Get your patient on Ethosuximide - Ethosuximide capsule (Ethosuximide)
Ethosuximide - Ethosuximide capsule prescribing information
INDICATIONS AND USAGE
Ethosuximide is indicated for the control of absence (petit mal) epilepsy.
DOSAGE AND ADMINISTRATION
Ethosuximide is administered by the oral route. The initial dose for patients 3 to 6 years of age is one capsule (250 mg) per day; for patients 6 years of age and older, 2 capsules (500 mg) per day. The dose thereafter must be individualized according to the patient's response. Dosage should be increased by small increments. One useful method is to increase the daily dose by 250 mg every four to seven days until control is achieved with minimal side effects. Dosages exceeding 1.5 g daily, in divided doses, should be administered only under the strictest supervision of the physician. The optimal dose for most pediatric patients is 20 mg/kg/day. This dose has given average plasma levels within the accepted therapeutic range of 40 to 100 mcg/mL. Subsequent dose schedules can be based on effectiveness and plasma level determinations.
Ethosuximide may be administered in combination with other anticonvulsants when other forms of epilepsy coexist with absence (petit mal). The optimal dose for most pediatric patients is 20 mg/kg/day.
CONTRAINDICATIONS
Ethosuximide should not be used in patients with a history of hypersensitivity to succinimides.
ADVERSE REACTIONS
Body As A Whole
Allergic reaction, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS).
Gastrointestinal System
Gastrointestinal symptoms occur frequently and include anorexia, vague gastric upset, nausea and vomiting, cramps, epigastric and abdominal pain, weight loss, and diarrhea. There have been reports of gum hypertrophy and swelling of the tongue.
Hemopoietic System
Hemopoietic complications associated with the administration of ethosuximide have included leukopenia, agranulocytosis, pancytopenia, with or without bone marrow suppression, eosinophilia and thrombocytopenia (see WARNINGS ).
Nervous System
Neurologic and sensory reactions reported during therapy with ethosuximide have included drowsiness, headache, dizziness, euphoria, hiccups, irritability, hyperactivity, lethargy, fatigue, and ataxia. Psychiatric or psychological aberrations associated with ethosuximide administration have included disturbances of sleep, night terrors, inability to concentrate, and aggressiveness. These effects may be noted particularly in patients who have previously exhibited psychological abnormalities. There have been rare reports of paranoid psychosis, increased libido, and increased state of depression with overt suicidal intentions.
Integumentary System
Dermatologic manifestations which have occurred with the administration of ethosuximide have included urticaria, pruritic erythematous rashes, Stevens-Johnson syndrome, and hirsutism.
Special Senses
Myopia .
Genitourinary System
Vaginal bleeding, microscopic hematuria.
Drug Interactions
Since ethosuximide may interact with concurrently administered antiepileptic drugs, periodic serum level determinations of these drugs may be necessary (e.g., ethosuximide may elevate phenytoin serum levels and valproic acid has been reported to both increase and decrease ethosuximide levels).
DESCRIPTION
Ethosuximide is an anticonvulsant succinimide, chemically designated as alpha-ethyl-alphamethyl-succinimide, with the following structural formula:
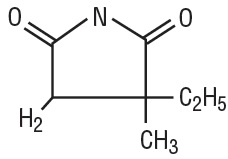
Each ethosuximide capsule contains 250 mg ethosuximide, USP. In addition, each capsule contains FD&C Red No. 3, FD&C Yellow No. 6, Gelatin, NF, Glycerin, USP, Polyethylene Glycol 400, NF, Purified Water. The white ink contains N-butyl Alcohol, Propylene Glycol, Shellac Glaze, and Titanium Dioxide.
It meets USP Dissolution Test 1.
CLINICAL PHARMACOLOGY
Ethosuximide suppresses the paroxysmal three cycle per second spike and wave activity associated with lapses of consciousness which is common in absence (petit mal) seizures. The frequency of epileptiform attacks is reduced, apparently by depression of the motor cortex and elevation of the threshold of the central nervous system to convulsive stimuli.
HOW SUPPLIED
Ethosuximide Capsules USP, 250 mg are clear orange oblong-shaped capsules, imprinted "HW01" with white ink and are supplied as follows:
NDC 42806-708-01: Bottles of 100.
Store at 20° to 25° C (68° to 77° F) [see USP Controlled Room Temperature].
Dispense with Medication Guide available at: www.epic-pharma.com/medguide/Ethosuximide-Capsule.pdf
Distributed by:
Epic Pharma, LLC
Laurelton, NY 11413
Manufactured by:
Humanwell PuraCap Pharmaceutical
Wuhan, Hubei 430206, China
MF708REV09/22
Rev. 09-2022-00
OE2940