Get your patient on Diazepam - Diazepam Oral solution (Diazepam Oral)
Diazepam - Diazepam Oral solution prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death.
Reserve concomitant prescribing of these drugs in patients for whom alternative treatment options are inadequate.
Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS AND PRECAUTIONS ).
- The use of benzodiazepines, including diazepam, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing diazepam and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS ).
- The continued use of benzodiazepines, including diazepam, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of diazepam after continued use may precipitate acute withdrawal reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue diazepam or reduce the dosage (see DOSAGE AND ADMINISTRATION and WARNINGS ).
INDICATIONS
Diazepam is indicated for the management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic.
In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis.
Diazepam is a useful adjunct for the relief of skeletal muscle spasm due to reflex spasm to local pathology (such as inflammation of the muscles or joints, or secondary to trauma), spasticity caused by upper motor neuron disorders (such as cerebral palsy and paraplegia), athetosis, and stiff-man syndrome.
Oral diazepam may be used adjunctively in convulsive disorders, although it has not proved useful as the sole therapy.
The effectiveness of diazepam in long-term use, that is, more than 4 months, has not been assessed by systematic clinical studies. The physician should periodically reassess the usefulness of the drug for the individual patient.
DOSAGE AND ADMINISTRATION
Dosage should be individualized for maximum beneficial effect. While the usual daily dosages given below will meet the needs of most patients, there will be some who may require higher doses. In such cases dosage should be increased cautiously to avoid adverse effects.
Adults: | Usual Daily Dosage: |
Management of Anxiety Disorders and Relief of Symptoms of Anxiety. | Depending upon severity of symptoms - 2 mg to 10 mg, 2 to 4 times daily. |
Symptomatic Relief in Acute Alcohol Withdrawal. | 10 mg, 3 or 4 times during the first 24 hours, reducing to 5 mg, 3 or 4 times daily as needed. |
Adjunctively for Relief of Skeletal Muscle Spasm. | 2 mg to 10 mg, 3 or 4 times daily. |
Adjunctively in Convulsive Disorders. | 2 mg to 10 mg, 2 to 4 times daily. |
Geriatric Patients, or in the presence of debilitating disease. | 2 mg to 2.5 mg, 1 or 2 times daily initially; increase gradually as needed and tolerated. |
Pediatric Patients: | |
Because of varied responses to CNS-acting drugs, initiate therapy with lowest dose and increase as required. Not for use in pediatric patients under 6 months. | 1 mg to 2.5 mg, 3 or 4 times daily initially; increase gradually as needed and tolerated. |
Discontinuation or Dosage Reduction of Diazepam
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue diazepam or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly (see WARNINGS: Dependence and Withdrawal Reactions and DRUG ABUSE AND DEPENDENCE: Dependence ).
Proper Use of Diazepam Oral Solution (Concentrate)
Diazepam Oral Solution (Concentrate) is a concentrated oral solution as compared to standard oral liquid medications. It is recommended that Diazepam Oral Solution (Concentrate) be mixed with liquid or semi-solid food such as water, juices, soda or soda-like beverages, applesauce and puddings.
Use only with a calibrated dropper. Draw into the dropper the amount prescribed for a single dose. Then squeeze the dropper contents into a liquid or semi-solid food. Stir the liquid or food gently for a few seconds. The Diazepam Oral Solution (Concentrate) formulation blends quickly and completely. The entire amount of the mixture, of drug and liquid or drug and food, should be consumed immediately. Do not store for future use.
CONTRAINDICATIONS
Diazepam is contraindicated in patients with a known hypersensitivity to this drug and, because of lack of sufficient clinical experience, in pediatric patients under 6 months of age. Diazepam is also contraindicated in patients with myasthenia gravis, severe respiratory insufficiency, severe hepatic insufficiency, and sleep apnea syndrome. It may be used in patients with open-angle glaucoma who are receiving appropriate therapy, but is contraindicated in acute narrow-angle glaucoma.
ADVERSE REACTIONS
Side effects most commonly reported were drowsiness, fatigue, muscle weakness and ataxia. The following have also been reported:
Central Nervous System:
confusion, depression, dysarthria, headache, slurred speech, tremor, vertigo
Gastrointestinal System:
constipation, nausea, gastrointestinal disturbances
Special Senses:
blurred vision, diplopia, dizziness
Cardiovascular System:
hypotension
Psychiatric and Paradoxical Reactions:
stimulation, restlessness, acute hyperexcited states, anxiety, agitation, aggressiveness, irritability, rage, hallucinations, psychoses, delusions, increased muscle spasticity, insomnia, sleep disturbances, and nightmares. Inappropriate behavior and other adverse behavioral effects have been reported when using benzodiazepines. Should these occur, use of the drug should be discontinued. They are more likely to occur in children and in the elderly.
Urogenital System:
incontinence, changes in libido, urinary retention
Skin and Appendages:
skin reactions
Laboratories:
elevated transaminases and alkaline phosphatase
Other:
changes in salivation, including dry mouth, hypersalivation
Antegrade amnesia may occur using therapeutic dosages, the risk increasing at higher dosages. Amnestic effects may be associated with inappropriate behavior.
Minor changes in EEG patterns, usually low-voltage fast activity, have been observed in patients during and after diazepam therapy and are of no known significance.
Because of isolated reports of neutropenia and jaundice, periodic blood counts and liver function tests are advisable during long-term therapy.
Postmarketing Experience
Injury, Poisoning and Procedural Complications
There have been reports of falls and fractures in benzodiazepine users. The risk is increased in those taking concomitant sedatives (including alcohol), and in the elderly.
DESCRIPTION
Each 5 mL of Diazepam Oral Solution contains:
diazepam USP ....................................................... 5 mg
Each mL of Diazepam Oral Solution (Concentrate) contains:
diazepam USP ....................................................... 5 mg
alcohol .................................................................. 19%
Inactive Ingredients:
Diazepam Oral Solution contains polyethylene glycol, propylene glycol, sorbitol solution, sodium citrate dihydrate, bitterness modifier flavor, anhydrous citric acid, peppermint flavor, FD&C Red No. 40, D&C Yellow No. 10, and purified water.
Diazepam Oral Solution (Concentrate) contains polyethylene glycol, propylene glycol, alcohol (19%), D&C Yellow No. 10, succinic acid, and purified water.
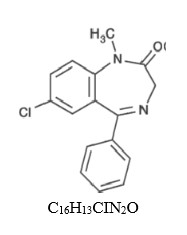
Diazepam is a benzodiazepine derivative. Chemically, diazepam is 7-Chloro-1,3-dihydro-1-methyl-5-phenyl-2 H -1,4-benzodiazepin-2-one. It is a white to practically white powder, insoluble in water and has a molecular weight of 284.75. Its structural formula is as follows:
CLINICAL PHARMACOLOGY
Diazepam is a benzodiazepine that exerts anxiolytic, sedative, muscle-relaxant, anticonvulsant and amnestic effects. Most of these effects are thought to result from a facilitation of the action of gamma aminobutyric acid (GABA), an inhibitory neurotransmitter in the central nervous system.
Pharmacokinetics
Absorption:
After oral administration of diazepam, > 90% of diazepam is absorbed and the average time to achieve peak plasma concentrations is 1 to 1.5 hours with a range of 0.25 to 2.5 hours. Absorption is delayed and decreased when administered with a moderate fat meal. In the presence of food, mean lag times are approximately 45 minutes as compared with 15 minutes when fasting. There is also an increase in the average time to achieve peak concentrations to about 2.5 hours in the presence of food as compared with 1.25 hours when fasting. This results in an average decrease in C max of 20% in addition to a 27% decrease in AUC (range 15% to 50%) when administered with food.
Distribution:
Diazepam and its metabolites are highly bound to plasma proteins (diazepam 98%). Diazepam and its metabolites cross the blood-brain and placental barriers and are also found in breast milk in concentrations approximately one tenth of those in maternal plasma (days 3 to 9 post-partum). In young healthy males, the volume of distribution at steady-state is 0.8 to 1.0 L/kg. The decline in the plasma concentration-time profile after oral administration is biphasic. The initial distribution phase has a half-life of approximately 1 hour, although it may range up to > 3 hours.
Metabolism:
Diazepam is N-demethylated by CYP3A4 and 2C19 to the active metabolite N-desmethyldiazepam, and is hydroxylated by CYP3A4 to the active metabolite temazepam. N-desmethyldiazepam and temazepam are both further metabolized to oxazepam. Temazepam and oxazepam are largely eliminated by glucuronidation.
Elimination:
The initial distribution phase is followed by a prolonged terminal elimination phase (half-life up to 48 hours).
The terminal elimination half-life of the active metabolite N-desmethyldiazepam is up to 100 hours. Diazepam and its metabolites are excreted mainly in the urine, predominantly as their glucuronide conjugates. The clearance of diazepam is 20 to 30 mL/min in young adults. Diazepam accumulates upon multiple dosing and there is some evidence that the terminal elimination half-life is slightly prolonged.
Pharmacokinetics in Special Populations
Children:
In children 3 to 8 years old the mean half-life of diazepam has been reported to be 18 hours.
Newborns:
In full term infants, elimination half-lives around 30 hours have been reported, with a longer average half-life of 54 hours reported in premature infants of 28 to 34 weeks gestational age and 8 to 81 days post-partum. In both premature and full term infants the active metabolite desmethyldiazepam shows evidence of continued accumulation compared to children. Longer half-lives in infants may be due to incomplete maturation of metabolic pathways.
Geriatric:
Elimination half-life increases by approximately 1 hour for each year of age beginning with a half-life of 20 hours at 20 years of age. This appears to be due to an increase in volume of distribution with age and a decrease in clearance. Consequently, the elderly may have lower peak concentrations, and on multiple dosing higher trough concentrations. It will also take longer to reach steady-state. Conflicting information has been published on changes of plasma protein binding in the elderly. Reported changes in free drug may be due to significant decreases in plasma proteins due to causes other than simply aging.
Hepatic Insufficiency:
In mild and moderate cirrhosis, average half-life is increased. The average increase has been variously reported from 2-fold to 5-fold, with individual half-lives over 500 hours reported. There is also an increase in volume of distribution, and average clearance decreases by almost half. Mean half-life is also prolonged with hepatic fibrosis to 90 hours (range 66 to 104 hours), with chronic active hepatitis to 60 hours (range 26 to 76 hours), and with acute viral hepatitis to 74 hours (range 49 to 129 hours). In chronic active hepatitis, clearance is decreased by almost half.
HOW SUPPLIED
Diazepam Oral Solution, 5 mg per 5 mL (Orange-colored, peppermint flavored solution) is supplied as follows:
NDC 62135-767-47: Bottle of 473 mL. NDC 62135-767-45: Unit Dose Cups of 5 mL NDC 62135-767-24: Tray of 20 x Unit Dose Cups of 5 mL
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Dispense in tight, light-resistant container as defined in the USP/NF.
Diazepam Oral Solution (Concentrate), 5 mg per mL (Clear, yellow solution) is supplied as follows:
NDC 62135-768-43: Bottle of 30 mL
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Discard opened bottle after 90 days.
PROTECT FROM LIGHT.
Manufactured for: Chartwell RX, LLC. Congers, NY 10920
L71008
Rev. 07/2024
Print Medication Guides at: www.chartwellpharma.com/medguides .