Get your patient on Doxorubicin Hydrochloride - Doxorubicin Hydrochloride injection, Solution (Doxorubicin Hydrochloride)
Doxorubicin Hydrochloride - Doxorubicin Hydrochloride injection, Solution prescribing information
WARNING
- Severe local tissue necrosis will occur if there is extravasation during administration (see DOSAGE AND ADMINISTRATION ). Doxorubicin must not be given by the intramuscular or subcutaneous route.
- Myocardial toxicity manifested in its most severe form by potentially fatal congestive heart failure (CHF) may occur either during therapy or months to years after termination of therapy. The probability of developing impaired myocardial function based on a combined index of signs, symptoms and decline in left ventricular ejection fraction (LVEF) is estimated to be 1 to 2% at a total cumulative dose of 300 mg/m 2 of doxorubicin, 3 to 5% at a dose of 400 mg/m 2 , 5 to 8% at 450 mg/m 2 and 6 to 20% at 500 mg/m 2 . The risk of developing CHF increases rapidly with increasing total cumulative doses of doxorubicin in excess of 400 mg/m 2 . Risk factors (active or dormant cardiovascular disease, prior or concomitant radiotherapy to the mediastinal/pericardial area, previous therapy with other anthracyclines or anthracenediones, concomitant use of other cardiotoxic drugs) may increase the risk of cardiac toxicity. Cardiac toxicity with doxorubicin may occur at lower cumulative doses whether or not cardiac risk factors are present. Pediatric patients are at increased risk for developing delayed cardiotoxicity.
- Secondary acute myelogenous leukemia (AML) or myelodysplastic syndrome (MDS) has been reported in patients treated with anthracyclines, including doxorubicin (see ADVERSE REACTIONS ). The occurrence of refractory secondary AML or MDS is more common when anthracyclines are given in combination with DNA-damaging anti-neoplastic agents or radiotherapy, when patients have been heavily pretreated with cytotoxic drugs, or when doses of anthracyclines have been escalated. The rate of developing secondary AML or MDS has been estimated in an analysis of 8,563 patients with early breast cancer treated in 6 studies conducted by the National Surgical Adjuvant Breast and Bowel Project (NSABP), including NSABP B-15. Patients in these studies received standard doses of doxorubicin and standard or escalated doses of cyclophosphamide (AC) adjuvant chemotherapy and were followed for 61,810 patient years. Among 4,483 such patients who received conventional doses of AC, 11 cases of AML or MDS were identified, for an incidence of 0.32 cases per 1,000 patient years (95% Cl, 0.16 to 0.57) and a cumulative incidence at 5 years of 0.21% (95% Cl, 0.11 to 0.41%). In another analysis of 1,474 patients with breast cancer who received adjuvant treatment with doxorubicin-containing regimens in clinical trials conducted at University of Texas M.D. Anderson Cancer Center, the incidence was estimated at 1.5% at 10 years. In both experiences, patients who received regimens with higher cyclophosphamide dosages, who received radiotherapy, or who were aged 50 or older had an increased risk of secondary AML or MDS. Pediatric patients are also at risk of developing secondary AML.
- Dosage should be reduced in patients with impaired hepatic function.
- Severe myelosuppression may occur.
- Doxorubicin should be administered only under the supervision of a physician who is experienced in the use of cancer chemotherapeutic agents.
INDICATIONS AND USAGE:
Doxorubicin Hydrochloride Injection, USP has been used successfully to produce regression in disseminated neoplastic conditions such as acute lymphoblastic leukemia, acute myeloblastic leukemia, Wilms' tumor, neuroblastoma, soft tissue and bone sarcomas, breast carcinoma, ovarian carcinoma, transitional cell bladder carcinoma, thyroid carcinoma, gastric carcinoma, Hodgkin's disease, malignant lymphoma and bronchogenic carcinoma in which the small cell histologic type is the most responsive compared to other cell types.
Doxorubicin is also indicated for use as a component of adjuvant therapy in women with evidence of axillary lymph node involvement following resection of primary breast cancer.
DOSAGE AND ADMINISTRATION:
When possible, to reduce the risk of developing cardiotoxicity in patients receiving doxorubicin after stopping treatment with other cardiotoxic agents, especially those with long half-lives such as trastuzumab, doxorubicin-based therapy should be delayed until the other agents have cleared from the circulation (see WARNINGS and PRECAUTIONS, General ).
Care in the administration of doxorubicin will reduce the chance of perivenous infiltration (see WARNINGS ). It may also decrease the chance of local reactions such as urticaria and erythematous streaking. On intravenous administration of doxorubicin, extravasation may occur with or without an accompanying burning or stinging sensation, even if blood returns well on aspiration of the infusion needle. If any signs or symptoms of extravasation have occurred, the injection or infusion should be immediately terminated and restarted in another vein. If extravasation is suspected, intermittent application of ice to the site for 15 min. q.i.d. x 3 days may be useful. The benefit of local administration of drugs has not been clearly established. Because of the progressive nature of extravasation reactions, close observation and plastic surgery consultation is recommended. Blistering, ulceration and/or persistent pain are indications for wide excision surgery, followed by split-thickness skin grafting.
The most commonly used dose schedule when used as a single agent is 60 to 75 mg/m 2 as a single intravenous injection administered at 21-day intervals. The lower dosage should be given to patients with inadequate marrow reserves due to old age, or prior therapy, or neoplastic marrow infiltration.
Doxorubicin has been used concurrently with other approved chemotherapeutic agents. Evidence is available that in some types of neoplastic disease, combination chemotherapy is superior to single agents. The benefits and risks of such therapy continue to be elucidated. When used in combination with other chemotherapy drugs, the most commonly used dosage of doxorubicin is 40 to 60 mg/m 2 given as a single intravenous injection every 21 to 28 days.
In a large randomized study (NSABP B-15) of patients with early breast cancer involving axillary lymph nodes (see CLINICAL PHARMACOLOGY , CLINICAL STUDIES and ADVERSE REACTIONS , Adverse Reactions in Patients with Early Breast Cancer Receiving Doxorubicin-Containing Adjuvant Therapy ), the combination dosage regimen of AC (doxorubicin 60 mg/m 2 and cyclophosphamide 600 mg/m 2 ) was administered intravenously on day 1 of each 21-day treatment cycle. Four cycles of treatment were administered.
Dose Modifications
Patients in the NSABP B-15 study could have dose modifications of AC to 75% of the starting doses for neutropenic fever/infection. When necessary, the next cycle of treatment cycle was delayed until the absolute neutrophil count (ANC) was ≥ 1,000 cells/mm 3 and the platelet count was ≥ 100,000 cells/mm 3 and nonhematologic toxicities had resolved.
Doxorubicin dosage must be reduced in case of hyperbilirubinemia as follows:
| Plasma bilirubin concentration (mg/dL) | Dosage reduction (%) |
| 1.2 to 3 | 50 |
| 3.1 to 5 | 75 |
Reconstitution Directions
It is recommended that doxorubicin be slowly administered into the tubing of a freely running intravenous infusion of Sodium Chloride Injection, USP, or 5% Dextrose Injection, USP. The tubing should be attached to a Butterfly ® needle inserted preferably into a large vein. If possible, avoid veins over joints or in extremities with compromised venous or lymphatic drainage. The rate of administration is dependent on the size of the vein and the dosage. However, the dose should be administered in not less than 3 to 5 minutes. Local erythematous streaking along the vein as well as facial flushing may be indicative of too rapid an administration. A burning or stinging sensation may be indicative of perivenous infiltration and, if this occurs, the infusion should be immediately terminated and restarted in another vein. Perivenous infiltration may occur painlessly.
Doxorubicin should not be mixed with heparin or fluorouracil since it has been reported that these drugs are incompatible to the extent that a precipitate may form. Contact with alkaline solutions should be avoided since this can lead to hydrolysis of doxorubicin. Until specific compatibility data are available, it is not recommended that doxorubicin be mixed with other drugs.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Handling and Disposal
Procedures for proper handling and disposal of anti-cancer drugs should be considered. Several guidelines on this subject have been published. 1-4 There is no general agreement that all the procedures recommended in the guidelines are necessary or appropriate. However, given the toxic nature of this substance, the following protective recommendations are provided:
- Personnel should be trained in good technique for reconstitution and handling.
- Pregnant staff should be excluded from working with this drug.
- Personnel handling doxorubicin should wear protective clothing: goggles, gowns and disposable gloves and masks.
- A designated area should be defined for reconstitution (preferably under a laminar flow system). The work surface should be protected by disposable, plastic-backed, absorbent paper.
- All items used for reconstitution, administration or cleaning, including gloves, should be placed in high-risk waste-disposal bags for high-temperature incineration.
- Spillage or leakage should be treated with dilute sodium hypochlorite (1% available chlorine) solution, preferably by soaking, and then water.
- All cleaning materials should be disposed of as indicated previously.
- In case of skin contact, thoroughly wash the affected area with soap and water or sodium bicarbonate solution. However, do not abrade the skin by using a scrub brush.
- In case of contact with the eye(s), hold back the eyelid(s) and flush the affected eye(s) with copious amounts of water for at least 15 minutes. Then seek medical evaluation by a physician.
- Always wash hands after removing gloves.
Caregivers of pediatric patients receiving doxorubicin should be counseled to take precautions (such as wearing latex gloves) to prevent contact with the patient’s urine and other body fluids for at least 5 days after each treatment.
CONTRAINDICATIONS:
Patients should not be treated with doxorubicin if they have any of the following conditions: baseline neutrophil count <1,500 cells/mm 3 ; severe hepatic impairment; recent myocardial infarction; severe myocardial insufficiency; severe arrhythmias; previous treatment with complete cumulative doses of doxorubicin, daunorubicin, idarubicin, and/or other anthracyclines and anthracenediones; or hypersensitivity to doxorubicin, any of its excipients, or other anthracyclines or anthracenediones (see WARNINGS and DOSAGE AND ADMINISTRATION ).
ADVERSE REACTIONS:
Dose limiting toxicities of therapy are myelosuppression and cardiotoxicity. Other reactions reported are:
Cardiotoxicity
(See WARNINGS ).
Cutaneous
Reversible complete alopecia occurs in most cases. Hyperpigmentation of nailbeds and dermal creases, primarily in pediatric patients, and onycholysis have been reported in a few cases. Radiation recall reaction has occurred with doxorubicin administration. Rash, itching, or photosensitivity may occur.
Gastrointestinal
Acute nausea and vomiting occurs frequently and may be severe. This may be alleviated by antiemetic therapy. Mucositis (stomatitis and esophagitis) may occur within 5 to 10 days of beginning therapy, and most patients recover from this adverse event within another 5 to 10 days. The effect may be severe leading to ulceration and represents a site of origin for severe infections. The dosage regimen consisting of administration of doxorubicin on three successive days results in greater incidence and severity of mucositis. Ulceration and necrosis of the colon, especially the cecum, may occur leading to bleeding or severe infections which can be fatal. This reaction has been reported in patients with acute non-lymphocytic leukemia treated with a 3-day course of doxorubicin combined with cytarabine. Anorexia, abdominal pain, dehydration, diarrhea, and hyperpigmentation of the oral mucosa have been occasionally reported.
Hematologic
(See WARNINGS ).
Hypersensitivity
Fever, chills and urticaria have been reported occasionally. Anaphylaxis may occur. A case of apparent cross sensitivity to lincomycin has been reported.
Neurological
Peripheral neurotoxicity in the form of local-regional sensory and/or motor disturbances have been reported in patients treated intra-arterially with doxorubicin, mostly in combination with cisplatin. Animal studies have demonstrated seizures and coma in rodents and dogs treated with intra-carotid doxorubicin. Seizures and coma have been reported in patients treated with doxorubicin in combination with cisplatin or vincristine.
Ocular
Conjunctivitis, keratitis, and lacrimation occur rarely.
Other
Malaise/asthenia have been reported.
Adverse Reactions in Patients with Early Breast Cancer Receiving Doxorubicin-Containing Adjuvant Therapy
Safety data were collected from approximately 2,300 women who participated in a randomized, open-label trial (NSABP B-15) evaluating the use of AC versus CMF in the treatment of early breast cancer involving axillary lymph nodes. In the safety analysis, the follow-up data from all patients receiving AC were combined (N=1,492 evaluable patients) and compared with data from patients receiving conventional CMF (i.e., oral cyclophosphamide; N=739 evaluable patients). The most relevant adverse events reported in this study are provided in Table 2.
Table 2. Relevant Adverse Events in Patients with Early Breast Cancer Involving Axillary Lymph Nodes
| AC• | Conventional CMF | |
| N=1,492 | N=739 | |
| Treatment administration | ||
| Mean number of cycles | 3.8 | 5.5 |
| Total cycles | 5,676 | 4,068 |
| Adverse events, % of patients | ||
| Leukopenia | ||
| Grade 3 (1,000 to 1,999 /mm 3 ) | 3.4 | 9.4 |
| Grade 4 (<1000/mm 3 ) | 0.3 | 0.3 |
| Thrombocytopenia | ||
| Grade 3 (25,000 to 49,999 /mm 3 ) | 0 | 0.3 |
| Grade 4 (<25,000 /mm 3 ) | 0.1 | 0 |
| Shock, sepsis | 1.5 | 0.9 |
| Systemic infection | 2.4 | 1.2 |
| Nausea and vomiting | ||
| Nausea only | 15.5 | 42.8 |
| Vomiting ≤ 12 hours | 34.4 | 25.2 |
| Vomiting >12 hours | 36.8 | 12 |
| Intractable | 4.7 | 1.6 |
| Alopecia | 92.4 | 71.4 |
| Partial | 22.9 | 56.3 |
| Complete | 69.5 | 15.1 |
| Weight loss | ||
| 5 to 10% | 6.2 | 5.7 |
| >10% | 2.4 | 2.8 |
| Weight gain | ||
| 5 to 10% | 10.6 | 27.9 |
| >10% | 3.8 | 14.3 |
| Cardiac function | ||
| Asymptomatic | 0.2 | 0.1 |
| Transient | 0.1 | 0 |
| Symptomatic | 0.1 | 0 |
| Treatment-related death | 0 | 0 |
• Includes pooled data from patients who received either AC alone for 4 cycles, or who were treated with AC for 4 cycles followed by 3 cycles of CMF
DESCRIPTION:
Doxorubicin is a cytotoxic anthracycline antibiotic isolated from cultures of Streptomyces peucetius var . caesius . Doxorubicin consists of a naphthacenequinone nucleus linked through a glycosidic bond at ring atom 7 to an amino sugar, daunosamine. Chemically, doxorubicin hydrochloride is: 5,12-Naphthacenedione, 10-[(3-amino-2,3,6-trideoxy-α-L- lyxo -hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11-trihydroxy-8-(hydroxylacetyl)-1-methoxy-, hydrochloride (8 S - cis )-. (8 S , 10 S )-10-[(3-Amino-2,3,6-trideoxy-α-L- lyxo -hexopyranosyl)-oxy]-8-glycoloyl-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-5,12-naphthacenedione hydrochloride [ 25316-40-9 ].
The structural formula is as follows:
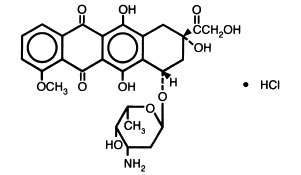
C 27 H 29 NO 11 • HCl M.W. 579.99 Doxorubicin binds to nucleic acids, presumably by specific intercalation of the planar anthracycline nucleus with the DNA double helix. The anthracycline ring is lipophilic, but the saturated end of the ring system contains abundant hydroxyl groups adjacent to the amino sugar, producing a hydrophilic center. The molecule is amphoteric, containing acidic functions in the ring phenolic groups and a basic function in the sugar amino group. It binds to cell membranes as well as plasma proteins.
Doxorubicin Hydrochloride Injection, USP is a sterile, isotonic, preservative-free solution for intravenous use. It is available in 5 mL (10 mg), 10 mL (20 mg) and 25 mL (50 mg) single dose vials and 100 mL (200 mg) multiple dose vials.
Each mL contains: Doxorubicin hydrochloride 2 mg; sodium chloride 9 mg for Isotonicity: Water for Injection q.s. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (2.5 to 4.5).
CLINICAL PHARMACOLOGY:
The cytotoxic effect of doxorubicin on malignant cells and its toxic effects on various organs are thought to be related to nucleotide base intercalation and cell membrane lipid binding activities of doxorubicin. Intercalation inhibits nucleotide replication and action of DNA and RNA polymerases. The interaction of doxorubicin with topoisomerase II to form DNA-cleavable complexes appears to be an important mechanism of doxorubicin cytocidal activity.
Doxorubicin cellular membrane binding may affect a variety of cellular functions. Enzymatic electron reduction of doxorubicin by a variety of oxidases, reductases and dehydrogenases generates highly reactive species including the hydroxyl free radical OH•. Free radical formation has been implicated in doxorubicin cardiotoxicity by means of Cu (II) and Fe (III) reduction at the cellular level.
Cells treated with doxorubicin have been shown to manifest the characteristic morphologic changes associated with apoptosis or programmed cell death. Doxorubicin-induced apoptosis may be an integral component of the cellular mechanism of action relating to therapeutic effects, toxicities, or both.
Animal studies have shown activity in a spectrum of experimental tumors, immunosuppression, carcinogenic properties in rodents, induction of a variety of toxic effects, including delayed and progressive cardiac toxicity, myelosuppression in all species and atrophy to testes in rats and dogs.
Pharmacokinetics
Pharmacokinetic studies, determined in patients with various types of tumors undergoing either single or multi-agent therapy have shown that doxorubicin follows a multiphasic disposition after intravenous injection. In four patients, doxorubicin has demonstrated dose-independent pharmacokinetics in the dose range of 30 to 70 mg/m 2 .
Distribution
The initial distribution half-life of approximately 5 minutes suggests rapid tissue uptake of doxorubicin, while its slow elimination from tissues is reflected by a terminal half-life of 20 to 48 hours. Steady-state distribution volume ranges from 809 to 1,214 L/m 2 and is indicative of extensive drug uptake into tissues. Binding of doxorubicin and its major metabolite, doxorubicinol, to plasma proteins is about 74 to 76% and is independent of plasma concentration of doxorubicin up to 1.1 mcg/mL.
Doxorubicin was excreted in the milk of one lactating patient, with peak milk concentration at 24 hours after treatment being approximately 4.4-fold greater than the corresponding plasma concentration. Doxorubicin was detectable in the milk up to 72 hours after therapy with 70 mg/m 2 of doxorubicin given as a 15-minute intravenous infusion and 100 mg/m 2 of cisplatin as a 26-hour intravenous infusion. The peak concentration of doxorubicinol in milk at 24 hours was 0.11 mcg/mL and AUC up to 24 hours was 9 mcg•h/mL while the AUC for doxorubicin was 5.4 mcg•h/mL.
Doxorubicin does not cross the blood brain barrier.
Metabolism
Enzymatic reduction at the 7 position and cleavage of the daunosamine sugar yields aglycones which are accompanied by free radical formation, the local production of which may contribute to the cardiotoxic activity of doxorubicin. Disposition of doxorubicinol (DOX-OL) in patients is formation rate limited, with the terminal half-life of DOX-OL being similar to doxorubicin. The relative exposure of DOX-OL, i.e., the ratio between the AUC of DOX-OL and the AUC of doxorubicin, compared to doxorubicin ranges between 0.4 and 0.6.
Excretion
Plasma clearance is in the range 324 to 809 mL/min/m 2 and is predominately by metabolism and biliary excretion. Approximately 40% of the dose appears in the bile in 5 days, while only 5 to 12% of the drug and its metabolites appear in the urine during the same time period. In urine, < 3% of the dose was recovered as DOX-OL over 7 days.
Systemic clearance of doxorubicin is significantly reduced in obese women with ideal body weight greater than 130%. There was a significant reduction in clearance without any change in volume of distribution in obese patients when compared with normal patients with less than 115% ideal body weight.
Pharmacokinetics in Special Populations
Pediatric
Following administration of 10 to 75 mg/m 2 doses of doxorubicin to 60 children and adolescents ranging from 2 months to 20 years of age, doxorubicin clearance averaged 1,443 ± 114 mL/min/m 2 . Further analysis demonstrated that clearance in 52 children greater than 2 years of age (1,540 mL/min/m 2 ) was increased compared with adults. However, clearance in infants younger than 2 years of age (813 mL/min/m 2 ) was decreased compared with older children and approached the range of clearance values determined in adults.
Geriatric
While the pharmacokinetics of elderly subjects (≥65 years of age) have been evaluated, no dosage adjustment is recommended based on age (see PRECAUTIONS , Geriatric Use ).
Gender
A published clinical study involving 6 men and 21 women with no prior anthracycline therapy reported a significantly higher median doxorubicin clearance in the men compared to the women (1,088 mL/min/m 2 versus 433 mL/min/m 2 ). However, the terminal half-life of doxorubicin was longer in men compared to the women (54 versus 35 hours).
Race
The influence of race on the pharmacokinetics of doxorubicin has not been evaluated.
Hepatic Impairment
The clearance of doxorubicin and doxorubicinol was reduced in patients with impaired hepatic function (see DOSAGE AND ADMINISTRATION ).
Renal Impairment
The influence of renal function on the pharmacokinetics of doxorubicin has not been evaluated.
CLINICAL STUDIES:
The effectiveness of doxorubicin-containing regimens in the adjuvant therapy of early breast cancer has primarily been established based on data collected in a meta-analysis published in 1998 by the Early Breast Cancer Trialists Collaborative Group (EBCTCG). The EBCTCG obtains primary data on all relevant studies, both published and unpublished, for early stage breast cancer and regularly updates these analyses. The principal endpoints for the adjuvant chemotherapy trials were disease-free survival (DFS) and overall survival (OS). The meta-analyses allowed comparisons of cyclophosphamide, methotrexate, and 5-fluorouracil (CMF) to no chemotherapy (19 trials including 7,523 patients) and comparisons of doxorubicin-containing regimens with CMF as an active control (6 trials including 3,510 patients). The pooled estimates of DFS and OS from these trials were used to calculate the effect of CMF relative to no therapy. The hazard ratio for DFS for CMF compared to no chemotherapy was 0.76 (95% Cl, 0.71 to 0.82) and for OS was 0.86 (95% Cl, 0.8 to 0.93). Based on a conservative estimate of CMF effect (lower 2-sided 95% confidence limit of hazard ratio) and 75% retention of CMF effect on DFS, it was determined that the doxorubicin-containing regimens would be considered as non-inferior to CMF if the upper 2-sided 95% confidence limit of the hazard ratio was less than 1.06, i.e., not more than 6% worse than CMF. A similar calculation for OS would require a non-inferiority margin of 1.02.
Six randomized trials in the EBCTCG meta-analysis compared doxorubicin-containing regimens to CMF. A total of 3,510 women with early breast cancer involving axillary lymph nodes were evaluated; approximately 70% were premenopausal and 30% were postmenopausal. At the time of the meta-analysis, 1,745 first recurrences and 1,348 deaths had occurred. Analyses demonstrated that doxorubicin-containing regimens retained at least 75% of the historical CMF adjuvant effect on DFS and are effective. The hazard ratio for DFS (dox:CMF) was 0.91 (95% Cl, 0.82 to 1.01) and for OS was 0.91 (95% Cl, 0.81 to 1.03). Results of these analyses for both DFS and OS are provided in Table 1 and Figures 1 and 2.
Table 1. Summary of Randomized Trials Comparing Doxorubicin-Containing Regimens Versus CMF in EBCTCG Meta-Analysis
| Study (starting year) | Regimens | No. of Cycles | No. of Patients | Doxorubicin-Containing Regimens vs CMF HR (95% CI) | |
| DFS | OS | ||||
| NSABP B-15 (1984) | AC | 4 | 1,562• | 0.93 (0.82 to 1.06) | 0.97 (0.83 to 1.12) |
| CMF | 6 | 776 | |||
| SECSG 2 (1976) | FAC | 6 | 260 | 0.86 (0.66 to 1.13) | 0.93 (0.69 to 1.26) |
| CMF | 6 | 268 | |||
| ONCOFRANCE (1978) | FACV | 12 | 138 | 0.71 (0.49 to 1.03) | 0.65 (0.44 to 0.96) |
| CMF | 12 | 113 | |||
| SE Sweden BCG A (1980) | AC | 6 | 21 | 0.59 (0.22 to 1.61) | 0.53 (0.21 to 1.37) |
| CMF | 6 | 22 | |||
| NSABC Israel Br0283 (1983) | AVbCMF † CMF | 4 6 6 | 55 50 | 0.91 (0.53 to 1.57) | 0.88 (0.47 to 1.63) |
| Austrian BCSG 3 (1984) | CMFVA | 6 | 121 | 1.07 (0.73 to 1.55) | 0.93 (0.64 to 1.35) |
| CMF | 8 | 124 | |||
| Combined Studies | Doxorubicin-Containing Regimens | 2,157 | 0.91 (0.82 to 1.01) | 0.91 (0.81 to 1.03) | |
| CMF | 1,353 | ||||
Abbreviations: DFS = disease free survival; OS = overall survival; AC = doxorubicin, cyclophosphamide; AVbCMF = doxorubicin, vinblastine, cyclophosphamide, methotrexate, 5-fluorouracil; CMF = cyclophosphamide, methotrexate, 5-fluorouracil; CMFVA = cyclophosphamide, methotrexate, 5-fluorouracil, vincristine, doxorubicin; FAC = 5-fluorouracil, doxorubicin, cyclophosphamide; FACV = 5-fluorouracil, doxorubicin, cyclophosphamide, vincristine; HR = hazard ratio; CI = confidence interval
_________________________________________________________________________________________________________________________
• Includes pooled data from patients who received either AC alone for 4 cycles, or who were treated with AC for 4 cycles followed by 3 cycles of CMF.
† Patients received alternating cycles of AVb and CMF.
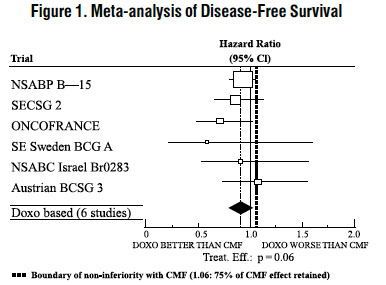
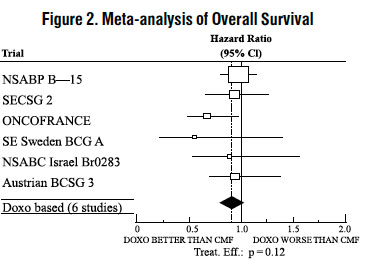
With respect to DFS, 2 of 6 studies (NSABP B-15 and ONCOFRANCE) met the non-inferiority standard individually and with respect to OS, 1 study met the non-inferiority margin individually (ONCOFRANCE). The largest of the 6 studies in the EBCTCG meta-analysis, a randomized, open-label, multicenter trial (NSABP B-15) was conducted in approximately 2,300 women (80% premenopausal; 20% postmenopausal) with early breast cancer involving axillary lymph nodes. In this trial, 6 cycles of conventional CMF was compared to 4 cycles of doxorubicin and cyclophosphamide (AC) and 4 cycles of AC followed by 3 cycles of CMF. No statistically significant differences in terms of DFS or OS were observed (see Table 1).
HOW SUPPLIED:
Doxorubicin Hydrochloride Injection, USP, 2 mg per mL, a sterile product which contains no preservatives, is available as follows:
| Product No. | NDC No. | |
| 88305 | 63323-883-05 | Doxorubicin hydrochloride 10 mg in a 5 mL single dose flip-top vial, packaged individually. |
| 88310 | 63323-883-10 | Doxorubicin hydrochloride 20 mg in a 10 mL single dose flip-top vial, packaged individually. |
| 88330 | 63323-883-30 | Doxorubicin hydrochloride 50 mg in a 25 mL single dose flip-top vial, packaged individually. |
| 100161 | 63323-101-61 | Doxorubicin hydrochloride 200 mg in a 100 mL multiple dose vial, packaged individually. |
REFRIGERATE AT: 2 ° to 8 °C ( 36 ° to 46 °F).
Protect from light (keep in outer carton). Preservative Free. Discard unused portion.
The container closure is not made with natural rubber latex.