Get your patient on Dacarbazine - Dacarbazine injection, Powder, For Solution (Dacarbazine)
Dacarbazine - Dacarbazine injection, Powder, For Solution prescribing information
WARNING
It is recommended that dacarbazine be administered under the supervision of a qualified physician experienced in the use of cancer chemotherapeutic agents.
1. Hemopoietic depression is the most common toxicity with dacarbazine (see WARNINGS ).
2. Hepatic necrosis has been reported (see WARNINGS ).
3. Studies have demonstrated this agent to have a carcinogenic and teratogenic effect when used in animals.
4. In treatment of each patient, the physician must weigh carefully the possibility of achieving therapeutic benefit against the risk of toxicity.
INDICATIONS AND USAGE
Dacarbazine for Injection is indicated in the treatment of metastatic malignant melanoma. In addition, Dacarbazine for Injection is also indicated for Hodgkin’s disease as a second-line therapy when used in combination with other effective agents.
DOSAGE AND ADMINISTRATION
Malignant Melanoma
The recommended dosage is 2 to 4.5 mg/kg/day for 10 days. Treatment may be repeated at 4 week intervals. 2
An alternate recommended dosage is 250 mg/square meter body surface/day I.V. for 5 days. Treatment may be repeated every 3 weeks. 3,4
Hodgkin's Disease
The recommended dosage of Dacarbazine for Injection, USP in the treatment of Hodgkin’s disease is 150 mg/square meter body surface/day for 5 days, in combination with other effective drugs. Treatment may be repeated every 4 weeks. 5 An alternative recommended dosage is 375 mg/square meter body surface on day 1, in combination with other effective drugs, to be repeated every 15 days. 6
Dacarbazine for Injection, USP 100 mg/vial and 200 mg/vial are reconstituted with 9.9 mL and 19.7 mL, respectively, of Sterile Water for Injection, USP. The resulting solution contains 10 mg/mL of dacarbazine having a pH of 3.0 to 4.0. The calculated dose of the resulting solution is drawn into a syringe and administered only intravenously.
The reconstituted solution may be further diluted with 5% Dextrose Injection or Sodium Chloride Injection and administered as an intravenous infusion.
After reconstitution and prior to use, the solution in the vial may be stored at 4°C for up to 72 hours or at normal room conditions (temperature and light) for up to 8 hours. If the reconstituted solution is further diluted in 5% Dextrose Injection or Sodium Chloride Injection, the resulting solution may be stored at 4°C for up to 24 hours or at normal room conditions for up to 8 hours.
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published. 7-13 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Dacarbazine for Injection is contraindicated in patients who have demonstrated a hypersensitivity to it in the past.
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Symptoms of anorexia, nausea, and vomiting are the most frequently noted of all toxic reactions. Over 90% of patients are affected with the initial few doses. The vomiting lasts 1-12 hours and is incompletely and unpredictably palliated with phenobarbital and/or prochlorperazine. Rarely, intractable nausea and vomiting have necessitated discontinuance of therapy with dacarbazine for injection. Rarely, dacarbazine has caused diarrhea. Some helpful suggestions include restricting the patient’s oral intake of food for 4-6 hours prior to treatment. The rapid toleration of these symptoms suggests that a central nervous system mechanism may be involved, and usually these symptoms subside after the first 1 or 2 days.
There are a number of minor toxicities that are infrequently noted. Patients have experienced an influenza-like syndrome of fever to 39°C, myalgias and malaise. These symptoms occur usually after large single doses, may last for several days, and they may occur with successive treatments.
Alopecia has been noted as has facial flushing and facial paresthesia. There have been few reports of significant liver or renal function test abnormalities in man. However, these abnormalities have been observed more frequently in animal studies.
Erythematous and urticarial rashes have been observed infrequently after administration of dacarbazine for injection. Rarely, photosensitivity reactions may occur.
DESCRIPTION
Dacarbazine for Injection, USP is a white to pale yellow colored solid which is light sensitive. Each vial contains 100 mg of dacarbazine, or 200 mg of dacarbazine (the active ingredient), citric acid and mannitol. Dacarbazine for Injection, USP is reconstituted and administered intravenously (pH 3-4). Dacarbazine for Injection, USP is an anticancer agent. Chemically, dacarbazine is 5-(3,3-Dimethyl-1-triazeno) imidazole-4-carboxamide with the following structural formula:
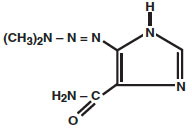
M.W. 182.19 C 6 H 10 N 6 O
CLINICAL PHARMACOLOGY
After intravenous administration of dacarbazine for injection, the volume of distribution exceeds total body water content suggesting localization in some body tissue, probably the liver. Its disappearance from the plasma is biphasic with initial half-life of 19 minutes and a terminal half-life of 5 hours. 1 In a patient with renal and hepatic dysfunctions, the half-lives were lengthened to 55 minutes and 7.2 hours. 1 The average cumulative excretion of unchanged dacarbazine in the urine is 40% of the injected dose in 6 hours. 1 Dacarbazine is subject to renal tubular secretion rather than glomerular filtration. At therapeutic concentrations dacarbazine is not appreciably bound to human plasma protein.
In man, dacarbazine for injection is extensively degraded. Besides unchanged dacarbazine, 5-aminoimidazole -4 carboxamide (AIC) is a major metabolite of dacarbazine excreted in the urine. AIC is not derived endogenously but from the injected dacarbazine, because the administration of radioactive dacarbazine labeled with 14 C in the imidazole portion of the molecule (dacarbazine-2- 14 C) gives rise to AIC-2- 14 C. 1
Although the exact mechanism of action of dacarbazine is not known, three hypotheses have been offered:
1. inhibition of DNA synthesis by acting as a purine analog
2. action as an alkylating agent
3. interaction with SH groups
HOW SUPPLIED
DACARBAZINE FOR INJECTION, USP is available in the following forms:
| Product Code | Unit of Sale | Strength | Each |
| 102710 | NDC 63323-127-10 Unit of 10 | 100 mg | NDC 63323-127-00 10 mL Single Dose Vial |
| 102820 | NDC 63323-128-20 Unit of 10 | 200 mg | NDC 63323-128-00 20 mL Single Dose Vial |
STORE IN A REFRIGERATOR 2°-8°C (36°-46°F).
PROTECT FROM LIGHT.
USE WITHIN 8 HOURS OF RECONSTITUTION.
Vial stoppers do not contain natural rubber latex.