Get your patient on Dextroamphetamine Sulfate extended - Release - Dextroamphetamine Sulfate capsule, Extended Release (Dextroamphetamine Sulfate)
Dextroamphetamine Sulfate extended - Release - Dextroamphetamine Sulfate capsule, Extended Release prescribing information
WARNING: ABUSE, MISUSE, AND ADDICTION
Dextroamphetamine sulfate extended-release capsules have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including dextroamphetamine sulfate extended-release capsules, can result in overdose and death ( see OVERDOSAGE ), and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing dextroamphetamine sulfate extended-release capsules, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout dextroamphetamine sulfate extended-release capsules treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction ( see WARNINGS and DRUG ABUSE AND DEPENDENCE ).
INDICATIONS AND USAGE
Dextroamphetamine sulfate extended-release capsules are indicated in:
Narcolepsy Attention Deficit Disorder with Hyperactivity
As an integral part of a total treatment program that typically includes other measures (psychological, educational, social) for patients (ages 6 years to 16 years) with this syndrome. A diagnosis of Attention Deficit Hyperactivity Disorder (ADHD; DSM-IV) implies the presence of the hyperactive-impulsive or inattentive symptoms that caused impairment and were present before age 7 years. The symptoms must cause clinically significant impairment, e.g., in social, academic, or occupational functioning, and be present in 2 or more settings, e.g., school (or work) and at home. The symptoms must not be better accounted for by another mental disorder. For the Inattentive Type, at least 6 of the following symptoms must have persisted for at least 6 months: lack of attention to details/careless mistakes; lack of sustained attention; poor listener; failure to follow through on tasks; poor organization; avoids tasks requiring sustained mental effort; loses things; easily distracted; forgetful. For the Hyperactive-Impulsive Type, at least 6 of the following symptoms must have persisted for at least 6 months: fidgeting/squirming; leaving seat; inappropriate running/climbing; difficulty with quiet activities; “on the go”; excessive talking; blurting answers; can’t wait turn; intrusive. The Combined Type requires both inattentive and hyperactive-impulsive criteria to be met. Special Diagnostic Considerations
Specific etiology of this syndrome is unknown, and there is no single diagnostic test. Adequate diagnosis requires the use of medical and special psychological, educational, and social resources. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the patient and not solely on the presences of the required number of DSM-IV characteristics. Need for Comprehensive Treatment Program
Dextroamphetamine sulfate extended-release capsules are indicated as an integral part of a total treatment program for ADHD that may include other measures (psychological, educational, social) for patients with this syndrome. Drug treatment may not be indicated for all patients with this syndrome. Stimulants are not intended for use in patients who exhibit symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Appropriate educational placement is essential and psychosocial intervention is often helpful. When remedial measures alone are insufficient, the decision to prescribe stimulant medication will depend upon the physician’s assessment of the chronicity and severity of the patient’s symptoms.
Limitations of Use
The use of dextroamphetamine sulfate extended-release capsules is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage ( see PRECAUTIONS, Pediatric Use ).
DOSAGE AND ADMINISTRATION
Amphetamines should be administered at the lowest effective dosage and dosage should be individually adjusted. Late evening doses should be avoided because of the resulting insomnia.
Prior to treating patients with dextroamphetamine sulfate extended-release capsules, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) ( see WARNINGS ).
- the family history and clinically evaluate patients for motor or verbal tics or Tourette’s syndrome ( see WARNINGS ).
Narcolepsy
Usual dose is 5 to 60 mg per day in divided doses, depending on the individual patient response.
Narcolepsy seldom occurs in children under 12 years of age; however, when it does, dextroamphetamine sulfate extended-release capsules may be used. The suggested initial dose for patients aged 6 to 12 is 5 mg daily; daily dose may be raised in increments of 5 mg at weekly intervals until an optimal response is obtained. In patients 12 years of age and older, start with 10 mg daily; daily dosage may be raised in increments of 10 mg at weekly intervals until an optimal response is obtained. If bothersome adverse reactions appear (e.g., insomnia or anorexia), dosage should be reduced. Dextroamphetamine sulfate extended-release capsules may be used for once-a-day dosage wherever appropriate.
Attention Deficit Disorder with Hyperactivity
The dextroamphetamine sulfate extended-release capsule formulation is not recommended for pediatric patients younger than 6 years of age.
In pediatric patients 6 years of age and older, start with 5 mg once or twice daily; daily dosage may be raised in increments of 5 mg at weekly intervals until optimal response is obtained. Only in rare cases will it be necessary to exceed a total of 40 mg per day. Dextroamphetamine sulfate extended-release capsules may be used for once-a-day dosage wherever appropriate.
CONTRAINDICATIONS
In patients known to be hypersensitive to amphetamine, or other components of dextroamphetamine sulfate extended-release capsules. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products ( see ADVERSE REACTIONS ).
Patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis ( see WARNINGS and Drug Interactions ).
ADVERSE REACTIONS
Cardiovascular
Palpitations, tachycardia, elevation of blood pressure. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use. Central Nervous System
Psychotic episodes at recommended doses (rare), overstimulation, restlessness, dizziness, insomnia, euphoria, dyskinesia, dysphoria, tremor, headache, exacerbation of motor and verbal tics and Tourette’s syndrome. Gastrointestinal
Dryness of the mouth, unpleasant taste, diarrhea, constipation, intestinal ischemia, and other gastrointestinal disturbances. Anorexia and weight loss may occur as undesirable effects. Allergic
Urticaria.
Endocrine
Impotence, changes in libido, frequent or prolonged erections.
Musculoskeletal
Rhabdomyolysis.
Skin and Subcutaneous Tissue Disorders
Alopecia.
To report SUSPECTED ADVERSE REACTIONS, contact Mallinckrodt at 1-800-778-7898 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Acidifying Agents
Lower blood levels and efficacy of amphetamines. Increase dose based on clinical response. Examples of acidifying agents include gastrointestinal acidifying agents (e.g., guanethidine, reserpine, glutamic acid HCl, ascorbic acid) and urinary acidifying agents (e.g., ammonium chloride, sodium acid phosphate, methenamine salts).
Adrenergic Blockers
Adrenergic blockers are inhibited by amphetamines.
Alkalinizing Agents
Increase blood levels and potentiate the action of amphetamine. Co-administration of dextroamphetamine sulfate extended-release capsules and gastrointestinal alkalinizing agents should be avoided. Examples of alkalinizing agents include gastrointestinal alkalinizing agents (e.g., sodium bicarbonate) and urinary alkalinizing agents (e.g., acetazolamide, some thiazides).
Tricyclic Antidepressants
May enhance the activity of tricyclic or sympathomimetic agents causing striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated. Monitor frequently and adjust or use alternative therapy based on clinical response. Examples of tricyclic antidepressants include desipramine, Protriptyline.
CYP2D6 Inhibitors
The concomitant use of dextroamphetamine sulfate extended-release capsules and CYP2D6 inhibitors may increase the exposure of dextroamphetamine sulfate extended-release capsules compared to the use of the drug alone and increase the risk of serotonin syndrome. Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during dextroamphetamine sulfate extended-release capsules initiation and after a dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine sulfate extended-release capsules and the CYP2D6 inhibitor ( see WARNINGS , OVERDOSAGE ). Examples of CYP2D6 Inhibitors include paroxetine and fluoxetine (also serotonergic drugs), quinidine, ritonavir.
Serotonergic Drugs
The concomitant use of dextroamphetamine sulfate extended-release capsules and serotonergic drugs increases the risk of serotonin syndrome. Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during dextroamphetamine sulfate extended-release capsules initiation or dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine sulfate extended-release capsules and the concomitant serotonergic drug(s) ( see WARNINGS and PRECAUTIONS ). Examples of serotonergic drugs include selective serotonin reuptake inhibitors (SSRI), serotonin norepinephrine reuptake inhibitors (SNRI), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John’s Wort.
MAO Inhibitors
Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure. Do not administer dextroamphetamine sulfate extended-release capsules concomitantly or within 14 days after discontinuing MAOI ( see CONTRAINDICATIONS and WARNINGS ). Examples of MAOIs include selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue.
Proton Pump Inhibitors
Time to maximum concentration (T max ) of amphetamine is decreased compared to when administered alone. Monitor patients for changes in clinical effect and adjust therapy based on clinical response. An example of a proton pump inhibitor is omeprazole.
Antihistamines
Amphetamines may counteract the sedative effect of antihistamines.
Antihypertensives
Amphetamines may antagonize the hypotensive effects of antihypertensives.
Chlorpromazine
Chlorpromazine blocks dopamine and norepinephrine reuptake, thus inhibiting the central stimulant effects of amphetamines, and can be used to treat amphetamine poisoning.
Ethosuximide
Amphetamines may delay intestinal absorption of ethosuximide.
Haloperidol
Haloperidol blocks dopamine and norepinephrine reuptake, thus inhibiting the central stimulant effects of amphetamines.
Lithium Carbonate
The stimulatory effects of amphetamines may be inhibited by lithium carbonate.
Meperidine
Amphetamines potentiate the analgesic effect of meperidine.
Methenamine Therapy
Urinary excretion of amphetamines is increased, and efficacy is reduced, by acidifying agents used in methenamine therapy.
Norepinephrine
Amphetamines enhance the adrenergic effect of norepinephrine.
Phenobarbital
Amphetamines may delay intestinal absorption of phenobarbital; co-administration of phenobarbital may produce a synergistic anticonvulsant action.
Phenytoin
Amphetamines may delay intestinal absorption of phenytoin; co-administration of phenytoin may produce a synergistic anticonvulsant action.
Propoxyphene
In cases of propoxyphene overdosage, amphetamine CNS stimulation is potentiated and fatal convulsions can occur.
Veratrum Alkaloids
Amphetamines inhibit the hypotensive effect of veratrum alkaloids.
Drug/Laboratory Test Interactions
Amphetamines can cause a significant elevation in plasma corticosteroid levels. This increase is greatest in the evening.
Amphetamines may interfere with urinary steroid determinations.
Carcinogenesis/Mutagenesis
Mutagenicity studies and long-term studies in animals to determine the carcinogenic potential of dextroamphetamine sulfate extended-release capsules have not been performed.
DESCRIPTION
Dextroamphetamine sulfate extended-release capsule is the dextro isomer of the compound d,l -amphetamine sulfate, a sympathomimetic amine of the amphetamine group. Chemically, dextroamphetamine is d -alpha-methylphenethylamine, and is present in all forms of dextroamphetamine sulfate extended-release capsules as the neutral sulfate.
Structural formula:
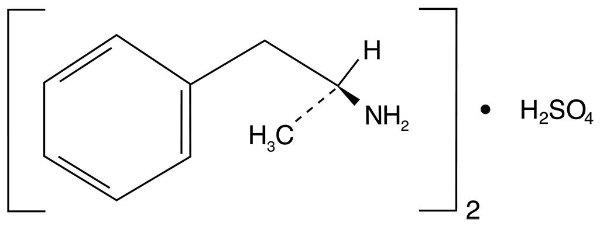
Dextroamphetamine Sulfate Extended-Release Capsules
Each dextroamphetamine sulfate extended-release capsule is so prepared that an initial dose is released promptly and the remaining medication is released gradually over a prolonged period.
Each capsule, with white opaque cap and white opaque body, contains dextroamphetamine sulfate USP. The 5 mg capsule is imprinted with an  ® on the cap and is imprinted 8960 5 mg on the body in black. The 10 mg capsule is imprinted with an
® on the cap and is imprinted 8960 5 mg on the body in black. The 10 mg capsule is imprinted with an  ® on the cap and is imprinted 8961 10 mg on the body in blue. The 15 mg capsule is imprinted with an
® on the cap and is imprinted 8961 10 mg on the body in blue. The 15 mg capsule is imprinted with an  ® on the cap and is imprinted 8962 15 mg on the body in pink. Inactive ingredients consist of sugar spheres, titanium dioxide, gelatin, shellac glaze-45%, SD-45 alcohol, iron oxide black, propylene glycol, FD&C Blue #2/Indigo Carmine Lake, FD&C Red #40/Allura Red AC Lake, FD&C Blue #1/Brilliant Blue FCF Lake, D&C Yellow #10 Lake, SD3A alcohol, shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, strong ammonia solution, FD&C Blue #2 Aluminum Lake, D&C Red #7 Calcium Lake, hydroxypropyl methylcellulose/hypromellose, macrogol/polyethylene glycol, purified water, ethylcellulose, ammonium hydroxide 28%, medium chain triglycerides, oleic acid.
® on the cap and is imprinted 8962 15 mg on the body in pink. Inactive ingredients consist of sugar spheres, titanium dioxide, gelatin, shellac glaze-45%, SD-45 alcohol, iron oxide black, propylene glycol, FD&C Blue #2/Indigo Carmine Lake, FD&C Red #40/Allura Red AC Lake, FD&C Blue #1/Brilliant Blue FCF Lake, D&C Yellow #10 Lake, SD3A alcohol, shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, strong ammonia solution, FD&C Blue #2 Aluminum Lake, D&C Red #7 Calcium Lake, hydroxypropyl methylcellulose/hypromellose, macrogol/polyethylene glycol, purified water, ethylcellulose, ammonium hydroxide 28%, medium chain triglycerides, oleic acid.
CLINICAL PHARMACOLOGY
Amphetamines are noncatecholamine, sympathomimetic amines with CNS stimulant activity. Peripheral actions include elevations of systolic and diastolic blood pressures and weak bronchodilator and respiratory stimulant action. There is neither specific evidence that clearly establishes the mechanism whereby amphetamines produce mental and behavioral effects in children, nor conclusive evidence regarding how these effects relate to the condition of the central nervous system. Dextroamphetamine sulfate extended-release capsules are formulated to release the active drug substance in vivo in a more gradual fashion than the standard formulation, as demonstrated by blood levels. The formulation has not been shown superior in effectiveness over the same dosage of the standard, noncontrolled-release formulations given in divided doses.
Pharmacokinetics
The pharmacokinetics of the tablet and sustained-release capsule were compared in 12 healthy subjects. The extent of bioavailability of the sustained-release capsule was similar compared to the immediate-release tablet. Following administration of three 5 mg tablets, average maximal dextroamphetamine plasma concentrations (C max ) of 36.6 ng/mL were achieved at approximately 3 hours.
Following administration of one 15 mg sustained-release capsule, maximal dextroamphetamine plasma concentrations were obtained approximately 8 hours after dosing. The average C max was 23.5 ng/mL. The average plasma T 1/2 was similar for both the tablet and sustained-release capsule and was approximately 12 hours. In 12 healthy subjects, the rate and extent of dextroamphetamine absorption were similar following administration of the sustained-release capsule formulation in the fed (58 g to 75 g fat) and fasted state.
HOW SUPPLIED
Dextroamphetamine Sulfate Extended-Release Capsules
Each capsule, with white opaque cap and white opaque body, contains dextroamphetamine sulfate.
The 5 mg capsule is imprinted with an  ® on the cap and is imprinted 8960 5 mg on the body in black.
® on the cap and is imprinted 8960 5 mg on the body in black.
The 10 mg capsule is imprinted with an  ® on the cap and is imprinted 8961 10 mg on the body in blue.
® on the cap and is imprinted 8961 10 mg on the body in blue.
The 15 mg capsule is imprinted with an  ® on the cap and is imprinted 8962 15 mg on the body in pink.
® on the cap and is imprinted 8962 15 mg on the body in pink.
5 mg Bottles of 100……………….NDC 0406-8960-01 10 mg Bottles of 100……………….NDC 0406-8961-01 15 mg Bottles of 100……………….NDC 0406-8962-01
Store at controlled room temperature between 20° to 25°C (68° to 77°F) [see USP].
Dispense in a tight, light-resistant container with a child-resistant closure.
Mallinckrodt, the “M” brand mark, the Mallinckrodt Pharmaceuticals logo and  are trademarks of a Mallinckrodt company.
are trademarks of a Mallinckrodt company.
© 2025 Mallinckrodt.
SpecGx LLC Webster Groves, MO 63119 USA
Rev 09/2025
Mallinckrodt™ Pharmaceuticals