Get your patient on Enrofloxacin 100 (Enrofloxacin)
Enrofloxacin 100 prescribing information
INDICATIONS:
Cattle - Single-Dose Therapy: Enrofloxacin 100 Injection is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and Mycoplasma bovis in beef and non-lactating dairy cattle; and for the control of BRD in beef and non-lactating dairy cattle at high risk of developing BRD associated with M. haemolytica, P. multocida, H. somni and M. bovis.
Cattle - Multiple-DayTherapy: Enrofloxacin 100 Injection is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida and Histophilus somni in beef and non-lactating dairy cattle.
Swine: Enrofloxacin 100 Injection is indicated for the treatment and control of swine respiratory disease (SRD) associated with Actinobacillus pleuropneumoniae, Pasteurella multocida, Haemophilus parasuis, Streptococcus suis, Bordetella bronchiseptica and Mycoplasma hyopneumoniae. Enrofloxacin 100 injection is indicated for the control of colibacillosis in groups or pens of weaned pigs where colibacillosis associated with Escherichia coli has been diagnosed.
DOSAGE AND ADMINISTRATION:
Enrofloxacin 100 Injection provides flexible dosages and durations of therapy. Enrofloxacin 100 Injection may be administered as a single dose for one day for treatment and control of BRD (cattle), for treatment and control of SRD or for control of colibacillosis (swine), or for multiple days for BRD treatment (cattle). Selection of the appropriate dose and duration of therapy for BRD treatment in cattle should be based on an assessment of the severity of the disease, pathogen susceptibility and clinical response.
Cattle:
Single-Dose Therapy (BRD Treatment): Administer, by subcutaneous injection, a single dose of 7.5-12.5 mg/kg of body weight (3.4-5.7 mL/100 lb).
Multiple-Day Therapy (BRD Treatment): Administer daily, a subcutaneous dose of 2.5-5 mg/kg of body weight (1.1-2.3 mL/100 lb). Treatment should be repeated at 24-hour intervals for three days. Additional treatments may be given on Days 4 and 5 to animals that have shown clinical improvement but not total recovery.
Single-Dose Therapy (BRD Control): Administer, by subcutaneous injection, a single dose of 7.5 mg/kg of body weight (3.4 mL/100 lb). Examples of conditions that may contribute to calves being at high risk for developing BRD include, but are not limited to, the following:
- Transportation with animals from two or more farm origins.
- An extended transport time with few to no rest stops.
- An environmental temperature change of ≥30°F during transportation.
- A ≥30°F range in temperature fluctuation within a 24-hour period.
- Exposure to wet or cold weather conditions.
- Excessive shrink (more than would be expected with a normal load of cattle).
- Stressful arrival processing procedures (e.g., castration or dehorning).
- Exposure within the prior 72 hours to animals showing clinical signs of BRD.
Administered dose volume should not exceed 20 mL per injection site.
•Dose volumes have been rounded to the nearest 0.5 mL within the dose range. | |||
| Treatment | Control | ||
| Weight (lb) | Single-Dose Therapy 7.5-12.5 mg/kg Dose Volume (mL) | Multiple-Day Therapy 2.5-5.0 mg/kg Dose Volume (mL) | Single-Dose Therapy 7.5 mg/kg Dose Volume (mL) |
| 100 | 3.5-5.5 | 1.5-2.0 | 3.5 |
| 200 | 7.0-11.0 | 2.5-4.5 | 7.0 |
| 300 | 10.5-17.0 | 3.5-6.5 | 10.5 |
| 400 | 14.0-22.5 | 4.5-9.0 | 14.0 |
| 500 | 17.0-28.5 | 5.5-11.5 | 17.0 |
| 600 | 20.5-34.0 | 7.0-13.5 | 20.5 |
| 700 | 24.0 - 39.5 | 8.0-16.0 | 24.0 |
| 800 | 27.5 - 45.5 | 9.0-18.0 | 27.5 |
| 900 | 31.0-51.0 | 10.0-20.5 | 31.0 |
| 1000 | 34.0 - 57.0 | 11.0-23.0 | 34.0 |
| 1100 | 37.5-62.5 | 12.5-25.0 | 37.5 |
Swine:
Administer, either by intramuscular or subcutaneous (behind the ear) injection, a single dose of 7.5 mg/kg of body weight (3.4 mL/100 lb). Administered dose volume should not exceed 5 mL per injection site.
For the control of colibacillosis, administration should be initiated within the first 60 days post-weaning when clinical signs are present in at least 2% of the animals in the group. If no improvement is noted within 48 hours, the diagnosis should be reevaluated.
| Weight (lb) | Dose Volume (mL) |
| 15 | 0.5 |
| 30 | 1.0 |
| 50 | 1.7 |
| 100 | 3.4 |
| 150 | 5.1 |
| 200 | 6.8 |
| 250 | 8.5 |
Dilution of Enrofloxacin 100 Injection: Enrofloxacin 100 Injection may be diluted with sterile water prior to injection. The diluted product should be used within 24 hours. Store diluted solution in amber glass bottles between 4-40°C (36-104°F).
•For 1 mL dose volume from diluted solution | |||
| Swine weight | mL of Enrofloxacin 100 Injection | mL of sterile water | Number of doses |
| 10 lb | 34 mL | 66 mL | 100 |
| 15 lb | 51 mL | 49 mL | 100 |
| 20 lb | 68 mL | 32 mL | 100 |
| 25 lb | 85 mL | 15 mL | 100 |
100 mL vial: Use within 30 days of first puncture and puncture a maximum of 36 times. When using a needle or draw-off spike larger than 16 gauge, discard any remaining product immediately after use.
250 mL and 500 mL vials: Use within 30 days of first puncture. Puncture a maximum of 36 times with a needle or dosage delivery device 16 gauge or smaller, or 4 times with a draw-off spike 5 mm or smaller. When using a needle larger than 16 gauge, or a draw-off spike larger than 5 mm, discard any remaining product immediately after use.
ADVERSE REACTIONS:
No adverse reactions were observed during clinical trials.
To report suspected adverse drug events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS), call Covetrus ® North America at (855) 724-3461.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1 -888-FDA-VETS or online at www.fda.gov/reportanimalae.
PRODUCT DESCRIPTION:
Enrofloxacin 100 Injection is a sterile, ready-to-use injectable antimicrobial solution that contains enrofloxacin, a broad-spectrum fluoroquinolone antimicrobial agent. Each mL of Enrofloxacin 100 Injection contains 100 mg of enrofloxacin. Excipients are L-arginine base 200 mg, n-butyl alcohol 30 mg, benzyl alcohol (as a preservative) 20 mg and water for injection q.s.
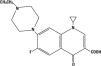
CHEMICAL NOMENCLATURE AND STRUCTURE:
1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1, 4-dihydro-4-oxo-3-quinolinecarboxylic acid.
HOW SUPPLIED:
Enrofloxacin 100 Injection:
| 100 mg/mL | 100 mL Bottle |
| 100 mg/mL | 250 mL Bottle |
| 100 mg/mL | 500 mL Bottle |