Get your patient on Dantrium - Dantrolene Sodium injection (Dantrolene Sodium)
Dantrium - Dantrolene Sodium injection prescribing information
INDICATIONS AND USAGE:
Dantrium Intravenous is indicated, along with appropriate supportive measures, for the management of the fulminant hypermetabolism of skeletal muscle characteristic of malignant hyperthermia crises in patients of all ages. Dantrium Intravenous should be administered by continuous rapid intravenous push as soon as the malignant hyperthermia reaction is recognized (i.e., tachycardia, tachypnea, central venous desaturation, hypercarbia, metabolic acidosis, skeletal muscle rigidity, increased utilization of anesthesia circuit carbon dioxide absorber, cyanosis and mottling of the skin, and, in many cases, fever).
Dantrium Intravenous is also indicated preoperatively, and sometimes postoperatively, to prevent or attenuate the development of clinical and laboratory signs of malignant hyperthermia in individuals judged to be malignant hyperthermia susceptible.
DOSAGE AND ADMINISTRATION:
As soon as the malignant hyperthermia reaction is recognized, all anesthetic agents should be discontinued; the administration of 100% oxygen is recommended. Dantrium Intravenous should be administered by continuous rapid intravenous push beginning at a minimum dose of 1 mg/kg, and continuing until symptoms subside or the maximum cumulative dose of 10 mg/kg has been reached.
If the physiologic and metabolic abnormalities reappear, the regimen may be repeated. It is important to note that administration of Dantrium Intravenous should be continuous until symptoms subside. The effective dose to reverse the crisis is directly dependent upon the individual's degree of susceptibility to malignant hyperthermia, the amount and time of exposure to the triggering agent, and the time elapsed between onset of the crisis and initiation of treatment.
Pediatric Dose:
Experience to date indicates that the dose of Dantrium Intravenous for pediatric patients is the same as for adults.
Preoperatively:
Dantrium Intravenous and/or Dantrium Capsules may be administered preoperatively to patients judged malignant hyperthermia susceptible as part of the overall patient management to prevent or attenuate the development of clinical and laboratory signs of malignant hyperthermia.
Dantrium Intravenous: The recommended prophylactic dose of Dantrium Intravenous is 2.5 mg/kg, starting approximately 1-1/4 hours before anticipated anesthesia and infused over approximately 1 hour. This dose should prevent or attenuate the development of clinical and laboratory signs of malignant hyperthermia provided that the usual precautions, such as avoidance of established malignant hyperthermia triggering agents, are followed.
Additional Dantrium Intravenous may be indicated during anesthesia and surgery because of the appearance of early clinical and/or blood gas signs of malignant hyperthermia or because of prolonged surgery (see also CLINICAL PHARMACOLOGY , WARNINGS , and PRECAUTIONS ). Additional doses must be individualized.
Oral Administration of Dantrium Capsules:
Administer 4 to 8 mg/kg/day of oral Dantrium Capsules in three or four divided doses for 1 or 2 days prior to surgery, with the last dose being given with a minimum of water approximately 3 to 4 hours before scheduled surgery. Adjustment can usually be made within the recommended dosage range to avoid incapacitation (weakness, drowsiness, etc.) or excessive gastrointestinal irritation (nausea and/or vomiting). See also the package insert for Dantrium Capsules .
Post Crisis Follow-Up:
Dantrium Capsules , 4 to 8 mg/kg/day, in four divided doses should be administered for 1 to 3 days following a malignant hyperthermia crisis to prevent recurrence of the manifestations of malignant hyperthermia.
Dantrium Intravenous may be used postoperatively to prevent or attenuate the recurrence of signs of malignant hyperthermia when oral administration of Dantrium Capsules is not practical. The dose of Dantrium Intravenous in the postoperative period must be individualized, starting with 1 mg/kg or more as the clinical situation dictates.
PREPARATION:
Each vial of Dantrium Intravenous should be reconstituted by adding 60 mL of sterile water for injection USP (without a bacteriostatic agent), and the vial shaken until the solution is clear. 5% Dextrose Injection USP, 0.9% Sodium Chloride Injection USP, and other acidic solutions are not compatible with Dantrium Intravenous and should not be used. The contents of the vial must be protected from direct light and used within 6 hours after reconstitution. Store reconstituted solutions between 15° to 30°C (59° to 86°F).
Reconstituted Dantrium Intravenous should not be transferred to large glass bottles for prophylactic infusion due to precipitate formation observed with the use of some glass bottles as reservoirs.
For prophylactic infusion, the required number of individual vials of Dantrium Intravenous should be reconstituted as outlined above. The contents of individual vials are then transferred to a larger volume sterile intravenous plastic bag. Stability data on file at Par Health indicate commercially available sterile plastic bags are acceptable drug delivery devices. However, it is recommended that the prepared infusion be inspected carefully for cloudiness and/or precipitation prior to dispensing and administration. Such solutions should not be used. While stable for 6 hours, it is recommended that the infusion be prepared immediately prior to the anticipated dosage administration time.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
CONTRAINDICATIONS:
None.
ADVERSE REACTIONS:
There have been occasional reports of death following malignant hyperthermia crisis even when treated with intravenous dantrolene; incidence figures are not available (the pre-dantrolene mortality of malignant hyperthermia crisis was approximately 50%). Most of these deaths can be accounted for by late recognition, delayed treatment, inadequate dosage, lack of supportive therapy, intercurrent disease and/or the development of delayed complications such as renal failure or disseminated intravascular coagulopathy. In some cases there are insufficient data to completely rule out therapeutic failure of dantrolene.
There are reports of fatality in malignant hyperthermia crisis, despite initial satisfactory response to intravenous dantrolene, which involve patients who could not be weaned from dantrolene after initial treatment.
The administration of Dantrium Intravenous to human volunteers is associated with loss of grip strength and weakness in the legs, as well as drowsiness and dizziness.
The following adverse reactions are in approximate order of severity:
There are rare reports of pulmonary edema developing during the treatment of malignant hyperthermia crisis in which the diluent volume and mannitol needed to deliver intravenous dantrolene possibly contributed.
There have been reported cases of hepatotoxicity following the use of intravenous dantrolene products. Elevated liver enzymes have occurred hours to days following use of intravenous dantrolene, though many of these cases were observed in patients with comorbidities (e.g., critical illness).
There have been reports of thrombophlebitis following administration of intravenous dantrolene; actual incidence figures are not available. Tissue necrosis secondary to extravasation has been reported.
There have been rare reports of urticaria and erythema possibly associated with the administration of Dantrium Intravenous . There has been one case of anaphylaxis.
Injection site reactions (pain, erythema, swelling), commonly due to extravasation, have been reported.
The following events have been reported in patients receiving oral dantrolene: aplastic anemia, leukopenia, lymphocytic lymphoma, and heart failure. (See package insert for Dantrium (dantrolene sodium) Capsules for a complete listing of adverse reactions.)
The published literature has included some reports of Dantrium use in patients with Neuroleptic Malignant Syndrome (NMS). Dantrium Intravenous is not indicated for the treatment of NMS and patients may expire despite treatment with Dantrium Intravenous .
For medical advice about adverse reactions contact your medical professional. To report SUSPECTED ADVERSE REACTIONS, contact Par Health at 1-800-828-9393 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions:
Dantrium is metabolized by the liver, and it is theoretically possible that its metabolism may be enhanced by drugs known to induce hepatic microsomal enzymes. However, neither phenobarbital nor diazepam appears to affect Dantrium metabolism. Binding to plasma protein is not significantly altered by diazepam, diphenylhydantoin, or phenylbutazone. Binding to plasma proteins is reduced by warfarin and clofibrate and increased by tolbutamide.
Cardiovascular collapse in association with marked hyperkalemia has been reported in patients receiving dantrolene in combination with calcium channel blockers. It is recommended that the combination of intravenous dantrolene sodium and calcium channel blockers, such as verapamil, not be used together during the management of malignant hyperthermia crisis.
Administration of dantrolene may potentiate vecuronium-induced neuromuscular block.
DESCRIPTION:
Dantrium Intravenous is a sterile, non-pyrogenic, lyophilized formulation of dantrolene sodium for injection. Dantrium Intravenous is supplied in 70 mL vials containing 20 mg dantrolene sodium, 3000 mg mannitol, and sufficient sodium hydroxide to yield a pH of approximately 9.5 when reconstituted with 60 mL sterile water for injection USP (without a bacteriostatic agent).
Dantrium is classified as a direct-acting skeletal muscle relaxant. Chemically, Dantrium is hydrated 1-[[[5-(4-nitrophenyl)-2-furanyl]methylene]amino]-2,4-imidazolidinedione sodium salt. The structural formula for the hydrated salt is:
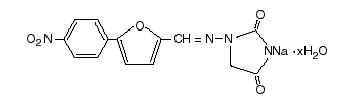
The hydrated salt contains approximately 15% water (3-1/2 moles) and has a molecular weight of 399. The anhydrous salt (dantrolene) has a molecular weight of 336.
CLINICAL PHARMACOLOGY:
In isolated nerve-muscle preparation, Dantrium has been shown to produce relaxation by affecting the contractile response of the muscle at a site beyond the myoneural junction. In skeletal muscle, Dantrium dissociates the excitation-contraction coupling, probably by interfering with the release of Ca++ from the sarcoplasmic reticulum. The administration of intravenous Dantrium to human volunteers is associated with loss of grip strength and weakness in the legs, as well as subjective CNS complaints (see also PRECAUTIONS, Information for Patients ). Information concerning the passage of Dantrium across the blood-brain barrier is not available.
In the anesthetic-induced malignant hyperthermia syndrome, evidence points to an intrinsic abnormality of skeletal muscle tissue. In affected humans, it has been postulated that "triggering agents" (e.g., general anesthetics and depolarizing neuromuscular blocking agents) produce a change within the cell which results in an elevated myoplasmic calcium. This elevated myoplasmic calcium activates acute cellular catabolic processes that cascade to the malignant hyperthermia crisis.
It is hypothesized that addition of Dantrium to the "triggered" malignant hyperthermic muscle cell reestablishes a normal level of ionized calcium in the myoplasm. Inhibition of calcium release from the sarcoplasmic reticulum by Dantrium reestablishes the myoplasmic calcium equilibrium, increasing the percentage of bound calcium. In this way, physiologic, metabolic, and biochemical changes associated with the malignant hyperthermia crisis may be reversed or attenuated. Experimental results in malignant hyperthermia susceptible swine show that prophylactic administration of intravenous or oral dantrolene prevents or attenuates the development of vital sign and blood gas changes characteristic of malignant hyperthermia in a dose related manner. The efficacy of intravenous dantrolene in the treatment of human and porcine malignant hyperthermia crisis, when considered along with prophylactic experiments in malignant hyperthermia susceptible swine, lends support to prophylactic use of oral or intravenous dantrolene in malignant hyperthermia susceptible humans. When prophylactic intravenous dantrolene is administered as directed, whole blood concentrations remain at a near steady state level for 3 or more hours after the infusion is completed. Clinical experience has shown that early vital sign and/or blood gas changes characteristic of malignant hyperthermia may appear during or after anesthesia and surgery despite the prophylactic use of dantrolene and adherence to currently accepted patient management practices. These signs are compatible with attenuated malignant hyperthermia and respond to the administration of additional intravenous dantrolene (see DOSAGE AND ADMINISTRATION ). The administration of the recommended prophylactic dose of intravenous dantrolene to healthy volunteers was not associated with clinically significant cardiorespiratory changes.
Specific metabolic pathways for the degradation and elimination of Dantrium in humans have been established. Dantrolene is found in measurable amounts in blood and urine. Its major metabolites in body fluids are 5-hydroxy dantrolene and an acetylamino metabolite of dantrolene. Another metabolite with an unknown structure appears related to the latter. Dantrium may also undergo hydrolysis and subsequent oxidation forming nitrophenylfuroic acid.
The mean biologic half-life of Dantrium after intravenous administration is variable, between 4 to 8 hours under most experimental conditions. Based on assays of whole blood and plasma, slightly greater amounts of dantrolene are associated with red blood cells than with the plasma fraction of blood. Significant amounts of dantrolene are bound to plasma proteins, mostly albumin, and this binding is readily reversible.
Cardiopulmonary depression has not been observed in malignant hyperthermia susceptible swine following the administration of up to 7.5 mg/kg intravenous dantrolene. This is twice the amount needed to maximally diminish twitch response to single supramaximal peripheral nerve stimulation (95% inhibition). A transient, inconsistent, depressant effect on gastrointestinal smooth muscles has been observed at high doses.
HOW SUPPLIED:
Dantrium Intravenous ( NDC 42023-123-06) is available in vials containing a sterile lyophilized mixture of 20 mg dantrolene sodium, 3000 mg mannitol, and sufficient sodium hydroxide to yield a pH of approximately 9.5 when reconstituted with 60 mL sterile water for injection USP (without a bacteriostatic agent).
Store unreconstituted product between 20° to 25°C (68° to 77°F). (See USP Controlled Room Temperature). Avoid prolonged exposure to light.