Get your patient on Dexamethasone - Dexamethasone elixir (Dexamethasone)
Dexamethasone - Dexamethasone elixir prescribing information
INDICATIONS AND USAGE
- Endocrine Disorders: Primary or secondary adrenocortical insufficiency (hydrocortisone or cortisone is the first choice; synthetic analogs may be used in conjunction with mineralocorticoids where applicable; in infancy mineralocorticoid supplementation is of particular importance). Congenital adrenal hyperplasia Nonsuppurative thyroiditis Hypercalcemia associated with cancer
- Rheumatic Disorders: As adjunctive therapy for short-term administration (to tide the patient over an acute episode or exacerbation) in: Psoriatic arthritis Rheumatoid arthritis, including juvenile rheumatoid arthritis (selected cases may require low-dose maintenance therapy) Ankylosing spondylitis Acute and subacute bursitis Acute nonspecific tenosynovitis Acute gouty arthritis Post-traumatic osteoarthritis Synovitis of osteoarthritis Epicondylitis
- Collagen Diseases: During an exacerbation or as maintenance therapy in selected cases of: Systemic lupus erythematosus Acute rheumatic carditis
- Dermatologic Diseases: Pemphigus Bullous dermatitis herpetiformis Severe erythema multiforme (Stevens-Johnson syndrome) Exfoliative dermatitis Mycosis fungoides Severe psoriasis Severe seborrheic dermatitis
- Allergic States: Control of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment: Seasonal or perennial allergic rhinitis Bronchial asthma Contact dermatitis Atopic dermatitis Serum sickness Drug hypersensitivity reactions
- Ophthalmic Diseases: Severe acute and chronic allergic and inflammatory processes involving the eye and its adnexa, such as: Allergic conjunctivitis Keratitis Allergic corneal marginal ulcers Herpes zoster ophthalmicus Iritis and iridocyclitis Chorioretinitis Anterior segment inflammation Diffuse posterior uveitis and choroiditis Optic neuritis Sympathetic ophthalmia
- Respiratory Diseases: Symptomatic sarcoidosis Loeffler's syndrome not manageable by other means Berylliosis Fulminating or disseminated pulmonary tuberculosis when used concurrently with appropriate antituberculous chemotherapy Aspiration pneumonitis
- Hematologic Disorders: Idiopathic thrombocytopenic purpura in adults Secondary thrombocytopenia in adults Acquired (autoimmune) hemolytic anemia Erythroblastopenia (RBC anemia) Congenital (erythroid) hypoplastic anemia
- Neoplastic Diseases : For palliative management of: Leukemia and lymphomas in adults Acute leukemia of childhood
- Edematous States: To induce a diuresis or remission of proteinuria in the nephrotic syndrome, without uremia, of the idiopathic type or that due to lupus erythematosus
- Gastrointestinal Diseases: To tide the patient over a critical period of the disease in: Ulcerative colitis Regional enteritis
- Miscellaneous: Tuberculous meningitis with subarachnoid block or impending block when used concurrently with appropriate antituberculous chemotherapy Trichinosis with neurologic or myocardial involvement
- Diagnostic testing of adrenocortical hyperfunction.
DOSAGE & ADMINISTRATION
For oral administration: DOSAGE REQUIREMENTS ARE VARIABLE AND MUST BE INDIVIDUALIZED ON THE BASIS OF THE DISEASE AND THE RESPONSE OF THE PATIENT. The initial dosage varies from 0.75 to 9 mg a day depending on the disease being treated. In less severe diseases doses lower than 0.75 mg may suffice, while in severe diseases doses higher than 9 mg may be required. The initial dosage should be maintained or adjusted until the patient’s response is satisfactory. If satisfactory clinical response does not occur after a reasonable period of time, discontinue Dexamethasone Elixir and transfer the patient to other therapy. After a favorable initial response, the proper maintenance dosage should be determined by decreasing the initial dosage in small amounts to the lowest dosage that maintains an adequate clinical response. Patients should be observed closely for signs that might require dosage adjustment, including changes in clinical status resulting from remissions or exacerbations of the disease, individual drug responsiveness, and the effect of stress (e.g., surgery, infection, trauma). During stress it may be necessary to increase dosage temporarily. If the drug is to be stopped after more than a few days of treatment, it usually should be withdrawn gradually. The following milligram equivalents facilitate changing to Dexamethasone Elixir from other glucocorticoids:
| Dexamethasone Elixir | Methylprednisolone and Triamcinolone | Prednisolone and Prednisone | Hydrocortisone | Cortisone |
| 0.75 mg = | 4 mg = | 5 mg = | 20 mg = | 25 mg |
Dexamethasone suppression tests 1. Tests for Cushing’s syndrome. Give 1 mg of Dexamethasone orally at 11:00 p.m. Blood is drawn for plasma cortisol determination at 8:00 a.m. the following morning. For greater accuracy, give 0.5 mg of Dexamethasone orally every 6 hours for 48 hours. Twenty-four hour urine collections are made for determination of 17-hydroxycorticosteroid excretion. 2. Test to distinguish Cushing’s syndrome due to pituitary ACTH excess from Cushing’s syndrome due to other causes. Give 2 mg of Dexamethasone orally every 6 hours for 48 hours. Twenty-four hour urine collections are made for determination of 17-hydroxycorticosteroid excretion.
CONTRAINDICATIONS
Systemic fungal infections Hypersensitivity to this product
ADVERSE REACTIONS
| Fluid and Electrolyte Disturbances: | |
| Sodium retention Fluid retention Congestive heart failure in susceptible patients | Potassium loss Hypokalemic alkalosis Hypertension |
| Musculoskeletal: | |
| Muscle weakness Steroid myopathy Osteoporosis Aseptic necrosis of femoral and humeral heads | Vertebral compression fractures Loss of muscle mass Pathologic fracture of long bones Tendon rupture |
| Gastrointestinal: | |
| Pancreatitis Abdominal distention Peptic ulcer with possible perforation and hemorrhage | Ulcerative esophagitis Perforation of the small and large bowel, particularly in patients with inflammatory bowel disease |
| Dermatologic: | |
| Impaired wound healing Thin fragile skin Erythema May suppress reactions to skin tests | Petechiae and ecchymoses Increased sweating Other cutaneous reactions, such as allergic dermatitis, urticaria, angioneurotic edema |
| Neurologic: | |
| Convulsions Vertigo Headache Psychic Disturbances | Increased intracranial pressure with papilledema (pseudotumor cerebri) usually after treatment |
| Endocrine: | |
| Menstrual irregularities Development of cushingoid state Manifestations of latent diabetes mellitus Secondary adrenocortical and pituitary unresponsiveness, particularly in times of stress, as in trauma, surgery, or illness | Decreased carbohydrate tolerance Suppression of growth in children Increased requirements for insulin or oral hypoglycemic agents in diabetes Hirsutism |
| Ophthalmic: | |
| Posterior subcapsular cataracts Increased intraocular pressure | Glaucoma Exophthalmos |
| Metabolic: | |
| Negative nitrogen balance due to protein catabolism | |
| Cardiovascular: | |
| Myocardial rupture following recent myocardial infarction (See WARNINGS) | |
| Other: | |
| Hypersensitivity Thromboembolism Weight gain Increased appetite | Nausea Malaise Hiccups |
DESCRIPTION
Each 5 mL (teaspoonful) contains: Dexamethasone, USP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 0.5 mg
Also contains: Benzoic Acid, USP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 0.1% (as preservative) Alcohol (% v/v) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.1%
Inactive Ingredients: artificial raspberry flavor; citric acid; FD&C red no. 40; sucrose; propylene glycol and purified water. It may also contain sodium citrate dihydrate.
Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which are readily absorbed from the gastrointestinal tract.
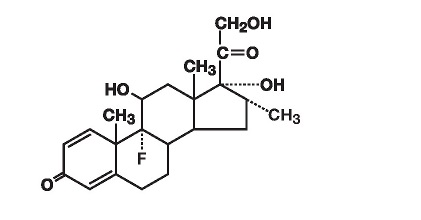
Dexamethasone, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is stable in air. It is practically insoluble in water. The molecular weight is 392.47. It is designated chemically as 9-fluoro-11β,17,21-trihydroxy-16α-methylpregna-1,4-diene-3,20-dione. The molecular formula is C 22 H 29 FO 5 and the structural formula is:
CLINICAL PHARMACOLOGY
Naturally occurring glucocorticoids, (hydrocortisone and cortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs, including dexamethasone, are primarily used for their potent anti-inflammatory effects in disorders of many organ systems.
Glucocorticoids cause profound and varied metabolic effects. In addition, they modify the body’s immune responses to diverse stimuli.
At equipotent anti-inflammatory doses, dexamethasone almost completely lacks the sodium-retaining property of hydrocortisone and closely related derivatives of hydrocortisone.
HOW SUPPLIED
Dexamethasone Elixir, USP 0.5 mg/5 mL is supplied as a clear, red, raspberry-flavored liquid in the following size: 8 fl oz (237 mL) bottle (NDC 64980-509-24)
RECOMMENDED STORAGE
Store at 20˚-25˚C (68˚-77˚F) [See USP Controlled Room Temperature].
KEEP TIGHTLY CLOSED
AVOID FREEZING
Dispense in a tight container as defined in the USP.
Rx Only
Manufactured for:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
Manufactured by:
Lyne Laboratories, Inc. Brockton, MA 02301
Revised : 08/2025
PIR50924-00
Each 5 mL (teaspoonful) contains:
Dexamethaosne, USP....................................................................................................0.5 mg
Also contains:
Benzoic Acid, USP (as preservative)................................................................................0.1%
Alcohol (%v/v)............................................................................................................5.1%
USUAL ADULT DOSAGE: See accompanying package insert.
WARNINGS: KEEP THIS AND ALL DRUGS OUT OF THE REACH OF CHILDREN.
In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Store at 20º – 25ºC (68º – 77ºF) [see USP Controlled Room Temperature]. KEEP TIGHTLY CLOSED AVOID FREEZING
Dispense in a tight container as defined in the USP.
Manufactured for:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
Manufactured by:
Lyne Laboratories, Inc.
Brockton, MA 02301
Revised: 08/2025
LR50924-00