Get your patient on Dextrose And Sodium Chloride - Dextrose And Sodium Chloride injection, Solution (Dextrose And Sodium Chloride)
Dextrose And Sodium Chloride - Dextrose And Sodium Chloride injection, Solution prescribing information
INDICATIONS AND USAGE
These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes, calories and water for hydration.
DOSAGE AND ADMINISTRATION
These solutions are for intravenous use only.
Dosage is to be directed by a physician and is dependent upon age, weight, clinical condition of the patient and laboratory determinations. Frequent laboratory determinations and clinical evaluation are essential to monitor changes in blood glucose and electrolyte concentrations, and fluid and electrolyte balance during prolonged parenteral therapy.
When a hypertonic solution is to be administered peripherally, it should be slowly infused through a small bore needle, placed well within the lumen of a large vein to minimize venous irritation. Carefully avoid infiltration.
In the average adult, daily requirements of sodium and chloride are met by the infusion of one liter of fluid containing 0.9% sodium chloride (154 mEq each of sodium and chloride).
Fluid administration should be based on calculated maintenance or replacement fluid requirements for each patient.
Some additives may be incompatible. Consult with pharmacist. When introducing additives, use aseptic techniques. Mix thoroughly. Do not store.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Pediatric Use
There is no specific pediatric dose. The dose is dependent on weight, clinical condition, and laboratory results. See WARNINGS and PRECAUTIONS .
CONTRAINDICATIONS
These solutions are contraindicated where the administration of sodium or chloride could be clinically detrimental.
Solutions containing dextrose may be contraindicated in patients with hypersensitivity to corn products.
ADVERSE REACTIONS
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
Too rapid infusion of hypertonic solutions may cause local pain and venous irritation. Rate of administration should be adjusted according to tolerance. Use of the largest peripheral vein and a small bore needle is recommended. (See DOSAGE AND ADMINISTRATION .)
Symptoms may result from an excess or deficit of one or more of the ions present in the solution; therefore, frequent monitoring of electrolyte levels is essential.
Hypernatremia may be associated with edema and exacerbation of congestive heart failure due to the retention of water, resulting in an expanded extracellular fluid volume. If infused in large amounts, chloride ions may cause a loss of bicarbonate ions, resulting in an acidifying effect.
The physician should also be alert to the possibility of adverse reactions to drug additives diluted and administered from the plastic container. Prescribing information for drug additives to be administered in this manner should be consulted.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary.
DESCRIPTION
(See chart below for quantitative information.)
Dextrose and Sodium Chloride Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration.
The formulas of the active ingredients are:
| Ingredients | Molecular Formula | Molecular Weight |
|---|---|---|
| Sodium Chloride USP | NaCl | 58.44 |
| Hydrous Dextrose USP | 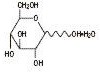 | 198.17 |
| Composition – Each 100 mL contains: | Concentration of Electrolytes (mEq/liter) | ||||||
|---|---|---|---|---|---|---|---|
| Hydrous Dextrose USP | Sodium Chloride USP | Calories per liter | Calculated Osmolarity mOsmol/liter | pH | |||
| Solution | Sodium | Chloride | |||||
| Water for Injection USP qs | |||||||
| 3.3% Dextrose and 0.30% Sodium Chloride Injection USP | 3.3 g | 0.3 g | 51 | 51 | 110 | 270 | 4.5 (3.5–6.5) |
| 5% Dextrose and 0.9% Sodium Chloride Injection USP | 5 g | 0.9 g | 154 | 154 | 170 | 560 | 4.4 (3.5–6.5) |
| 5% Dextrose and 0.45% Sodium Chloride Injection USP | 5 g | 0.45 g | 77 | 77 | 170 | 405 | 4.4 (3.5–6.5) |
| 5% Dextrose and 0.33% Sodium Chloride Injection USP | 5 g | 0.33 g | 56 | 56 | 170 | 365 | 4.4 (3.5–6.5) |
| 5% Dextrose and 0.20% Sodium Chloride Injection USP | 5 g | 0.2 g | 34 | 34 | 170 | 320 | 4.4 (3.5–6.5) |
| 10% Dextrose and 0.45% Sodium Chloride Injection USP | 10 g | 0.45 g | 77 | 77 | 340 | 660 | 4.3 (3.5–6.5) |
| 10% Dextrose and 0.20% Sodium Chloride Injection USP | 10 g | 0.2 g | 34 | 34 | 340 | 575 | 4.3 (3.5–6.5) |
Not made with natural rubber latex, PVC or DEHP.
The plastic container is made from a multilayered film specifically developed for parenteral drugs. It contains no plasticizers and exhibits virtually no leachables. The solution contact layer is a rubberized copolymer of ethylene and propylene. The container is nontoxic and biologically inert. The container-solution unit is a closed system and is not dependent upon entry of external air during administration. The container is overwrapped to provide protection from the physical environment and to provide an additional moisture barrier when necessary.
Addition of medication should be accomplished using complete aseptic technique.
The closure system has two ports; the one for the administration set has a tamper evident plastic protector and the other is a medication addition site. Refer to the Directions for Use of the container.
CLINICAL PHARMACOLOGY
Dextrose and Sodium Chloride Injections USP provide electrolytes and calories and are a source of water for hydration. All are capable of inducing diuresis depending on the clinical condition of the patient.
Sodium, the major cation of the extracellular fluid, functions primarily in the control of water distribution, fluid balance, and osmotic pressure of body fluids. Sodium is also associated with chloride and bicarbonate in the regulation of the acid-base equilibrium of body fluid.
Chloride, the major extracellular anion, closely follows the metabolism of sodium, and changes in the acid-base balance of the body are reflected by changes in the chloride concentration.
Dextrose provides a source of calories. Dextrose is readily metabolized, may decrease losses of body protein and nitrogen, promotes glycogen deposition and decreases or prevents ketosis if sufficient doses are provided.
HOW SUPPLIED
Dextrose and Sodium Chloride Injections USP are supplied sterile and nonpyrogenic in EXCEL ® Containers. The 1000 mL containers are packaged 12 per case; the 500 mL and 250 mL containers are packaged 24 per case.
| Canada DIN | NDC | REF | Size |
|---|---|---|---|
| 3.3% Dextrose and 0.30% Sodium Chloride Injection USP | |||
| 01927981 | 0264-7608-00 | L6080-00 | 1000 mL |
| 0264-7608-10 | L6081-00 | 500 mL | |
| 5% Dextrose and 0.9% Sodium Chloride Injection USP | |||
| 01924435 | 0264-7610-00 | L6100 | 1000 mL |
| 0264-7610-10 | L6101 | 500 mL | |
| 5% Dextrose and 0.45% Sodium Chloride Injection USP | |||
| 01927531 | 0264-7612-00 | L6120 | 1000 mL |
| 0264-7612-10 | L6121 | 500 mL | |
| 0264-7612-20 | L6122 | 250 mL | |
| 5% Dextrose and 0.33% Sodium Chloride Injection USP | |||
| 0264-7614-00 | L6140 | 1000 mL | |
| 0264-7614-10 | L6141 | 500 mL | |
| 5% Dextrose and 0.20% Sodium Chloride Injection USP | |||
| 01927558 | 0264-7616-00 | L6160 | 1000 mL |
| 0264-7616-10 | L6161 | 500 mL | |
| 0264-7616-20 | L6162 | 250 mL | |
| 10% Dextrose and 0.45% Sodium Chloride Injection USP | |||
| 0264-7622-00 | L6220 | 1000 mL | |
| 10% Dextrose and 0.20% Sodium Chloride Injection USP | |||
| 0264-7623-20 | L6232 | 250 mL | |
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product.