Get your patient on Cysto - Conray Ii - Iothalamate Meglumine injection (Iothalamate Meglumine)
Cysto - Conray Ii - Iothalamate Meglumine injection prescribing information
NOT FOR INTRAVASCULAR ADMINISTRATION
NOT FOR INTRATHECAL USE
INDICATIONS AND USAGE
Cysto-Conray II is indicated for use in retrograde cystography and cystourethrography.
DOSAGE AND ADMINISTRATION
Patient Preparation
Unless contraindicated, an appropriate laxative is given the night before the examination.
Radiographic Technique
The radiographic procedure normally employed for cystography and cystourethrography should be employed. A preliminary radiograph is recommended before the contrast agent is administered.
Administration
Sterile catherization is essential. Cysto-Conray II may be introduced by gravity flow using an appropriate venoclysis set or by syringe. Excessive pressure should be avoided with any method of administration.
Usual Dosage
Cystography and Cystourethrography – Either Conray 43 supplied at 43% w/v or Cysto-Conray II supplied at a 17.2% w/v concentration may be used for these procedures. The desired concentration will vary depending upon the patient’s size and age and also with the technique and equipment used. (SEE CONRAY 43 PACKAGE INSERT FOR SPECIFIC INFORMATION ABOUT THE USE OF THIS PRODUCT.) Sufficient volume of contrast medium is administered to adequately fill the urinary bladder. The volume of solution required will vary depending upon the individual patient. Adults usually require a volume in the range of 200 to 400 mL. Children require a volume in proportion to their body size. The usual dose ranges from 30 to 300 mL.
CONTRAINDICATIONS
See WARNINGS concerning inadvertant intrathecal administration.
ADVERSE REACTIONS
Irritation of the bladder or ureter, common to some degree to all contrast media administered for retrograde urographic procedures, may occasionally occur.
As with all contrast media, intravasation may lead to hypersensitivity reactions such as a sense of warmth, flushing, sneezing, sweating, chills, fever, urticaria, laryngeal edema, bronchospasm, hypertension, hypotension, cardiac arrhythmias and cardiac arrest.
Adverse reactions associated with procedural technique include injury to the urethra, bladder, ureter, and introduction of infection.
In the event of serious or anaphylactoid reactions, it should be kept in mind that the reactions known to occur with intravenous administration of radiopaque contrast materials are possible.
DESCRIPTION
Cysto-Conray II is a sterile aqueous solution intended for instillation as a diagnostic radiopaque medium. Cysto-Conray II contains 17.2% w/v iothalamate meglumine which is 1-Deoxy-1-(methylamino)-D-glucitol 5-acetamido-2,4,6-triiodo-N-methylisophthalamate (salt) and has the following structural formula:
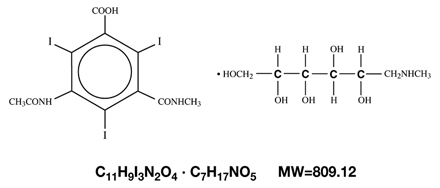
Each milliliter of Cysto-Conray II contains 172 mg of iothalamate meglumine, equivalent to 81 mg (8.1% w/v) of organically bound iodine, 0.110 mg edetate calcium disodium as a stabilizer and 0.115 mg of monobasic sodium phosphate as a buffer.
Cysto-Conray II is hypertonic under conditions of use and is supplied in containers from which the air has been displaced by nitrogen. The pH of Cysto-Conray II is 6.6 to 7.6.
CLINICAL PHARMACOLOGY
The most important characteristic of contrast media is the iodine content. The relatively high atomic weight of iodine contributes sufficient radiodensity for radiographic contrast.
Following instillation by sterile catheter, Cysto-Conray II provides for visualization of the lower urinary tract. Clinical literature reports indicate that routinely less than 1 percent of a retrograde urographic radiopaque is absorbed systemically, however, as much as 12 percent absorption was observed with pyelorenal back flow and may produce iodine medicated thyrotropic effects described under PRECAUTIONS .
HOW SUPPLIED
| Cysto-Conray ® II Glass Bottles | NDC Number |
| 12 x 250 mL single dose bottles | 0019-0862-50 |
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Exposing this product to very cold temperatures may result in crystallization of the salt. If this occurs, the containers should be brought to room temperature. Shake vigorously to assure complete dissolution of any crystals. The speed of dissolution may be increased by heating with circulating warm air. Before use, examine the product to assure that all solids are redissolved and that the container and closure have not been damaged.
This product is sensitive to light and must be protected from strong daylight or direct exposure to the sun.
As with all contrast media, the containers should be inspected prior to use to ensure that breakage or other damage has not occurred during shipping and handling. All containers should be inspected for closure integrity. Damaged containers should not be used.
Manufactured by: Liebel-Flarsheim Company LLC Raleigh, NC 27616
Made in USA
GBT 0862A0817 Revised 08/17
Guerbet