Get your patient on Ethacrynic Acid - Ethacrynic Acid tablet (Ethacrynic Acid)
Ethacrynic Acid - Ethacrynic Acid tablet prescribing information
INDICATIONS AND USAGE
Ethacrynic Acid Tablets USP are indicated for treatment of edema when an agent with greater diuretic potential than those commonly employed is required.
- Treatment of the edema associated with congestive heart failure, cirrhosis of the liver, and renal disease, including the nephrotic syndrome.
- Short-term management of ascites due to malignancy, idiopathic edema, and lymphedema.
- Short-term management of hospitalized pediatric patients, other than infants, with congenital heart disease or the nephrotic syndrome.
Intravenous ethacrynate sodium is indicated when a rapid onset of diuresis is desired, e.g., in acute pulmonary edema, or when gastrointestinal absorption is impaired or oral medication is not practicable.
DOSAGE AND ADMINISTRATION
Dosage must be regulated carefully to prevent a more rapid or substantial loss of fluid or electrolyte than is indicated or necessary. The magnitude of diuresis and natriuresis is largely dependent on the degree of fluid accumulation present in the patient. Similarly, the extent of potassium excretion is determined in large measure by the presence and magnitude of aldosteronism.
Oral Use
Ethacrynic Acid Tablets USP are available for oral use as 25 mg tablets.
Dosage
To Initiate Diuresis
In Adults
The smallest dose required to produce gradual weight loss (about 1 to 2 pounds per day) is recommended. Onset of diuresis usually occurs at 50 to 100 mg for adults. After diuresis has been achieved, the minimally effective dose (usually from 50 to 200 mg daily) may be given on a continuous or intermittent dosage schedule. Dosage adjustments are usually in 25 to 50 mg increments to avoid derangement of water and electrolyte excretion.
The patient should be weighed under standard conditions before and during the institution of diuretic therapy with this compound. Small alterations in dose should effectively prevent a massive diuretic response. The following schedule may be helpful in determining the smallest effective dose.
- Day 1 — 50 mg once daily after a meal
- Day 2 — 50 mg twice daily after meals, if necessary
- Day 3 — 100 mg in the morning and 50 to 100 mg following the afternoon or evening meal, depending upon response to the morning dose.
A few patients may require initial and maintenance doses as high as 200 mg twice daily. These higher doses, which should be achieved gradually, are most often required in patients with severe, refractory edema.
In Pediatric Patients
(excluding infants, see CONTRAINDICATIONS ): The initial dose should be 25 mg. Careful stepwise increments in dosage of 25 mg should be made to achieve effective maintenance.
Maintenance Therapy
It is usually possible to reduce the dosage and frequency of administration once dry weight has been achieved.
Ethacrynic Acid Tablets USP may be given intermittently after an effective diuresis is obtained with the regimen outlined above
Dosage may be on an alternate daily schedule or more prolonged periods of diuretic therapy may be interspersed with rest periods. Such an intermittent dosage schedule allows time for correction of any electrolyte imbalance and may provide a more efficient diuretic response.
The chloruretic effect of this agent may give rise to retention of bicarbonate and a metabolic alkalosis. This may be corrected by giving chloride (ammonium chloride or arginine chloride). Ammonium chloride should not be given to cirrhotic patients.
Ethacrynic acid has additive effects when used with other diuretics. For example, a patient who is on maintenance dosage of an oral diuretic may require additional intermittent diuretic therapy, such as an organomercurial, for the maintenance of basal weight. The intermittent use of ethacrynic acid orally may eliminate the need for injections of organomercurials. Small doses of ethacrynic acid may be added to existing diuretic regimens to maintain basal weight. This drug may potentiate the action of carbonic anhydrase inhibitors, with augmentation of natriuresis and kaliuresis. Therefore, when adding ethacrynic acid, the initial dose and changes of dose should be in 25 mg increments, to avoid electrolyte depletion. Rarely, patients who failed to respond to ethacrynic acid have responded to older established agents.
While many patients do not require supplemental potassium, the use of potassium chloride or potassium-sparing agents, or both, during treatment with ethacrynic acid is advisable, especially in cirrhotic or nephrotic patients and in patients receiving digitalis.
Salt liberalization usually prevents the development of hyponatremia and hypochloremia. During treatment with ethacrynic acid, salt may be liberalized to a greater extent than with other diuretics. Cirrhotic patients, however, usually require at least moderate salt restriction concomitant with diuretic therapy.
Intravenous Use
Ethacrynate sodium is for intravenous use when oral intake is impractical or in urgent conditions, such as acute pulmonary edema
CONTRAINDICATIONS
All diuretics, including ethacrynic acid, are contraindicated in anuria. If increasing electrolyte imbalance, azotemia, and/or oliguria occur during treatment of severe, progressive renal disease, the diuretic should be discontinued.
In a few patients this diuretic has produced severe, watery diarrhea. If this occurs, it should be discontinued and not used again. Until further experience in infants is accumulated, therapy with oral ethacrynic acid is contraindicated. Hypersensitivity to any component of this product.
ADVERSE REACTIONS
Gastrointestinal
Anorexia, malaise, abdominal discomfort or pain, dysphagia, nausea, vomiting, and diarrhea have occurred. These are more frequent with large doses or after one to three months of continuous therapy. A few patients have had sudden onset of profuse, watery diarrhea. Discontinue Ethacrynic Acid Tablets USP if diarrhea is severe and do not give it again. Gastrointestinal bleeding has occurred in some patients. Rarely, acute pancreatitis has been reported.
Metabolic
Reversible hyperuricemia and acute gout have been reported. Acute symptomatic hypoglycemia with convulsions occurred in two uremic patients who received doses above those recommended. Hyperglycemia has been reported. Rarely, jaundice and abnormal liver function tests have been reported in seriously ill patients receiving multiple drug therapy, including Ethacrynic Acid Tablets USP.
Hematologic
Agranulocytosis or severe neutropenia has been reported in a few critically ill patients also receiving agents known to produce this effect. Thrombocytopenia has been reported rarely. Henoch-Schönlein purpura has been reported rarely in patients with rheumatic heart disease receiving multiple drug therapy, including Ethacrynic Acid Tablets USP.
Special Senses
(see WARNINGS )
Deafness, tinnitus and vertigo with a sense of fullness in the ears, and blurred vision have occurred.
Central Nervous System
Headache, fatigue, apprehension, confusion.
Miscellaneous
Skin rash, fever, chills, hematuria.
To report SUSPECTED ADVERSE REACTIONS, contact Edenbridge Pharmaceuticals, LLC at 1-877-381-3336 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Lithium generally should not be given with diuretics because they reduce its renal clearance and add a high risk of lithium toxicity.
Read circulars for lithium preparations before use of such concomitant therapy.
Ethacrynic acid may increase the ototoxic potential of other drugs such as aminoglycoside and some cephalosporin antibiotics. Their concurrent use should be avoided.
A number of drugs, including ethacrynic acid, have been shown to displace warfarin from plasma protein; a reduction in the usual anticoagulant dosage may be required in patients receiving both drugs.
In some patients, the administration of a non-steroidal anti-inflammatory agent can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing and thiazide diuretics. Therefore, when ethacrynic acid and non-steroidal antiinflammatory agents are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained.
DESCRIPTION
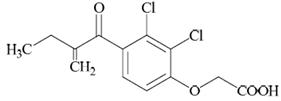
Ethacrynic acid is an unsaturated ketone derivative of an aryloxyacetic acid. It is designated chemically as [2,3-dichloro-4-(2-methylene-1-oxobutyl)phenoxy] acetic acid, and has a molecular weight of 303.14. Ethacrynic acid is a white, or practically white, crystalline powder, very slightly soluble in water, but soluble in most organic solvents such as alcohols, chloroform, and benzene. Its empirical formula is C 13 H 12 Cl 2 O 4 and its structural formula is:
Ethacrynic Acid Tablets USP are supplied as 25 mg tablets for oral use. The tablets contain the following inactive ingredients: calcium stearate, colloidal silicon dioxide, lactose monohydrate, pre-gelatinized starch and talc.
USP Dissolution Test is pending.
CLINICAL PHARMACOLOGY
Pharmacokinetics and Metabolism
Ethacrynic acid acts on the ascending limb of the loop of Henle and on the proximal and distal tubules. Urinary output is usually dose dependent and related to the magnitude of fluid accumulation. Water and electrolyte excretion may be increased several times over that observed with thiazide diuretics, since ethacrynic acid inhibits reabsorption of a much greater proportion of filtered sodium than most other diuretic agents. Therefore, ethacrynic acid is effective in many patients who have significant degrees of renal insufficiency (see WARNINGS concerning deafness). Ethacrynic acid has little or no effect on glomerular filtration or on renal blood flow, except following pronounced reductions in plasma volume when associated with rapid diuresis.
The electrolyte excretion pattern of ethacrynic acid varies from that of the thiazides and mercurial diuretics. Initial sodium and chloride excretion is usually substantial and chloride loss exceeds that of sodium. With prolonged administration, chloride excretion declines, and potassium and hydrogen ion excretion may increase. Ethacrynic acid is effective whether or not there is clinical acidosis or alkalosis.
Although ethacrynic acid, in carefully controlled studies in animals and experimental subjects, produces a more favorable sodium/potassium excretion ratio than the thiazides, in patients with increased diuresis excessive amounts of potassium may be excreted. Onset of action is rapid, usually within 30 minutes after an oral dose of ethacrynic acid tablets. After oral use, diuresis peaks in about 2 hours and lasts about 6 to 8 hours.
The sulfhydryl binding propensity of ethacrynic acid differs somewhat from that of the organomercurials. Its mode of action is not by carbonic anhydrase inhibition.
Ethacrynic acid does not cross the blood-brain barrier.
HOW SUPPLIED
Ethacrynic Acid Tablets USP, 25 mg, are white, capsule shaped, scored tablets, debossed with "4" on left side of the score and "05" on the right side of the score on one side and plain on the other side. They are supplied as follows:
NDC 42799-405-01 in bottles of 100.
Storage
Store in a tightly closed container at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].