known toxicities of agents of this class, it would be prudent to proceed carefully in patients with known or suspected renal insufficiency or severe bone marrow impairment of any etiology. Patients should be monitored closely for hematologic and non-hematologic toxicity (see WARNINGS and PRECAUTIONS ).
Preparation and Administration of Intravenous Solutions
Cladribine injection, USP must be diluted with the designated diluent prior to administration. Since the drug product does not contain any antimicrobial preservative or bacteriostatic agent, aseptic technique and proper environmental precautions must be observed in preparation of cladribine injection solutions.
To prepare a single daily dose
Cladribine injection, USP should be passed through a sterile 0.22µm disposable hydrophilic syringe filter prior to introduction into the infusion bag, prior to each daily infusion. Add the calculated dose (0.09 mg/kg or 0.09 mL/kg) of cladribine injection, USP through the sterile filter to an infusion bag containing 500 mL of 0.9% Sodium Chloride Injection, USP. Infuse continuously over 24 hours. Repeat daily for a total of 7 consecutive days. The use of 5% dextrose as a diluent is not recommended because of increased degradation of cladribine. Admixtures of cladribine injection, USP are chemically and physically stable for at least 24 hours at room temperature under normal room fluorescent light in Baxter Viaflex ® PVC infusion containers. Since limited compatibility data are available, adherence to the recommended diluents and infusion systems is advised.
| Dose of Cladribine Injection, USP | Recommended Diluent | Quantity of Diluent | |
|---|---|---|---|
| 24-hour infusion method | 1(day) × 0.09 mg/kg | 0.9% Sodium Chloride Injection, USP | 500 mL |
To prepare a 7-day infusion
The 7-day infusion solution should only be prepared with Bacteriostatic 0.9% Sodium Chloride Injection, USP (0.9% benzyl alcohol preserved). In order to minimize the risk of microbial contamination, both cladribine injection, USP and the diluent should be passed through a sterile 0.22 µm disposable hydrophilic syringe filter as each solution is being introduced into the infusion reservoir. First add the calculated dose of cladribine injection, USP (7 days × 0.09 mg/kg or mL/kg) to the infusion reservoir through the sterile filter.
Then add a calculated amount of Bacteriostatic 0.9% Sodium Chloride Injection, USP (0.9% benzyl alcohol preserved) also through the filter to bring the total volume of the solution to 100 mL. After completing solution preparation, clamp off the line, disconnect and discard the filter. Aseptically aspirate air bubbles from the reservoir as necessary using the syringe and a dry second sterile filter or a sterile vent filter assembly. Reclamp the line and discard the syringe and filter assembly. Infuse continuously over 7 days. Solutions prepared with Bacteriostatic Sodium Chloride Injection for individuals weighing more than 85 kg may have reduced preservative effectiveness due to greater dilution of the benzyl alcohol preservative. Admixtures for the 7-day infusion have demonstrated acceptable chemical and physical stability for at least 7 days in the SIMS Deltec MEDICATION CASSETTE™ Reservoir.
| Dose of Cladribine Injection, USP | Recommended Diluent | Quantity of Diluent | |
|---|---|---|---|
| 7-day infusion method (use sterile 0.22µ filter when preparing infusion solution) | 7 (days) × 0.09 mg/kg | Bacteriostatic 0.9% Sodium Chloride Injection, USP (0.9% benzyl alcohol) | q.s. to 100 mL |
Since limited compatibility data are available, adherence to the recommended diluents and infusion systems is advised. Solutions containing cladribine injection, USP should not be mixed with other intravenous drugs or additives or infused simultaneously via a common intravenous line, since compatibility testing has not been performed. Preparations containing benzyl alcohol should not be used in neonates (see WARNINGS ).
Care must be taken to assure the sterility of prepared solutions. Once diluted, solutions of cladribine injection, USP should be administered promptly or stored in the refrigerator (2° to 8° C) for no more than 8 hours prior to start of administration. Vials of cladribine injection, USP are for single-use only. Any unused portion should be discarded in an appropriate manner (see Handling and Disposal ).
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. A precipitate may occur during the exposure of cladribine injection, USP to low temperatures; it may be resolubilized by allowing the solution to warm naturally to room temperature and by shaking vigorously. DO NOT HEAT OR MICROWAVE.
Chemical Stability of Vials
When stored in refrigerated conditions between 2° to 8°C (36° to 46°F) protected from light, unopened vials of cladribine injection, USP are stable until the expiration date indicated on the package. Freezing does not adversely affect the solution. If freezing occurs, thaw naturally to room temperature. DO NOT heat or microwave. Once thawed, the vial of cladribine injection, USP is stable until expiry if refrigerated. DO NOT refreeze. Once diluted, solutions containing cladribine injection, USP should be administered promptly or stored in the refrigerator (2° to 8°C) for no more than 8 hours prior to administration.
Handling and Disposal
The potential hazards associated with cytotoxic agents are well established and proper precautions should be taken when handling, preparing, and administering cladribine injection, USP. The use of disposable gloves and protective garments is recommended. If cladribine injection, USP contacts the skin or mucous membranes, wash the involved surface immediately with copious amounts of water. Several guidelines on this subject have been published. (2–8) There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate. Refer to your Institution's guidelines and all applicable state/local regulations for disposal of cytotoxic waste.
CONTRAINDICATIONS
Cladribine Injection, USP is contraindicated in those patients who are hypersensitive to this drug or any of its components.
ADVERSE REACTIONS
Clinical Trials Experience
Adverse drug reactions reported by ≥ 1% of cladribine-treated patients with HCL noted in the HCL clinical dataset (studies K90-091 and L91-048, n=576) are shown in the table below.
| System Organ Class Preferred Term | Cladribine (n=576) % |
|---|---|
| Blood and Lymphatic System Disorder (see also sections WARNINGS and PRECAUTIONS ) | |
| Anemia | 1 |
| Febrile neutropenia | 8 |
| Psychiatric Disorders | |
| Anxiety | 1 |
| Insomnia | 3 |
| Nervous System Disorders | |
| Dizziness | 6 |
| Headache | 14 |
| Cardiac Disorders | |
| Tachycardia | 2 |
| Respiratory, Thoracic and Mediastinal Disorders | |
| Breath sounds abnormal | 4 |
| Cough | 7 |
| Dyspnea Dyspnea includes dyspnea, dyspnea exertional, and wheezing | 5 |
| Rales | 1 |
| Gastrointestinal Disorders | |
| Abdominal pain Abdominal pain includes abdominal discomfort, abdominal pain, and abdominal pain (lower and upper) | 4 |
| Constipation | 4 |
| Diarrhea | 7 |
| Flatulence | 1 |
| Nausea | 22 |
| Vomiting | 9 |
| Skin and Subcutaneous Tissue Disorders | |
| Ecchymosis | 2 |
| Hyperhidrosis | 3 |
| Petechiae | 2 |
| Pruritus | 2 |
| Rash Rash includes erythema, rash, and rash (macular, macula-papular, papular, pruritic, pustular and erythematous) | 16 |
| Musculoskeletal, Connective Tissue, and Bone Disorders | |
| Arthralgia | 3 |
| Myalgia | 6 |
| Pain Pain includes pain, back pain, chest pain, arthritis pain, bone pain, and pain in extremity | 6 |
| General Disorders and Administration Site Conditions (see also sections WARNINGS and PRECAUTIONS ) | |
| Administration site reaction Administration site reaction includes administration site reaction, catheter site (cellulitis, erythema, hemorrhage, and pain), and infusion site reaction(erythema, edema, and pain) | 11 |
| Asthenia | 6 |
| Chills | 2 |
| Decreased appetite | 8 |
| Fatigue | 31 |
| Malaise | 5 |
| Muscular weakness | 1 |
| Edema peripheral | 2 |
| Pyrexia | 33 |
| Injury, Poisoning and Procedural Complications | |
| Contusion | 1 |
The following safety data are based on 196 patients with Hairy Cell Leukemia: the original cohort of 124 patients plus an additional 72 patients enrolled at the same two centers after the original enrollment cutoff. In Month 1 of the Hairy Cell Leukemia clinical trials, severe neutropenia was noted in 70% of patients, fever in 69%, and infection was documented in 28%. Most non-hematologic adverse experiences were mild to moderate in severity.
Myelosuppression was frequently observed during the first month after starting treatment. Neutropenia (ANC < 500 × 10 6 /L) was noted in 70% of patients, compared with 26% in whom it was present initially. Severe anemia (Hemoglobin < 8.5 g/dL) developed in 37% of patients, compared with 10% initially and thrombocytopenia (Platelets < 20 × 10 9 /L) developed in 12% of patients, compared to 4% in whom it was noted initially.
During the first month, 54 of 196 patients (28%) exhibited documented evidence of infection. Serious infections (e.g., septicemia, pneumonia) were reported in 6% of all patients; the remainder were mild or moderate. Several deaths were attributable to infection and/or complications related to the underlying disease. During the second month, the overall rate of documented infection was 6%; these infections were mild to moderate and no severe systemic infections were seen. After the third month, the monthly incidence of infection was either less than or equal to that of the months immediately preceding cladribine therapy.
During the first month, 11% of patients experienced severe fever (i.e., ≥104°F). Documented infections were noted in fewer than one-third of febrile episodes. Of the 196 patients studied, 19 were noted to have a documented infection in the month prior to treatment. In the month following treatment, there were 54 episodes of documented infection: 23 (42%) were bacterial, 11 (20%) were viral and 11 (20%) were fungal. Seven (7) of 8 documented episodes of herpes zoster occurred during the month following treatment. Fourteen (14) of 16 episodes of documented fungal infections occurred in the first two months following treatment. Virtually all of these patients were treated empirically with antibiotics. (see WARNINGS and PRECAUTIONS )
Analysis of lymphocyte subsets indicates that treatment with cladribine is associated with prolonged depression of the CD4 counts. Prior to treatment, the mean CD4 count was 766/µL. The mean CD4 count nadir, which occurred 4 to 6 months following treatment, was 272/µL. Fifteen (15) months after treatment, mean CD4 counts remained below 500/µL. CD8 counts behaved similarly, though increasing counts were observed after 9 months. The clinical significance of the prolonged CD4 lymphopenia is unclear.
Another event of unknown clinical significance includes the observation of prolonged bone marrow hypocellularity. Bone marrow cellularity of < 35% was noted after 4 months in 42 of 124 patients (34%) treated in the two pivotal trials. This hypocellularity was noted as late as Day 1010. It is not known whether the hypocellularity is the result of disease related marrow fibrosis or if it is the result of cladribine toxicity. There was no apparent clinical effect on the peripheral blood counts.
The vast majority of rashes were mild. Most episodes of nausea were mild, not accompanied by vomiting, and did not require treatment with antiemetics. In patients requiring antiemetics, nausea was easily controlled, most frequently with chlorpromazine.
When used in other clinical settings the following ADRs were reported: bacteremia, cellulitis, localized infection, pneumonia, anemia, thrombocytopenia (with bleeding or petechiae), phlebitis, purpura, crepitations, localized edema and edema.
For a description of adverse reactions associated with use of high doses in non-Hairy Cell Leukemia patients, see WARNINGS .
Postmarketing Experience
The following additional adverse reactions have been reported since the drug became commercially available. These adverse reactions have been reported primarily in patients who received multiple courses of cladribine injection:
Infections and infestations: Septic shock. Opportunistic infections have occurred in the acute phase of treatment.
Blood and lymphatic system disorders : Bone marrow suppression with prolonged pancytopenia, including some reports of aplastic anemia; hemolytic anemia (including autoimmune hemolytic anemia), which was reported in patients with lymphoid malignancies, occurring within the first few weeks following treatment. Rare cases of myelodysplastic syndrome have been reported.
Immune system disorders : Hypersensitivity.
Metabolism and nutrition disorders: Tumor lysis syndrome.
Psychiatric disorders : Confusion (including disorientation).
Hepatobiliary disorders : Reversible, generally mild increases in bilirubin (uncommon) and transaminases.
Nervous System disorders : Depressed level of consciousness, neurological toxicity (including peripheral sensory neuropathy, motor neuropathy (paralysis), polyneuropathy, paraparesis); however, severe neurotoxicity has been reported rarely following treatment with standard cladribine dosing regimens.
Eye disorders : Conjunctivitis.
Respiratory, thoracic and mediastinal disorders : Pulmonary interstitial infiltrates (including lung infiltration, interstitial lung disease, pneumonitis and pulmonary fibrosis); in most cases, an infectious etiology was identified.
Skin and tissue disorders : Urticaria, hypereosinophilia; Stevens-Johnson. In isolated cases toxic epidermal necrolysis has been reported in patients who were receiving or had recently been treated with other medications (e.g., allopurinol or antibiotics) known to cause these syndromes.
Renal and urinary disorders: Renal failure (including renal failure acute, renal impairment).
Drug Interactions
There are no known drug interactions with cladribine injection. Caution should be exercised if cladribine injection is administered before, after, or in conjunction with other drugs known to cause immunosuppression or myelosuppression. (see WARNINGS )
DESCRIPTION
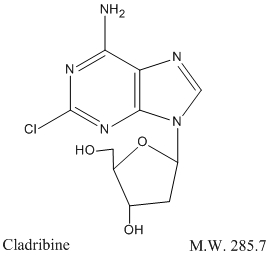
Cladribine Injection, USP (also commonly known as 2-chloro-2΄-deoxy- β -D-adenosine) is a synthetic antineoplastic agent for continuous intravenous infusion. It is a clear, colorless, sterile, preservative-free, isotonic solution. Cladribine injection, USP is available in single-dose vials containing 10 mg (1 mg/mL) of cladribine, a chlorinated purine nucleoside analog. Each milliliter of cladribine injection, USP contains 1 mg of the active ingredient and 9 mg (0.15 mEq) of sodium chloride as an inactive ingredient. The solution has a pH range of 5.5 to 8.0. Phosphoric acid and/or dibasic sodium phosphate may have been added to adjust the pH to 6.3±0.3.
The chemical name for cladribine is 2-chloro-6-amino-9-(2-deoxy-β-D-erythropento-furanosyl) purine and the structure is represented below:
CLINICAL PHARMACOLOGY
Cellular Resistance and Sensitivity
The selective toxicity of 2-chloro-2΄-deoxy-β-D-adenosine towards certain normal and malignant lymphocyte and monocyte populations is based on the relative activities of deoxycytidine kinase and deoxynucleotidase. Cladribine passively crosses the cell membrane. In cells with a high ratio of deoxycytidine kinase to deoxynucleotidase, it is phosphorylated by deoxycytidine kinase to 2-chloro-2΄-deoxy- β -D-adenosine monophosphate (2-CdAMP). Since 2-chloro-2΄-deoxy- β -D-adenosine is resistant to deamination by adenosine deaminase and there is little deoxynucleotide deaminase in lymphocytes and monocytes, 2-CdAMP accumulates intracellularly and is subsequently converted into the active triphosphate deoxynucleotide, 2-chloro-2΄-deoxy- β -D-adenosine triphosphate (2-CdATP). It is postulated that cells with high deoxycytidine kinase and low deoxynucleotidase activities will be selectively killed by 2-chloro-2΄-deoxy- β -D-adenosine as toxic deoxynucleotides accumulate intracellularly.
Cells containing high concentrations of deoxynucleotides are unable to properly repair single-strand DNA breaks. The broken ends of DNA activate the enzyme poly (ADP-ribose) polymerase resulting in NAD and ATP depletion and disruption of cellular metabolism. There is evidence, also, that 2-CdATP is incorporated into the DNA of dividing cells, resulting in impairment of DNA synthesis. Thus, 2-chloro-2΄-deoxy- β -D-adenosine can be distinguished from other chemotherapeutic agents affecting purine metabolism in that it is cytotoxic to both actively dividing and quiescent lymphocytes and monocytes, inhibiting both DNA synthesis and repair.
Pharmacokinetics
In a clinical investigation, 17 patients with Hairy Cell Leukemia and normal renal function were treated for 7 days with the recommended treatment regimen of cladribine injection (0.09 mg/kg/day) by continuous intravenous infusion. The mean steady-state serum concentration was estimated to be 5.7 ng/mL with an estimated systemic clearance of 663.5 mL/h/kg when cladribine was given by continuous infusion over 7 days. In Hairy Cell Leukemia patients, there does not appear to be a relationship between serum concentrations and ultimate clinical outcome.
In another study, 8 patients with hematologic malignancies received a two (2) hour infusion of cladribine injection (0.12 mg/kg). The mean end-of-infusion plasma cladribine concentration was 48±19 ng/mL. For 5 of these patients, the disappearance of cladribine could be described by either a biphasic or triphasic decline. For these patients with normal renal function, the mean terminal half-life was 5.4 hours. Mean values for clearance and steady-state volume of distribution were 978±422 mL/h/kg and 4.5±2.8 L/kg, respectively.
Cladribine plasma concentration after intravenous administration declines multi-exponentially with an average half-life of 6.7 +/- 2.5 hours. In general, the apparent volume of distribution of cladribine is approximately 9 L/kg, indicating an extensive distribution in body tissues.
Cladribine penetrates into cerebrospinal fluid. One report indicates that concentrations are approximately 25% of those in plasma.
Cladribine is bound approximately 20% to plasma proteins.
Except for some understanding of the mechanism of cellular toxicity, no other information is available on the metabolism of cladribine in humans. An average of 18% of the administered dose has been reported to be excreted in urine of patients with solid tumors during a 5-day continuous intravenous infusion of 3.5–8.1 mg/m 2 /day of cladribine. The effect of renal and hepatic impairment on the elimination of cladribine has not been investigated in humans.
CLINICAL STUDIES
Two single-center open label studies of cladribine injection have been conducted in patients with Hairy Cell Leukemia with evidence of active disease requiring therapy. In the study conducted at the Scripps Clinic and Research Foundation (Study A), 89 patients were treated with a single course of cladribine injection given by continuous intravenous infusion for 7 days at a dose of 0.09 mg/kg/day. In the study conducted at the M.D. Anderson Cancer Center (Study B), 35 patients were treated with a 7-day continuous intravenous infusion of cladribine injection at a comparable dose of 3.6 mg/m 2 /day. A complete response (CR) required clearing of the peripheral blood and bone marrow of hairy cells and recovery of the hemoglobin to 12 g/dL, platelet count to 100 × 10 9 /L, and absolute neutrophil count to 1500 × 10 6 /L. A good partial response (GPR) required the same hematologic parameters as a complete response, and that fewer than 5% hairy cells remain in the bone marrow. A partial response (PR) required that hairy cells in the bone marrow be decreased by at least 50% from baseline and the same response for hematologic parameters as for complete response. A pathologic relapse was defined as an increase in bone marrow hairy cells to 25% of pretreatment levels. A clinical relapse was defined as the recurrence of cytopenias, specifically, decreases in hemoglobin ≥ 2 g/dL, ANC ≥ 25% or platelet counts ≥ 50,000. Patients who met the criteria for a complete response but subsequently were found to have evidence of bone marrow hairy cells (< 25% of pretreatment levels) were reclassified as partial responses and were not considered to be complete responses with relapse.
Among patients evaluable for efficacy (N=106), using the hematologic and bone marrow response criteria described above, the complete response rates in patients treated with cladribine injection were 65% and 68% for Study A and Study B, respectively, yielding a combined complete response rate of 66%. Overall response rates (i.e., Complete plus Good Partial plus Partial Responses) were 89% and 86% in Study A and Study B, respectively, for a combined overall response rate of 88% in evaluable patients treated with cladribine injection.
Using an intent-to-treat analysis (N=123) and further requiring no evidence of splenomegaly as a criterion for CR (i.e., no palpable spleen on physical examination and ≤ 13 cm on CT scan), the complete response rates for Study A and Study B were 54% and 53%, respectively, giving a combined CR rate of 54%. The overall response rates (CR + GPR + PR) were 90% and 85%, for Studies A and B, respectively, yielding a combined overall response rate of 89%.
| CR | Overall | |
|---|---|---|
| Evaluable Patients N=106 | 66% | 88% |
| Intent-to-treat Population N=123 | 54% | 89% |
In these studies, 60% of the patients had not received prior chemotherapy for Hairy Cell Leukemia or had undergone splenectomy as the only prior treatment and were receiving cladribine as a first-line treatment. The remaining 40% of the patients received cladribine as a second-line treatment, having been treated previously with other agents, including α-interferon and/or deoxycoformycin. The overall response rate for patients without prior chemotherapy was 92%, compared with 84% for previously treated patients. Cladribine is active in previously treated patients; however, retrospective analysis suggests that the overall response rate is decreased in patients previously treated with splenectomy or deoxycoformycin and in patients refractory to α-interferon.
| OVERALL RESPONSE (N = 123) | NR + RELAPSE | |
|---|---|---|
| NR = No Response | ||
| No Prior Chemotherapy | 68/74 92% | 6 + 4 14% |
| Any Prior Chemotherapy | 41/49 84% | 8 + 3 22% |
| Previous Splenectomy | 32/41 P < 0.05 78% | 9 + 1 24% |
| Previous Interferon | 40/48 83% | 8 + 3 23% |
| Interferon Refractory | 6/11 55% | 5 + 2 64% |
| Previous Deoxycoformycin | 3/6 50% | 3 + 1 66% |
After a reversible decline, normalization of peripheral blood counts (Hemoglobin >12.0 g/dL, Platelets >100 × 10 9 /L, Absolute Neutrophil Count (ANC) >1500 × 10 6 /L) was achieved by 92% of evaluable patients. The median time to normalization of peripheral counts was 9 weeks from the start of treatment (Range: 2 to 72). The median time to normalization of Platelet Count was two weeks, the median time to normalization of ANC was 5 weeks and the median time to normalization of Hemoglobin was 8 weeks. With normalization of Platelet Count and Hemoglobin, requirements for platelet and RBC transfusions were abolished after Months 1 and 2, respectively, in those patients with complete response. Platelet recovery may be delayed in a minority of patients with severe baseline thrombocytopenia. Corresponding to normalization of ANC, a trend toward a reduced incidence of infection was seen after the third month, when compared to the months immediately preceding cladribine therapy. (see also WARNINGS , PRECAUTIONS and ADVERSE REACTIONS )
| Parameter | Median Time to Normalization of Count Day 1 = First day of infusion |
|---|---|
| Platelet Count | 2 weeks |
| Absolute Neutrophil Count | 5 weeks |
| Hemoglobin | 8 weeks |
| ANC, Hemoglobin and Platelet Count | 9 weeks |
For patients achieving a complete response, the median time to response (i.e., absence of hairy cells in bone marrow and peripheral blood together with normalization of peripheral blood parameters), measured from treatment start, was approximately 4 months. Since bone marrow aspiration and biopsy were frequently not performed at the time of peripheral blood normalization, the median time to complete response may actually be shorter than that which was recorded. At the time of data cut-off, the median duration of complete response was greater than 8 months and ranged to 25+ months. Among 93 responding patients, seven had shown evidence of disease progression at the time of the data cut-off. In four of these patients, disease was limited to the bone marrow without peripheral blood abnormalities (pathologic progression), while in three patients there were also peripheral blood abnormalities (clinical progression). Seven patients who did not respond to a first course of cladribine received a second course of therapy. In the five patients who had adequate follow-up, additional courses did not appear to improve their overall response.
HOW SUPPLIED
Cladribine Injection, USP is supplied as a sterile, preservative-free, isotonic solution containing 10 mg (1 mg/mL) of cladribine as 10 mL filled into a single-dose clear flint glass 20 mL vial, individually boxed. NDC 42658-010-01.
Store refrigerated 2° to 8°C (36° to 46°F). Protect from light during storage.
Cladribine - Cladribine injection, Solution PubMed™ news
- Journal Article • 2026 MayCladribine treatment of a young patient with isolated progressive pulmonary Langerhans histiocytosis complicated by invasive aspergillosis.
- Journal Article • 2026 MayEfficacy and Tolerance of Cladribine for Non-Langerhans Cell Histiocytosis.
- Journal Article • 2026 MayUpdated systematic literature review and network meta-analysis of cladribine tablets versus currently approved disease-modifying treatments for active relapsing-remitting multiple sclerosis.
- Journal Article • 2026 AprReal-world outcomes of cladribine in people ≥55 years of age with multiple sclerosis.
- Journal Article • 2026 AprCladribine in multiple sclerosis with autoimmune comorbidities: Further clinical insights from patients with rheumatologic disease treated in combination with secukinumab.