Get your patient on Diclofenac Sodium - Diclofenac Sodium tablet, Extended Release (Diclofenac Sodium)
Diclofenac Sodium - Diclofenac Sodium tablet, Extended Release prescribing information
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
Cardiovascular Thrombotic Events • Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( see WARNINGS ). • Diclofenac Sodium extended-release tablets, USP are contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( see CONTRAINDICATIONS , WARNINGS ). Gastrointestinal Bleeding, Ulceration, and Perforation • NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events, including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( see WARNINGS ).
INDICATIONS & USAGE
Carefully consider the potential benefits and risks of diclofenac sodium extended release tablets, USP and other treatment options before deciding to use diclofenac sodium extended-release tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals ( see WARNINGS; Gastrointestinal Bleeding, Ulceration, and Perforation ). Diclofenac sodium extended-release tablets are indicated: • for relief of the signs and symptoms of osteoarthritis • for relief of the signs and symptoms of rheumatoid arthritis
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of diclofenac sodium extended-release tablets, USP and other treatment options before deciding to use diclofenac sodium extended release tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals ( see WARNINGS ). After observing the response to initial therapy with diclofenac sodium extended-release tablets, the dose and frequency should be adjusted to suit an individual patient’s needs. For the relief of osteoarthritis, the recommended dosage is 100 mg daily. For the relief of rheumatoid arthritis, the recommended dosage is 100 mg daily. In the rare patient where diclofenac sodium extended-release tablets 100 mg/day are unsatisfactory, the dose may be increased to 100 mg twice a day if the benefits outweigh the clinical risks of increased side effects. Different formulations of diclofenac (diclofenac sodium enteric-coated tablets; diclofenac sodium extended-release tablets, USP, diclofenac potassium immediate-release tablets) are not necessarily bioequivalent even if the milligram strength is the same.
CONTRAINDICATIONS
Diclofenac sodium extended-release tablets, USP are contraindicated in the following patients: • Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product ( see WARNINGS; Anaphylactic Reactions, Serious Skin Reactions ). • History of asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactice reactions to NSAIDs have been reported in such patients ( see WARNINGS; Anaphylactic Reactions , PRECAUTIONS; Exacerbation of Asthma Related to Aspirin Sensitivity ) . • In the setting of coronary artery bypass graft (CABG) surgery ( see WARNINGS; Cardiovascular Thrombotic Events ).
ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling: • Cardiovascular Thrombotic Events (see WARNINGS ) • GI Bleeding, Ulceration and Perforation (see WARNINGS ) • Hepatotoxicity (see WARNINGS ) • Hypertension (see WARNINGS ) • Heart Failure and Edema (see WARNINGS ) • Renal Toxicity and Hyperkalemia (see WARNINGS ) • Anaphylactic Reactions (see WARNINGS ) • Serious Skin Reactions (see WARNINGS ) • Hematologic Toxicity (see WARNINGS )
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. In patients taking diclofenac sodium extended-release tablets, USP or other NSAIDs, the most frequently reported adverse experiences occurring in approximately 1% to 10% of patients are: Gastrointestinal experiences including: abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gross bleeding/perforation, heartburn, nausea, GI ulcers (gastric/duodenal) and vomiting. Abnormal renal function, anemia, dizziness, edema, elevated liver enzymes, headaches, increased bleeding time, pruritus, rashes and tinnitus. Additional adverse experiences reported occasionally include: Body as a Whole: fever, infection, sepsis Cardiovascular System: congestive heart failure, hypertension, tachycardia, syncope Digestive System: dry mouth, esophagitis, gastric/peptic ulcers, gastritis, gastrointestinal bleeding, glossitis, hematemesis, hepatitis, jaundice Hemic and Lymphatic System: ecchymosis, eosinophilia, leukopenia, melena, purpura, rectal bleeding, stomatitis, thrombocytopenia Metabolic and Nutritional: weight changes Nervous System: anxiety, asthenia, confusion, depression, dream abnormalities, drowsiness, insomnia, malaise, nervousness, paresthesia, somnolence, tremors, vertigo Respiratory System: asthma, dyspnea Skin and Appendages: alopecia, photosensitivity, sweating increased Special Senses: blurred vision Urogenital System: cystitis, dysuria, hematuria, interstitial nephritis, oliguria/polyuria, proteinuria, renal failure. Other adverse reactions, which occur rarely are: Body as a Whole: anaphylactic reactions, appetite changes, death Cardiovascular System: arrhythmia, hypotension, myocardial infarction, palpitations, vasculitis Digestive System: colitis, eructation, fulminant hepatitis with and without jaundice, liver failure, liver necrosis, pancreatitis Hemic and Lymphatic System: agranulocytosis, hemolytic anemia, aplastic anemia, lymphadenopathy, pancytopenia Metabolic and Nutritional: hyperglycemia Nervous System: convulsions, coma, hallucinations, meningitis Respiratory System: respiratory depression, pneumonia Skin and Appendages: angioedema, toxic epidermal necrolysis, erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome, fixed drug eruption (FDE), urticaria Special Senses: conjunctivitis, hearing impairment. To report SUSPECTED ADVERSE EVENTS, contact Edenbridge Pharmaceuticals, LLC at 877-381-3336 or FDA at 1-800-FDA- 1088 or http://www.fda.gov/medwatch for voluntary reporting of adverse reactions.
Drug Interactions
See Table 2 for clinically significant drug interactions with diclofenac.
Table 2. Clinically Significant Drug Interactions with Diclofenac
| Drugs That Interfere with Hemostasis | |
| Clinical Impact: |
|
| Intervention: | Monitor patients with concomitant use of diclofenac sodium extended-release tablets with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding (see WARNINGS; Hematological Toxicity ) . |
| Aspirin | |
| Clinical Impact: | Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone (see Warnings; Gastrointestinal Bleeding, Ulceration, and Perforation ) . |
| Intervention: | Concomitant use of diclofenac sodium extended-release tablets and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding (see WARNINGS; Hematological Toxicity ) . Diclofenac sodium extended-release tablets are not a substitute for low dose aspirin for cardiovascular protection. |
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta- Blockers | |
| Clinical Impact: | • NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). • In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, coadministration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. |
| Intervention: | • During concomitant use of diclofenac sodium extended-release tablets and ACE-inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. • During concomitant use of diclofenac sodium extended-release tablets and ACE-inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function (see WARNINGS; Renal Toxicity and Hyperkalemia ) . • When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. |
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAIDinhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of diclofenac sodium extended-release tablets with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy, including antihypertensive effects (see WARNINGS; Renal Toxicity and Hyperkalemia ) . |
| Digoxin | |
| Clinical Impact: | The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
| Intervention: | During concomitant use of diclofenac sodium extended-release tablets and digoxin, monitor serum digoxin levels. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of diclofenac sodium extended-release tablets and lithium, monitor patients for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
| Intervention: | During concomitant use of diclofenac sodium extended-release tablets and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of diclofenac sodium extended-release tablets and cyclosporine may increase cyclosporine’s nephrotoxicity. |
| Intervention: | During concomitant use of diclofenac sodium extended-release tablets and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of diclofenac with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy (see WARNINGS; Gastrointestinal Bleeding, Ulceration, and Perforation ) . |
| Intervention: | The concomitant use of diclofenac with other NSAIDs or salicylates is not recommended. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of diclofenac sodium extended-release tablets and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of diclofenac sodium extended-release tablets and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of 2 days before, the day of, and 2 days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least 5 days before, the day of, and 2 days following pemetrexed administration. |
| CYP2C9 Inhibitors or Inducers: | |
| Clinical Impact: | Diclofenac is metabolized by cytochrome P450 enzymes, predominantly by CYP2C9. Coadministration of diclofenac with CYP2C9 inhibitors (e.g., voriconazole) may enhance the exposure and toxicity of diclofenac whereas coadministration with CYP2C9 inducers (e.g., rifampin) may lead to compromised efficacy of diclofenac. |
| Intervention: | A dosage adjustment may be warranted when diclofenac is administered with CYP2C9 inhibitors or inducers (see CLINICAL PHARMACOLOGY; Pharmacokinetics ) . |
DESCRIPTION
Diclofenac sodium extended-release tablets, USP is a benzeneacetic acid derivative. Diclofenac sodium extended-release tablets of 100 mg (pink) are available for oral administration. Diclofenac sodium is a white or slightly yellowish crystalline powder and is sparingly soluble in water at 25°C. The chemical name is 2-[(2,6-dichlorophenyl)amino] benzeneacetic acid, monosodium salt. The molecular weight is 318.14. Its molecular formula is C 14 H 10 Cl 2 NNaO 2 , and it has the following structural formula:
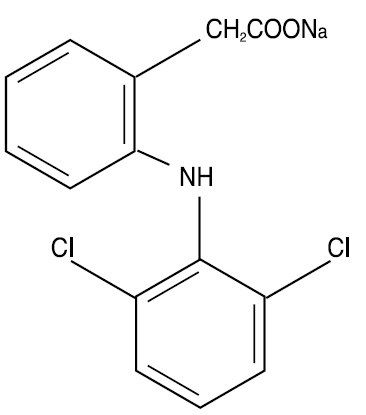
The inactive ingredients in diclofenac sodium extended-release tablets include: carnauba wax, cetostearyl alcohol, colloidal silicon dioxide, compressible sugar, copovidone, gum acacia, hydroxypropyl methylcellulose, iron oxide red, magnesium stearate, polyethylene glycol, povidone, sucrose, talc, titanium dioxide.
Meets USP Dissolution Test 2.
CLINICAL PHARMACOLOGY
Mechanism of Action
Diclofenac sodium extended-release tablets have analgesic, anti-inflammatory, and antipyretic properties. The mechanism of action of diclofenac sodium extended-release tablets, like that of other NSAIDs, is not completely understood but involves inhibition of cyclooxygenase (COX-1 and COX-2). Diclofenac is a potent inhibitor of prostaglandin synthesis in vitro. Diclofenac concentrations reached during therapy have produced in vivo effects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Because diclofenac is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
Pharmacokinetics
Absorption Diclofenac is 100% absorbed after oral administration compared to intravenous (IV) administration as measured by urine recovery. However, due to firstpass metabolism, only about 50% of the absorbed dose is systemically available (see Table 1). When diclofenac sodium extended-release tablet are taken with food, there is a delay of 1 to 2 hours in the T max and a 2-fold increase in C max values. The extent of absorption of diclofenac, however, is not significantly affected by food intake.
Table 1. Pharmacokinetic Parameters for Diclofenac
| PK Parameter | Normal Healthy Adults (18 to 48 years) | |
| Mean | Coefficient of Variation (%) | |
| Absolute Bioavailability (%) [N=7] | 55 | 40 |
| T max (hr) [N = 12] | 5.3 | 28 |
| Oral clearance (CL/F; mL/ min) [N = 12] | 895 | 56 |
| Renal clearance (% unchanged drug in urine) [N = 7] | <1 | - |
| Apparent volume of Distribution (V/F; L/kg) [N = 56] | 1.4 | 58 |
| Terminal half-life (hr) [N = 56] | 2.3 | 4.8 |
Distribution
The apparent volume of distribution (V/F) of diclofenac sodium is 1.4 L/kg. Diclofenac is more than 99% bound to human serum proteins, primarily to albumin. Serum protein binding is constant over the concentration range (0.15 to 105 mcg/mL) achieved with recommended doses. Diclofenac diffuses into and out of the synovial fluid. Diffusion into the joint occurs when plasma levels are higher than those in the synovial fluid, after which the process reverses and synovial fluid levels are higher than plasma levels. It is not known whether diffusion into the joint plays a role in the effectiveness of diclofenac.
Elimination
Metabolism Five diclofenac metabolites have been identified in human plasma and urine. The metabolites include 4’-hydroxy-, 5-hydroxy-, 3’-hydroxy-, 4’, 5-dihydroxy- and 3’-hydroxy-4’-methoxy-diclofenac. The major diclofenac metabolite, 4'-hydroxy-diclofenac, has very weak pharmacologic activity. The formation of 4'-hydroxy diclofenac is primarily mediated by CYP2C9. Both diclofenac and its oxidativemetabolites undergo glucuronidation or sulfation followed by biliary excretion. Acyl glucuronidation mediated by UGT2B7 and oxidation mediated by CYP2C8 may also play a role in diclofenac metabolism. CYP3A4 is responsible for the formation of minor metabolites, 5-hydroxy- and 3’-hydroxy-diclofenac. In patients with renal dysfunction, peak concentrations of metabolites 4’-hydroxy- and 5-hydroxydiclofenac were approximately 50% and 4% of the parent compound after single oral dosing compared to 27% and 1% in normal healthy subjects. Excretion Diclofenac is eliminated through metabolism and subsequent urinary and biliary excretion of the glucuronide and the sulfate conjugates of the metabolites. Little or no free unchanged diclofenac is excreted in the urine. Approximately 65% of the dose is excreted in the urine and approximately 35% in the bile as conjugates of unchanged diclofenac plus metabolites. Because renal elimination is not a significant pathway of elimination for unchanged diclofenac, dosing adjustment in patients with mild to moderate renal dysfunction is not necessary. The terminal half-life of unchanged diclofenac is approximately 2 hours.
Special Populations
Pediatric: The pharmacokinetics of diclofenac sodium extended- release tablets has not been investigated in pediatric patients. Race: Pharmacokinetic differences due to race have not been identified. Hepatic Impairment: Hepatic metabolism accounts for almost 100% of diclofenac sodium extended-release tablets elimination, so patients with hepatic disease may require reduced doses of diclofenac sodium extended-release tablets compared to patients with normal hepatic function. Renal Impairment: Diclofenac pharmacokinetics has been investigated in subjects with renal insufficiency. No differences in the pharmacokinetics of diclofenac have been detected in studies of patients with renal impairment. In patients with renal impairment (inulin clearance 60 to 90, 30 to 60, and less than 30 mL/min; N=6 in each group), area under the curve (AUC) values and elimination rate were comparable to those in healthy subjects.
Drug Interaction Studies
Voriconazole : When co-administered with voriconazole (inhibitor of CYP2C9, 2C19 and 3A4 enzyme), the C max and AUC of diclofenac increased by 114% and 78%, respectively ( see PRECAUTIONS; Drug Interactions ). Aspirin : When NSAIDs were administered with aspirin, the protein binding of NSAIDs were reduced, although the clearance of free NSAID was not altered. The clinical significance of this interaction is not known. See Table 2 for clinically significant drug interactions of NSAIDs with aspirin ( see PRECAUTIONS; Drug Interactions ).
HOW SUPPLIED
Diclofenac sodium extended-release tablets, USP 100 mg - Pink round convex film coated tablet debossed with DX 41 on one side. Bottle of 100 NDC 42799-953-01. Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Protect from moisture. Dispense in tight container (USP).
Manufactured for: Edenbridge Pharmaceuticals, LLC
DBA Dexcel Pharma USA
Parsippany, NJ 07054
877-381-3336
Manufactured by: Dexcel Pharma Technologies Ltd.,
Israel
Revised: 02/2025