Get your patient on Cyklx (Articaine Hydrochloride)
Cyklx prescribing information
INDICATIONS AND USAGE
CYKLX is indicated for ocular surface anesthesia prior to ocular procedures and/or intraocular injections in adults and pediatric patients.
DOSAGE AND ADMINISTRATION
The recommended dose of CYKLX is 2 drops applied 30 seconds apart to the ocular surface.
DOSAGE FORMS AND STRENGTHS
Ophthalmic Solution: clear and colorless solution containing 8% articaine in single-dose vial.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no adequate and well-controlled studies in pregnant women with CYKLX to inform a drug associated risk. In animal reproduction studies, subcutaneous administration of articaine hydrochloride and epinephrine (1:100,000) to rabbits throughout organogenesis resulted in maternal toxicity, embryofetal death and increased fetal skeletal variations at doses approximately 160 times the maximum recommended human ophthalmic dose (MRHOD) of CYKLX (see Data ) .
The background risk of major birth defects and miscarriage for the indicated population is unknown. However, the background risk in the U.S. general population of major birth defects is 2 to 4%, and of miscarriage is 15 to 20%, of clinically recognized pregnancies.
Data
Human Data
No adequate and well-controlled trials of CYKLX have been conducted in pregnant women.
Animal Data
In embryo-fetal toxicity studies, subcutaneous administration of articaine hydrochloride and epinephrine (1:100,000) to pregnant rabbits throughout organogenesis produced maternal toxicity, including seizures, at doses of 80 mg/kg/day (approximately 160-fold higher than the bilateral maximum recommended human ophthalmic dose [MRHOD] of 9.6 mg, based on body surface area). Embryofetal death and skeletal malformations were observed at the same dose
Articaine hydrochloride and epinephrine were not maternally toxic and did not produce adverse embryofetal effects in rats and rabbits at oral doses up to 40 mg/kg/day (approximately 40- and 80-fold higher than the MRHOD, respectively).
In a pre-/ postnatal developmental study in rats, subcutaneous administration of articaine hydrochloride throughout gestation and lactation resulted in severe maternal toxicity, increased the number of stillbirths and adversely affected passive avoidance, a measure of learning, in pups at a dose of 80 mg/kg (approximately 80 times the MRHOD based on body surface area). A dose of 40 mg/kg (approximately 40 times the MRHOD based on body surface area) did not produce these effects.
Lactation
Risk Summary
There is no information regarding the presence of CYKLX or its metabolites in human or animal milk, the effects on the breastfed infant, or the effects on milk production to inform risk of CYKLX to an infant during lactation.
Pediatric Use
The safety and effectiveness of CYKLX for ocular surface anesthesia prior to ocular procedures and/or intraocular injections have been established in pediatric patients. Use of CYKLX for this indication is supported by evidence from adequate and well-controlled studies in adults with additional safety data from a single active-controlled study in pediatric patients aged birth to 11 years old [see Clinical Studies (14) ] .
A similar safety profile was observed between pediatric and adult patients.
Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger adult subjects.
CONTRAINDICATIONS
CYKLX is contraindicated in patients with known hypersensitivity to any component of this preparation.
WARNINGS AND PRECAUTIONS
- CYKLX is not for injection or intraocular administration (5.1 )
- Patients should not touch the eye for at least 10 to 20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye (5.2 )
- Corneal Opacification : Prolonged use of topical ocular anesthetics may produce permanent corneal opacification and ulceration with accompanying visual loss. (5.3 )
- Do not touch the dropper tip to the eye, eyelids, or any other surface as this may contaminate the solution (5.4 )
- For Administration by Healthcare Provider : CYKLX is not intended for patient self-administration (5.5 )
For Topical Ophthalmic Use
CYKLX is not for injection or intraocular administration.
Corneal Injury Due to Insensitivity
Patients should not touch the eye for at least 10 to 20 minutes after using CYKLX as accidental injuries can occur due to insensitivity of the eye.
Corneal Opacification
Prolonged use of topical ocular anesthetics may produce permanent corneal opacification and ulceration with accompanying visual loss.
Risk of Contamination
Do not touch the dropper tip to the eye, eyelids, or any other surface as this may contaminate the solution.
For Administration by Healthcare Provider
CYKLX is indicated for administration under the direct supervision of a healthcare provider. CYKLX is not intended for patient self-administration.
ADVERSE REACTIONS
The most common adverse reaction was instillation site pain (incidence 24.5%). (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact American Genomics at 855-242-9559 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled trials, the most common adverse reaction with CYKLX was instillation site pain reported in 24.5% (70/286) of subjects, compared to 6.9% (14/203) of subjects in the placebo group.
DESCRIPTION
CYKLX (articaine ophthalmic solution) 8% contains articaine, an amide local anesthetic. CYKLX is an aseptically prepared, sterile, clear and colorless solution for topical ophthalmic use with a pH range of 4.5- 5.0.
The chemical name of articaine hydrochloride is methyl 4-methyl-3-[2-(propylamino)propionamido]-2-thiophenecarboxylate, monohydrochloride and the molecular formula is C 13 H 20 N 2 O 3 S∙HCl. The molecular weight of articaine hydrochloride is 320.84 g/mol, or 283.374 g/mol as articaine (free base). Articaine hydrochloride is represented by the following structural formula:
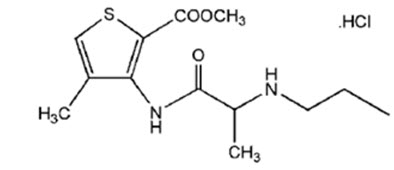
Each mL of CYKLX contains 80 mg of articaine (equivalent to 90.2 mg articaine hydrochloride) as the active ingredient. Inactive ingredients: boric acid, edetate disodium dihydrate, glacial acetic acid, mannitol, sodium acetate trihydrate, water for injection. CYKLX does not contain an antimicrobial preservative.
CLINICAL PHARMACOLOGY
Mechanism of Action
Articaine hydrochloride is an amide local anesthetic. Local anesthetics block the generation and conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of the affected nerve fibers.
Pharmacokinetics
Following ocular administration of CYKLX (2 drops), the geometric mean (geometric CV%) maximum plasma concentration (C max ) is 5.4 ng/mL and total systemic exposure (AUC) is 5.1 ng•hr/mL.
Absorption
Articaine median (min, max) time to maximum plasma concentration (T max ) is 0.25 hour (0.25 – 1.03 hour)
Distribution
Approximately 60 to 80% of articaine HCl is bound to human serum albumin and γ-globulins at 37°C in vitro.
Elimination
Articaine estimated elimination half-life is 1.5 hours with an apparent clearance of 872 L/hr.
Metabolism
Articaine HCl is metabolized by plasma carboxyesterase to its primary metabolite, articainic acid, which is inactive. In vitro studies show that the human liver microsome P450 isoenzyme system metabolizes approximately 5% to 10% of available articaine with nearly quantitative conversion to articainic acid.
Excretion
Articaine is excreted primarily through urine with 53-57% of the administered dose eliminated in the first 24 hours following submucosal administration. Articainic acid is the primary metabolite in urine. A minor metabolite, articainic acid glucuronide, is also excreted in urine.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long term studies in animals have not been performed to evaluate carcinogenic potential.
Mutagenesis
Articaine hydrochloride did not show any potential to cause mutagenicity or genetic toxicity in five standard tests, including three in vitro tests (the nonmammalian Ames test, the mammalian Chinese hamster ovary chromosomal aberration test, and a mammalian gene mutation test with articaine HCl) and two in vivo mouse micronucleus tests (one with articaine and epinephrine 1:100,000 and one with articaine HCl alone).
Impairment of Fertility
Subcutaneous administration of articaine and epinephrine (1:100,000) produced no adverse effects on fertility in rats at doses up to 80 mg/kg/day (approximately 80 times the MRHOD based on body surface area).
CLINICAL STUDIES
The efficacy of CYKLX was evaluated in two randomized, placebo-controlled, double masked, parallel-arm studies in 240 healthy subjects. A total of 120 subjects were randomized to CYKLX (2 drops 30 seconds apart) and 120 subjects were randomized to vehicle.
More than two-thirds of subjects treated with CYKLX achieved ocular anesthesia at 5 minutes. The treatment effect (difference from placebo) was 65% in both studies (P<0.01).
HOW SUPPLIED/STORAGE AND HANDLING
CYKLX 8%, is supplied as an aseptically prepared, sterile, clear and colorless solution for topical ophthalmic use contained in a low-density polyethylene, single-dose vial with a 0.4 mL fill. 5 single-dose vials are packaged into a foil pouch, with 10 foil pouches in a carton.
NDC 87047-7852-01: Carton of 10 foil pouches, 50 single-dose vials.
Storage and Handling:
Store at 20°C to 25°C (68°F to 77°F), not to exceed the expiration date printed on the carton and pouch. Store unopened single-dose vials in pouch until ready for use. Discard opened vial after use.
Mechanism of Action
Articaine hydrochloride is an amide local anesthetic. Local anesthetics block the generation and conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of the affected nerve fibers.