Get your patient on Ergotamine Tartrate And Caffeine - Ergotamine Tartrate And Caffeine tablet, Film Coated (Ergotamine Tartrate And Caffeine)
Ergotamine Tartrate And Caffeine - Ergotamine Tartrate And Caffeine tablet, Film Coated prescribing information
WARNING
Serious and/or life-threatening peripheral ischemia has been associated with the coadministration of ergotamine tartrate and caffeine tablets with potent CYP 3A4 inhibitors including protease inhibitors and macrolide antibiotics. Because CYP 3A4 inhibition elevates the serum levels of ergotamine tartrate and caffeine tablets, the risk for vasospasm leading to cerebral ischemia and/or ischemia of the extremities is increased. Hence, concomitant use of these medications is contraindicated (see also CONTRAINDICATIONS and WARNINGS sections).
INDICATIONS AND USAGE
Ergotamine tartrate and caffeine tablets are indicated as therapy to abort or prevent vascular headache; e.g., migraine, migraine variants or so-called “histaminic cephalalgia.”
DOSAGE AND ADMINISTRATION
Procedure: For the best results, dosage should start at the first sign of an attack. Adults: Take 2 tablets at the start of attack; 1 additional tablet every ½ hour, if needed for full relief (maximum 6 tablets per attack, 10 per week).
Maximum Adult Dosage:
Total daily dose for any one attack should not exceed 6 tablets. Total weekly dosage should not exceed 10 tablets. Ergotamine tartrate and caffeine tablets should not be used for chronic daily administration. In carefully selected patients, with due consideration of maximum dosage recommendations, administration of the drug at bedtime may be an appropriate short-term preventive measure.
CONTRAINDICATIONS
Coadministration of ergotamine with potent CYP 3A4 inhibitors (ritonavir, nelfinavir, indinavir, erythromycin, clarithromycin, and troleandomycin) has been associated with acute ergot toxicity (ergotism) characterized by vasospasm and ischemia of the extremities (see PRECAUTIONS : Drug Interactions ), with some cases resulting in amputation. There have been rare reports of cerebral ischemia in patients on protease inhibitor therapy when ergotamine tartrate and caffeine tablets was coadministered, at least one resulting in death. Because of the increased risk of ergotism and other serious vasospastic adverse events, ergotamine use is contraindicated with these drugs and other potent inhibitors of CYP 3A4 (e.g., ketoconazole, itraconazole) (see WARNINGS : CYP 3A4 Inhibitors ).
Ergotamine tartrate and caffeine tablets may cause fetal harm when administered to pregnant women. Ergotamine tartrate and caffeine tablets is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy or if the patient becomes pregnant while taking this product, the patient should be apprised of the potential hazard to the fetus.
Peripheral vascular disease, coronary heart disease, hypertension, impaired hepatic or renal function and sepsis.
Hypersensitivity to any of the components.
ADVERSE REACTIONS
Cardiovascular: Vasoconstrictive complications of a serious nature may occur at times. These include ischemia, cyanosis, absence of pulse, cold extremities, gangrene, precordial distress and pain, EKG changes and muscle pains. Although these effects occur most commonly with long-term therapy at relatively high doses, they have also been reported with short-term or normal doses. Other cardiovascular adverse effects include transient tachycardia or bradycardia and hypertension.
Gastrointestinal: Nausea and vomiting
Neurological: Paresthesias, numbness, weakness, and vertigo
Allergic: Localized edema and itching.
Fibrotic Complications: (see WARNINGS ).
To report SUSPECTED ADVERSE REACTIONS, call 1-855-899-4237, or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
CYP 3A4 Inhibitors (e.g. Macrolide Antibiotics and Protease Inhibitors)
See CONTRAINDICATIONS and WARNINGS .
Ergotamine tartrate and caffeine tablets should not be administered with other vasoconstrictors. Use with sympathomimetics (pressor agents) may cause extreme elevation of blood pressure. The beta-blocker Inderal (propranolol) has been reported to potentiate the vasoconstrictive action of ergotamine tartrate and caffeine tablets by blocking the vasodilating property of epinephrine. Nicotine may provoke vasoconstriction in some patients, predisposing to a greater ischemic response to ergot therapy.
The blood levels of ergotamine-containing drugs are reported to be elevated by the concomitant administration of macrolide antibiotics and vasospastic reactions have been reported with therapeutic doses of the ergotamine-containing drugs when coadministered with these antibiotics.
DESCRIPTION
Each tablet for oral administration contains 1 mg ergotamine tartrate, USP, and 100 mg caffeine, USP.
ERGOTAMINE TARTRATE:
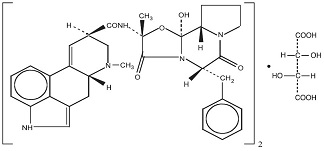
(C 33 H 35 N 5 O 5 ) 2 • C 4 H 6 O 6
M.W. 1313.41
Ergotaman-3’,6’,18-trione, 12’-hydroxy-2’-methyl-5’-(phenyl-methyl)-, (5’α), [R-(R•, R•)]-2,3-dihydroxy-butanedioate (2:1) (salt)
CAFFEINE:
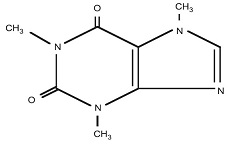
C 8 H 10 N 4 O 2 (anhydrous)
M.W. 194.19
1 H -Purine-2,6-dione, 3,7-dihydro-1,3,7-trimethyl-
Inactive ingredients include colloidal silicon dioxide, crospovidone, magnesium stearate, and microcrystalline cellulose. Film coating includes the following ingredients: macrogol/PEG 3350, polyvinyl alcohol, purified water, talc, titanium dioxide, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, and FD&C yellow #5 tartrazine aluminum lake.
CLINICAL PHARMACOLOGY
Ergotamine is an alpha adrenergic blocking agent with a direct stimulating effect on the smooth muscle of peripheral and cranial blood vessels and produces depression of central vasomotor centers. The compound also has the properties of serotonin antagonism. In comparison to hydrogenated ergotamine, the adrenergic blocking actions are less pronounced and vasoconstrictive actions are greater.
Caffeine, also a cranial vasoconstrictor, is added to further enhance the vasoconstrictive effect without the necessity of increasing ergotamine dosage.
Many migraine patients experience excessive nausea and vomiting during attacks, making it impossible for them to retain any oral medication. In such cases, therefore, the only practical means of medication is through the rectal route where medication may reach the cranial vessels directly, evading the splanchnic vasculature and the liver.
Pharmacokinetics
Interactions : Pharmacokinetic interactions (increased blood levels of ergotamine) have been reported in patients treated orally with ergotamine and macrolide antibiotics (e.g., troleandomycin, clarithromycin, erythromycin), and in patients treated orally with ergotamine and protease inhibitors (e.g. ritonavir) presumably due to inhibition of cytochrome P450 3A metabolism of ergotamine (see CONTRAINDICATIONS ). Ergotamine has also been shown to be an inhibitor of cytochrome P450 3A catalyzed reactions. No pharmacokinetic interactions involving other cytochrome P450 isoenzymes are known.
HOW SUPPLIED
Ergotamine tartrate and caffeine tablets, USP for oral administration are available as:
1 mg/100 mg: beige (film coated), round, convex face tablets, debossed “123” on one side and “MH” on the other side.
Bottles of 100......NDC 24470-917-10
Bottles of 40 .......NDC 24470-917-40
Store at 20° to 25°C (68° to 77°F) (see USP Controlled Room Temperature).
Dispense in a tight, light-resistant container.
KEEP OUT OF THE REACH OF CHILDREN.
Distributed by
Cintex Services, LLC
Dallas, TX 75243
Manufactured by
MIKART, LLC
Atlanta, GA 30318
Code 1098C00
Rev. 11/24