Get your patient on Evomela - Melphalan injection, Powder, Lyophilized, For Solution (Melphalan)
Evomela - Melphalan injection, Powder, Lyophilized, For Solution prescribing information
WARNING: SEVERE BONE MARROW SUPPRESSION, HYPERSENSITIVITY, and LEUKEMOGENICITY
- Severe bone marrow suppression with resulting infection or bleeding may occur. Controlled trials comparing intravenous (IV) melphalan to oral melphalan have shown more myelosuppression with the IV formulation. Monitor hematologic laboratory parameters. [see Warnings and Precautions (5.1 )]
- Hypersensitivity reactions, including anaphylaxis, have occurred in approximately 2% of patients who received the IV formulation of melphalan. Discontinue treatment with Evomela for serious hypersensitivity reactions. [see Warnings and Precautions (5.4 )]
- Melphalan produces chromosomal aberrations in vitro and in vivo. Evomela should be considered potentially leukemogenic in humans. [see Warnings and Precautions (5.5 )]
INDICATIONS AND USAGE
Evomela is an alkylating drug indicated for use as a high-dose conditioning treatment prior to hematopoietic progenitor (stem) cell transplantation in patients with multiple myeloma. (1.1 )
Multiple Myeloma-Conditioning Treatment
Evomela is indicated for use as a high-dose conditioning treatment prior to hematopoietic progenitor (stem) cell transplantation in patients with multiple myeloma.
DOSAGE AND ADMINISTRATION
For Conditioning Treatment , the recommended dose of Evomela is 100 mg/m 2 /day administered over 30 minutes by intravenous infusion for 2 consecutive days (Day -3 and Day -2) prior to autologous stem cell transplantation (ASCT, Day 0). (2.1 )
Recommended Dosage for Conditioning Treatment
The recommended dose of Evomela for conditioning treatment is 100 mg/m 2 /day administered over 30 minutes by intravenous infusion for 2 consecutive days (Day -3 and Day -2) prior to autologous stem cell transplantation (ASCT, Day 0). For patients who weigh more than 130% of their ideal body weight, body surface area should be calculated based on adjusted ideal body weight.
Administer prophylactic antiemetics [see Warnings and Precautions (5.2 )] .
Preparation and Administration
Evomela is a hazardous drug. Follow applicable special handling and disposal procedures 1 .
Evomela is light sensitive. Retain in original carton until use.
Do not mix Evomela with other melphalan hydrochloride for injection drug products.
Reconstitution and Infusion Instructions:
1.000000000000000e+00 Use 0.9% Sodium Chloride Injection, USP (8.6 mL as directed) to reconstitute Evomela and make a 50 mg/10 mL (5 mg/ mL) nominal concentration of melphalan.
The reconstituted Evomela drug product is stable for 24 hours at refrigerated temperature (5 o C) without any precipitation due to the high solubility.
The reconstituted Evomela drug product is stable for 1 hour at room temperature.
2.000000000000000e+00 Calculate the required volume of Evomela needed for a patient’s dose and withdraw that volume from the vial(s).
3.000000000000000e+00 Add the required volume of Evomela to the appropriate volume of 0.9% Sodium Chloride Injection, USP to a final concentration of 0.45 mg/mL. The Evomela admixture solution is stable for 4 hours at room temperature in addition to the 1 hour following reconstitution. 4.000000000000000e+00 Infuse over 30 minutes via an injection port or central venous catheter. Evomela may cause local tissue damage should extravasation occur. Do not administer by direct injection into a peripheral vein. Administer Evomela by injecting slowly into a fast-running IV infusion via a central venous access line. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
DOSAGE FORMS AND STRENGTHS
For injection: 50 mg, white to off-white lyophilized powder in single-dose vial for reconstitution (after reconstitution the solution is clear and colorless to light yellow). Each vial contains 50 mg melphalan free base equivalent to 56 mg melphalan hydrochloride.
USE IN SPECIFIC POPULATIONS
- Lactation: Advise not to breastfeed (8.2 )
Pregnancy
Risk Summary
Based on its mechanism of action, Evomela can cause fetal harm when administered to a pregnant woman, including teratogenicity and/or embryo-fetal lethality [see Clinical Pharmacology (12.1 )] . Melphalan is a genotoxic drug and can cause chromatid or chromosome damage in humans [see Nonclinical Toxicology (13.1 )] . In animal studies, melphalan was embryolethal and teratogenic in rats at doses below the recommended clinical doses [see Data] . Advise a pregnant woman of the potential risk to a fetus..
The background risk of major birth defects and miscarriage for the indicated populations are unknown. However, the background risk in the U.S. general population of major birth defects is 2-4% and of miscarriage is 15-20% of clinically recognized pregnancies.
Data
Animal Data
Adequate animal studies have not been conducted with intravenous melphalan. Melphalan was embryolethal and teratogenic in rats following oral administration of 6 to 18 mg/m 2 /day for 10 days (0.06 to 0.18 times the highest recommended clinical dose of 100 mg/m 2 /day) and intraperitoneal administration of 18 mg/m 2 (0.18 times the highest recommended clinical dose). Malformations resulting from melphalan administration included alterations of the brain (underdevelopment, deformation, meningocele, and encephalocele) and eye (anophthalmia and microphthalmos), reduction of the mandible and tail, and hepatocele (exomphaly).
Lactation
Risk Summary
It is not known whether melphalan is present in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing children from melphalan, breastfeeding is not recommended during treatment with Evomela and for one week after the last dose.
Females and Males of Reproductive Potential
Evomela can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1 )] .
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with Evomela and for 6 months after the last dose.
Males
Evomela administration may damage spermatozoa and testicular tissue, resulting in possible genetic fetal abnormalities. Advise males with female partners of reproductive potential to use effective contraception during treatment with Evomela and for 3 months after the last dose [see Nonclinical Toxicology (13.1 )].
Infertility
Females
Melphalan causes suppression of ovarian function in premenopausal women, resulting in amenorrhea in a significant number of patients.
Males
Reversible and irreversible testicular suppression has been reported in male patients after administration of melphalan.
Pediatric Use
Pediatric patients were not included in clinical trials. Safety and effectiveness have not been established in pediatric patients.
Geriatric Use
Of the total number of subjects in the single-arm pivotal study of Evomela, 30% were 65 and over, but no patients were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. A greater incidence of engraftment syndrome was observed in older patients; 7% (3 of 43) of patients younger than 65 years old versus 28% (5 of 18) of patients 65 years old and over.
CONTRAINDICATIONS
History of serious allergic reaction to melphalan.
WARNINGS AND PRECAUTIONS
- Gastrointestinal toxicity: Nausea, vomiting, diarrhea or oral mucositis may occur; provide supportive care using antiemetic and antidiarrheal medications as needed. (2.1 , 5.2 )
- Embryo-fetal toxicity: Can cause fetal harm. Advise females of reproductive potential and males with female partners of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.6 , 8.1 , 8.3 )
- Infertility: Melphalan may cause ovarian function suppression or testicular suppression. (5.7 )
Bone Marrow Suppression
For patients receiving Evomela as part of a conditioning regimen, myeloablation occurs in all patients. Do not begin the conditioning regimen if a stem cell product is not available for rescue. Monitor complete blood counts, provide supportive care for infections, anemia and thrombocytopenia until there is adequate hematopoietic recovery.
Gastrointestinal Toxicity
For patients receiving Evomela as part of a conditioning regimen, nausea, vomiting, mucositis, and diarrhea may occur in over 50% of patients. Use prophylactic antiemetic medication. Provide supportive care for nausea, vomiting, diarrhea, and mucositis. The frequency of grade 3/4 mucositis in clinical studies was 13%. Provide nutritional support and analgesics for patients with severe mucositis. [see Dosage and Administration (2.1 ) and Adverse Reactions (6.1 )] .
Hepatotoxicity
Hepatic disorders ranging from abnormal liver function tests to clinical manifestations such as hepatitis and jaundice have been reported after treatment with melphalan. Hepatic veno-occlusive disease has also been reported. Monitor liver chemistries.
Hypersensitivity
Acute hypersensitivity reactions, including anaphylaxis, have occurred in approximately 2% of patients who received an intravenous formulation of melphalan. Symptoms may include urticaria, pruritus, edema, and skin rashes and, in some patients, tachycardia, bronchospasm, dyspnea, and hypotension. Discontinue treatment with Evomela for serious hypersensitivity reactions.
Secondary Malignancies
Melphalan has been shown to cause chromatid or chromosome damage in humans. Secondary malignancies such as myeloproliferative syndrome or acute leukemia have been reported in multiple myeloma patients treated with melphalan-containing chemotherapy regimens. The potential benefit of Evomela therapy must be considered against the possible risk of the induction of a secondary malignancy.
Embryo-Fetal Toxicity
Based on its mechanism of action, Evomela can cause fetal harm when administered to a pregnant woman. Melphalan is genotoxic, targets actively dividing cells, and was embryolethal and teratogenic in rats. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Evomela and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with Evomela and for 3 months after the last dose [see Use in Specific Populations (8.1 , 8.3 )] .
Infertility
Melphalan-based chemotherapy regimens have been reported to cause suppression of ovarian function in premenopausal women, resulting in persistent amenorrhea in approximately 9% of patients. Reversible or irreversible testicular suppression has also been reported [see Use in Specific Populations (8.3 )].
ADVERSE REACTIONS
Most common adverse reactions observed in at least 50% of patients treated with Evomela are neutrophil count decreased, white blood cell count decreased, lymphocyte count decreased, platelet count decreased, diarrhea, nausea, fatigue, hypokalemia, anemia, and vomiting. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Acrotech Biopharma Inc. at 1-888-292-9617 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
The following serious adverse reactions are described in more detail in other sections of the prescribing information.
Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of Evomela may not reflect the rates observed in practice.
The most common adverse reactions observed in at least 50% of patients with multiple myeloma treated with Evomela were neutrophil count decreased, white blood cell count decreased, lymphocyte count decreased, platelet count decreased, diarrhea, nausea, fatigue, hypokalemia, anemia, and vomiting.
Myeloablative Conditioning in Multiple Myeloma Patients Undergoing ASCT
The safety of Evomela was evaluated in 61 patients with multiple myeloma in a single arm clinical trial in which patients were administered Evomela at a dosage of 100 mg/m 2 /day administered over ~30 minutes (range: 24-48 minutes) by intravenous (IV) infusion for 2 consecutive days (Day -3 and Day -2) prior to autologous stem cell transplant (ASCT, Day 0). [see Clinical Studies (14.1 )].
Table 1 summarizes the adverse reactions from the single-arm trial in patients with multiple myeloma. Severe myelosuppression is expected and these adverse reactions are not listed below.
| Adverse Reactions | Number (%) of Patients (N=61) | |
| All Grades | Grade 3or 4 | |
| All Adverse Reactions | 61 | 61 |
| Diarrhea | 57 (93%) | 2 (3%) |
| Nausea | 55 (90%) | 1 (2%) |
| Fatigue | 47 (77%) | 1 (2%) |
| Hypokalemia | 45 (74%) | 17 (28%) |
| Vomiting | 39 (64%) | 0 (0%) |
| Hypophosphatemia | 30 (49%) | 29 (2%) |
| Decreased Appetite | 30 (49%) | 0 (0%) |
| Pyrexia | 29 (48%) | 2 (3%) |
| Constipation | 29 (48%) | 0 (0%) |
| Febrile Neutropenia | 25 (41%) | 17 (28%) |
| Mucosal Inflammation | 23 (38%) | 6 (10%) |
| Dizziness | 23 (38%) | 0 (0%) |
| Edema Peripheral | 20 (33%) | 0 (0%) |
| Stomatitis | 17 (28%) | 3 (5%) |
| Abdominal Pain | 17 (28%) | 0 (0%) |
| Dysgeusia | 17 (28%) | 0 (0%) |
| Dyspepsia | 16 (26%) | 0 (0%) |
Serious Adverse Reactions
Twelve (20%) patients experienced a treatment emergent serious adverse reaction while on study. The most common serious adverse reactions (>1 patient, 1.6%) were pyrexia, hematochezia, febrile neutropenia, and renal failure. Treatment-related serious adverse reactions reported in >1 patient were pyrexia (n=2, 3%), febrile neutropenia (n=2, 3%), and hematochezia (n=2, 3%).
DRUG INTERACTIONS
No formal drug interaction studies have been conducted. The development of severe renal impairment has been reported in patients treated with a single dose of intravenous melphalan 140-250 mg/m 2 followed by standard oral doses of cyclosporine. Intravenous melphalan may also reduce the threshold for BCNU lung toxicity.
DESCRIPTION
Evomela contains melphalan hydrochloride, an alkylating drug, as the active ingredient. The chemical name of melphalan hydrochloride is 4-[bis(2-chloroethyl)amino]-L-phenylalanine hydrochloride. Its molecular formula is C 13 H 18 Cl 2 N 2 O 2 • HCl and the molecular weight is 341.67. The structural formula is:
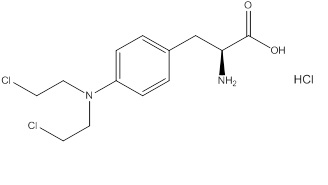
Melphalan hydrochloride is a white to off-white powder, with a melting range of 199°C − 201°C. It is practically insoluble in water, but freely soluble in 1N HCl and methanol.
Evomela (melphalan) for injection is supplied as a sterile white to off-white lyophilized powder in a single-dose vial for intravenous use. Each vial contains 50 mg melphalan free base equivalent to 56 mg melphalan hydrochloride and 2700 mg Betadex Sulfobutyl Ether Sodium, NF. Sodium hydroxide and if necessary, hydrochloric acid are added as a pH adjuster.
CLINICAL PHARMACOLOGY
Mechanism of Action
Melphalan is an alkylating agent of the bischloroethylamine type. As a result, its cytotoxicity appears to be related to the extent of its interstrand cross-linking with DNA, probably by binding at the N 7 position of guanine. Like other bifunctional alkylating agents, it is active against both resting and rapidly dividing tumor cells.
Pharmacodynamics
The peak mean heart rate increased 20 bpm from baseline following melphalan 100 mg/m 2 for 2 consecutive days in multiple myeloma patients undergoing autologous stem cell transplantation.
Cardiac Electrophysiology
No large mean increase in QTc (i.e. > 20 ms) was detected following melphalan 100 mg/m 2 .
Pharmacokinetics
Mean (± SD) peak plasma concentrations and AUC 0-inf were 5.8 ± 1.5 mcg/mL and 451 ± 109 mcg•min/mL, respectively, following administration of melphalan 100 mg/m 2 in multiple myeloma patients.
Distribution
The volume of distribution of melphalan ranges from approximately 35.5 to 185.7 L/m 2 . Melphalan penetrates into cerebrospinal fluid (CSF). Protein binding of melphalan ranges from approximately 50% to 90%, primarily to serum albumin (40% to 60%) and to a lesser extent to α1-acid glycoprotein (20%). Approximately 30% of melphalan is (covalently) irreversibly bound to plasma proteins.
Elimination
Melphalan terminal elimination half-life is approximately 75 minutes. Average total body clearance (CL) ranges from approximately 250 to 325 mL/min/m 2 .
Metabolism
Melphalan primarily undergoes chemical hydrolysis to inactive metabolites.
Excretion
Mean values of melphalan excreted in urine range from 5.8% to 21.3%.
Specific Populations
Patient Body Weight
Melphalan clearance changes with ideal body weight (IBW), which decreased by 28% and increased by 31% with IBW of 45 kg and 100 kg compared to 70 kg IBW, respectively.
Renal Impairment
A decrease in estimated creatinine CL from 100 mL/min to 30 mL/min results in 28.2% reduction in CL for a typical person with an IBW of 70 kg.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Adequate and well-controlled carcinogenicity studies have not been conducted in animals. However, intraperitoneal (IP) administration of melphalan in rats (5.4 to 10.8 mg/m 2 ) and in mice (2.25 to 4.5 mg/m 2 ) 3 times per week for 6 months followed by 12 months post-dose observation produced peritoneal sarcomas and lung tumors, respectively.
Intramuscular administration of melphalan at 6 and 60 mg/m 2 produced structural aberrations of the chromatid and chromosomes in bone marrow cells of Wistar rats.
CLINICAL STUDIES
Myeloablative Conditioning in Patients with Multiple Myeloma Undergoing ASCT
An open-label, single-arm, non-randomized trial of Evomela was conducted at 5 US centers (NCT 01660633). The 61 patients enrolled had symptomatic multiple myeloma and had at least 2 × 10 6 CD34+ cells/kg cryopreserved stem cells available. The median age was 62 years (range 32 to 73); 57% male, 80% white, 18% black, 2% Asian. Evomela was administered at 100 mg/m 2 /day over 30 minutes by IV infusion for two consecutive days (Day -3 and Day -2) prior to ASCT (Day 0).
The objective of the trial was to determine the overall safety and toxicity profile of 200 mg/m 2 of Evomela in patients with multiple myeloma undergoing ASCT. The efficacy was evaluated by the International Myeloma Working Group response criteria comparing the disease response immediately prior to the ASCT procedure to the disease response assessed 90 to 100 days post-transplant. In addition, successful myeloablation, and time to engraftment were evaluated.
The overall response rate (partial response or better) improved from 79% (48 of 61) prior to the ASCT procedure to 95% (58 of 61) at 90 to 100 days post-transplant. There was also an increase in the number of patients with a stringent complete response from 0 patients prior to the ASCT procedure to 16% (10 of 61) at 90 to 100 days post-transplant.
Myeloablation and engraftment were evaluated by complete blood cell count tests daily until neutrophil and platelet engraftment, and then weekly until Day 30, and at Day 60 and Day 90-100. Myeloablation was defined as any of the following: absolute neutrophil count (ANC) < 500/mm 3 , absolute lymphocyte count < 100/mm 3 , or platelet count < 20,000/mm 3 ). Neutrophil engraftment was defined as ANC > 500/mm 3 ×3 consecutive daily assessments. Platelet engraftment was defined as untransfused platelet counts > 20,000/mm 3 ×3 consecutive daily assessments. Nonengraftment was defined as failure to reach an ANC > 500/mm 3 ×3 consecutive daily assessments by Day 90-100.
Myeloablation, neutrophil engraftment and platelet engraftment were achieved by all 61 patients. Myeloablation occurred on ASCT Day 5 (range ASCT days -1 to 6) with the median time to myeloablation from dosing of 8 days. The median time to neutrophil engraftment was 12 days (range ASCT days 10 to 16). The median time to platelet engraftment was 13 days (range ASCT days 10 to 28).
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Evomela is supplied in a single carton containing one (1) vial. Each 50 mg vial contains a white to off- white lyophilized powder in single-dose vial for reconstitution (after reconstitution the solution is clear and coloress to light yellow). Each vial contains 50 mg melphalan free base equivalent to 56 mg melphalan hydrochloride.
NDC 72893-001-01: Individual carton of Evomela single-dose vial containing 50 mg melphalan free base.
Storage and Handling
Store Evomela at room temperature 25°C (77°F). Temperature excursions are permitted between 15- 30°C (59-86°F). [see USP Controlled Room Temperature]
Evomela is light sensitive. Retain in original carton until use.
Melphalan is a hazardous drug. Follow applicable special handling and disposal procedures. 1
Mechanism of Action
Melphalan is an alkylating agent of the bischloroethylamine type. As a result, its cytotoxicity appears to be related to the extent of its interstrand cross-linking with DNA, probably by binding at the N 7 position of guanine. Like other bifunctional alkylating agents, it is active against both resting and rapidly dividing tumor cells.