Cryselle prescribing information
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs are contraindicated in women who are over 35 years of age and smoke [see Contraindications ] .
INDICATIONS AND USAGE
Cryselle is indicated for use by females of reproductive potential to prevent pregnancy.
In a study of 1,287 women with a total of 11,085 cycles or 852.7 women-years of usage, the pregnancy rate in women age 15 to 40 years was approximately 1 pregnancy per 100 women-years of use.
CONTRAINDICATIONS
Cryselle is contraindicated in females who are known to have or develop the following conditions:
- A high risk of arterial or venous thrombotic diseases. Examples include women who are known to:
- Smoke, if over age 35
- Have deep-vein thrombosis or pulmonary embolism, now or in the past
- Have inherited or acquired coagulopathies
- Have cerebrovascular disease
- Have coronary artery disease
- Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease or atrial fibrillation)
- Have uncontrolled hypertension
- Have diabetes mellitus with vascular disease
- Headaches with focal neurological symptoms or migraine headaches with aura
- Women over age 35 with any migraine headaches
- Liver tumors, benign or malignant, or liver disease
- Undiagnosed abnormal uterine bleeding
- Pregnancy, because there is no reason to use COCs during pregnancy
- Current diagnosis or history of breast cancer, which may be hormone sensitive
- Hypersensitivity to any of the components of Cryselle
Women who are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see Warnings , Risk of liver enzyme elevations with concomitant hepatitis c treatment ).
ADVERSE REACTIONS
An increased risk of the following serious adverse reactions (see Warnings section for additional information) has been associated with the use of oral contraceptives:
- Serious cardiovascular events and stroke [see Boxed Warning ]
- Vascular events
- Liver disease
Adverse reactions commonly reported by COC users are:
- Irregular uterine bleeding
- Nausea
- Breast tenderness
- Headache
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of Cryselle was evaluated in 1,343 healthy women of child-bearing potential who participated in 9 clinical trials and received at least one dose of Cryselle for contraception. Subjects were exposed for a total of 11,085 cycles, with 429 women completing one year of exposure. Subjects ranged in age from 15 to 40 years. Demographics were 69% Caucasian, 28% Black, and 3% other.
Common Adverse Reactions (≥ 2% of women):
- Weight increase (11%)
- Cervical erosion (9%)
- Weight decrease (6%)
- Acne (4%)
- Dysmenorrhea (4%)
- Vaginal discharge (4%)
- Abdominal pain, cramps, and bloating (3%)
- Appetite increase (3%)
- Depression (3%)
- Nervousness (3%)
- Chloasma/melasma (2%)
- Fatigue (2%)
- Varicose veins, aggravation of (2%)
A total of 8% of subjects discontinued the trials prematurely due to an adverse reaction, most commonly due to unscheduled bleeding, spotting, headache (including migraine), nausea, acne, changes in menstrual flow, weight increase, nervousness, high blood pressure, and depression.
Postmarketing Experience
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 to 1.12 (Figure 1).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 1). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 to 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure 1: Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives
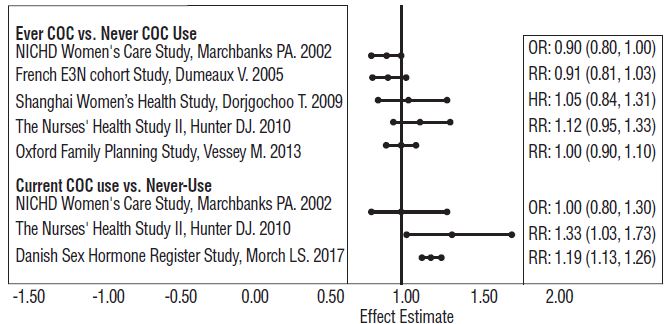
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
The following additional adverse drug reactions have been reported from worldwide postmarketing experience with Cryselle. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Arterial Events: Arterial thromboembolism, Myocardial infarction, Cerebral hemorrhage
Eye Disorder: Optic neuritis, which may lead to partial or complete loss of vision, Intolerance to contact lenses, Change (steepening) in corneal curvature
Gastrointestinal Disorders: Colitis, Nausea, Pancreatitis
Hepatobiliary Disorders: Gallbladder disease, Cholestatic jaundice, Budd-Chiari syndrome
Immune System Disorders: Anaphylactic/anaphylactoid reactions, including urticaria, angioedema, and severe reactions with respiratory and circulatory symptoms
Metabolism and Nutrition Disorders: Carbohydrate and lipid effects, Porphyria, exacerbation of Porphyria
Neoplasms, Benign, Malignant, and Unspecified: Carcinoma of the reproductive organs and breasts , Hepatic neoplasia (including hepatic adenomas or benign liver tumors)
Psychiatric Disorders: Mood changes
Reproductive System and Breast Disorders: Temporary infertility after discontinuation of treatment, Changes in libido, Vaginitis, including candidiasis; Breast secretion
Skin and Subcutaneous Tissue Disorders: Melasma/chloasma, which may persist; Erythema multiforme, Erythema nodosum, Hemorrhagic eruption, Hirsutism
Vascular Events: Venous thrombosis, Pulmonary embolism, Cerebral thrombosis, Mesenteric thrombosis, Retinal vascular thrombosis
OVERDOSAGE
There have been no reports of serious ill effects from overdosage of oral contraceptives, including ingestion by children. Overdosage may cause withdrawal bleeding in females and nausea.
DOSAGE AND ADMINISTRATION
To achieve maximum contraceptive effectiveness, Cryselle (norgestrel and ethinyl estradiol tablets) must be taken exactly as directed and at intervals not exceeding 24 hours. The dosage of Cryselle is one white tablet daily for 21 consecutive days, followed by one light green colored inert tablet daily for 7 consecutive days, according to prescribed schedule. It is recommended that Cryselle tablets be taken by mouth at the same time each day.
How to Start Cryselle
Consider the possibility of ovulation and conception prior to initiation of medication.
Instruct the patient to begin taking Cryselle on the first Sunday after the onset of menstruation. If menstruation begins on a Sunday, the first tablet (white) is taken that day. The patient should take one white tablet daily for 21 consecutive days followed by one light green colored inert tablet daily for 7 consecutive days. Withdrawal bleeding will usually occur within 3 days following discontinuation of white tablets and may not have finished before the next pack is started. During the first cycle, the patient should not rely on Cryselle for contraception until a white tablet has been taken daily for 7 consecutive days and she should use a non-hormonal back-up method of birth control during those 7 days.
After the first cycle of use
The patient is to begin her next and all subsequent 28-day courses of tablets on the same day of the week (Sunday) on which she began her first course, following the same schedule: 21 days of white tablets, followed by 7 days of light green colored inert tablets. If in any cycle the patient starts tablets later than the proper day, instruct her to protect herself against pregnancy by using a non-hormonal back-up method of birth control until she has taken a white tablet daily for 7 consecutive days.
Switching from another hormonal method of contraception
- When the patient is switching from a 21-day regimen of tablets, instruct her to wait 7 days after her last tablet before she starts Cryselle. She will probably experience withdrawal bleeding during that week. Instruct her not to let more than 7 days pass after her previous 21-day regimen before she starts Cryselle.
- When the patient is switching from a 28-day regimen of tablets, instruct her to start her first pack of Cryselle on the day after her last tablet. She should not wait any days between packs.
- The patient may switch any day from a progestin-only pill and should begin Cryselle the next day. If switching from an implant or injection, instruct the patient to start Cryselle on the day of implant removal or the day the next injection would be due. If switching from a contraceptive vaginal ring or transdermal patch instruct the patient to start Cryselle on the day they would have inserted the next ring or applied the next patch. If switching from an Intrauterine Device (IUD) or Intrauterine System (IUS), instruct the patient to start Cryselle on the day of IUD/IUS removal. If the IUD/IUS is not removed on the first day of the patient’s menstrual cycle, instruct her to use a non-hormonal back-up method of birth control for the first 7 days of tablet-taking.
Use after pregnancy, abortion, or miscarriage
- Initiate Cryselle no earlier than day 28 postpartum in the nonlactating mother or after a second-trimester abortion due to the increased risk for thromboembolism (see Contraindications , Warnings and Precautions concerning thromboembolic disease). Advise the patient to use a non-hormonal back-up method for the first 7 days of tablet-taking.
- Cryselle may be initiated immediately after a first-trimester abortion or miscarriage. If the patient starts Cryselle immediately, back-up contraception is not needed.
If spotting or breakthrough bleeding occurs
If spotting or breakthrough bleeding occurs, instruct the patient to continue on the same regimen. This type of bleeding is usually transient and without significance; however, advise the patient to consult her healthcare provider if the bleeding is persistent or prolonged.
Missed Tablets
The possibility of ovulation and pregnancy increases with each successive day that scheduled white tablets are missed. If withdrawal bleeding does not occur, the possibility of pregnancy must be considered. If the patient has not adhered to the prescribed schedule (if she missed one or more tablets or started taking them on a day later than she should have), consider the probability of pregnancy at the time of the first missed period and take appropriate diagnostic measures. If the patient has adhered to the prescribed regimen and misses two consecutive periods, rule out pregnancy.
For additional patient instructions regarding missed tablets, see the WHAT TO DO IF YOU MISS PILLS section in FDA-Approved Patient Labeling below.
Advice in Case of Gastrointestinal Disturbances
In case of severe vomiting or diarrhea, absorption may not be complete and additional contraceptive measures should be taken. If vomiting or diarrhea occurs within 3 to 4 hours after taking an active tablet, handle this as a missed tablet [see FDA-Approved Patient Labeling] .
Nursing Mothers
Advise the nursing mother to use other forms of contraception, when possible, until she has weaned her child. COCs can reduce milk production in breastfeeding mothers. This is less likely to occur once breastfeeding is well-established; however, it can occur at any time in some women. Small amounts of oral contraceptive steroids and/or metabolites are present in breast milk.
DESCRIPTION
Cryselle ® is a combination oral contraceptive containing the progestational compound norgestrel, USP and the estrogenic compound ethinyl estradiol, USP. Norgestrel is designated as (2) (±)-13-Ethyl-17-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one and ethinyl estradiol is designated as (19-nor-17α-pregna-1,3,5 (10)-trien-20-yne-3,17-diol). Each white active Cryselle tablet contains 0.3 mg norgestrel, USP and 0.03 mg ethinyl estradiol, USP. The inactive ingredients present are lactose monohydrate, magnesium stearate, microcrystalline cellulose and pregelatinized corn starch. The light green inactive tablets also contain D&C Yellow No. 10 Aluminum Lake, FD&C Blue No. 1 Aluminum Lake and FD&C Yellow No. 6 Aluminum Lake.
Norgestrel, USP
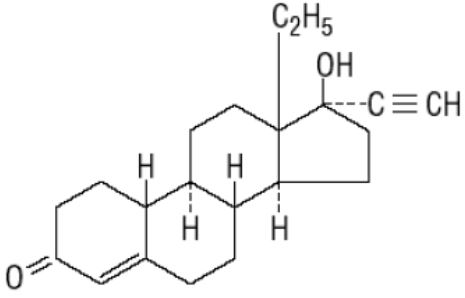
C 21 H 28 O 2 MW: 312.45
Ethinyl Estradiol, USP
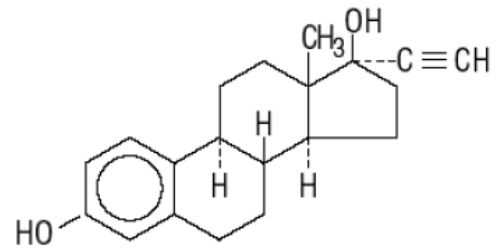
C 20 H 24 O 2 MW: 296.40
CLINICAL PHARMACOLOGY
Mechanism of Action
Combined oral contraceptives (COCs) prevent pregnancy primarily by suppressing ovulation.
HOW SUPPLIED
Cryselle ® (norgestrel and ethinyl estradiol tablets USP), 0.3 mg/0.03 mg are available in packages of 6 blister card dispensers (NDC 0480-3493-16), each containing 28 tablets as follows: 21 active, white, round, biconvex tablets debossed with C33 on one side and TV on the other side and 7 inert, light green, round tablets debossed with T on one side and PL2 on the other side.
Store at 20º to 25°C (68° to 77º F) [see USP Controlled Room Temperature].
Keep this and all medications out of the reach of children.
Manufactured For: Teva Pharmaceuticals Parsippany, NJ 07054
Rev. H 12/2024