Get your patient on Exenatide - Exenatide injection (Exenatide)
Exenatide - Exenatide injection prescribing information
INDICATIONS AND USAGE
Exenatide injection is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Limitations of Use
- Exenatide injection contains exenatide. Co-administration with other exenatide-containing products is not recommended.
DOSAGE AND ADMINISTRATION
Recommended Dosing
- Initiate exenatide injection at 5 mcg administered subcutaneously twice daily at any time within the 60-minute period before the morning and evening meals (or before the two main meals of the day, approximately 6 hours or more apart). Do not administer after a meal.
- Based on clinical response, the dose of exenatide injection can be increased to 10 mcg twice daily which is recommended after 1 month of therapy, in order to reduce the risk of gastrointestinal adverse reactions [see Warnings and Precautions (5.5) and Adverse Reactions (6.1) ] .
- Administer as a subcutaneous injection in the thigh, abdomen, or upper arm.
- Rotate injections sites with each dose. Do not use the same site for each injection.
- Inspect visually for particulate matter and discoloration. Only use exenatide injection if the solution appears clear, colorless and contains no particles.
- When using exenatide injection with insulin, administer as separate injections and never mix. It is acceptable to inject exenatide injection and insulin in the same body region, but the injections should not be adjacent to each other.
- If a dose is missed, resume the treatment regimen as prescribed with the next scheduled dose.
DOSAGE FORMS AND STRENGTHS
Exenatide Injection, USP is a clear, colorless sterile solution for subcutaneous injection containing 250 mcg/mL exenatide, USP supplied as follows:
- 5 mcg per dose in a single-patient-use prefilled pen containing 300 mcg/1.2 mL (250mcg/mL), 60 doses.
- 10 mcg per dose in a single-patient-use prefilled pen containing 600 mcg/2.4 mL (250mcg/mL), 60 doses.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Limited data with exenatide in pregnant women are not sufficient to determine a drug-associated risk for major birth defects or miscarriage. There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy (see Clinical Considerations) . Based on animal reproduction studies, there may be risks to the fetus from exposure to exenatide during pregnancy. Exenatide should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Animal reproduction studies identified increased adverse fetal and neonatal outcomes from exposure to exenatide during pregnancy and lactation in association with maternal effects. In mice, exenatide administered during gestation and lactation caused increased neonatal deaths at systemic exposure 3-times the human exposure resulting from the maximum recommended human dose (MRHD) of 20 mcg/day for exenatide (see Data) .
The estimated background risk of major birth defects is 6% to 10% in women with pre-gestational diabetes with an HbA 1c > 7 and has been reported to be as high as 20% to 25% in women with HbA 1c > 10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryofetal risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, preeclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
Data
Animal Data
In studies evaluating reproduction and development in pregnant mice and rabbits, maternal animals were administered exenatide, the active ingredient in exenatide injection, by subcutaneous injection twice a day.
In pregnant mice given 6, 68, 460, or 760 mcg/kg/day exenatide during fetal organogenesis, skeletal variations associated with slowed fetal growth, including changes in number of rib pairs or vertebral ossifications sites, and wavy ribs were observed at 760 mcg/kg/day, a dose that produced maternal toxicity and yielded systemic exposure 390-times the human exposure resulting from the MRHD of exenatide based on AUC comparison.
In pregnant rabbits given 0.2, 2, 22, 156, or 260 mcg/kg/day exenatide during fetal organogenesis, irregular fetal skeletal ossifications were observed at 2 mcg/kg/day, a dose yielding systemic exposure up to 12-times the human exposure from the MRHD of exenatide based on AUC comparison.
In maternal mice given 6, 68, or 760 mcg/kg/day exenatide from gestation day 6 through lactation day 20 (weaning), an increased number of neonatal deaths was observed on postpartum days 2 to 4 in dams given 6 mcg/kg/day, a dose yielding a systemic exposure 3-times the human exposure from the MRHD of exenatide based on AUC comparison.
Lactation
Risk Summary
There is no information regarding the presence of exenatide, in human milk, the effects of exenatide on the breastfed infant, or the effects of exenatide on milk production. Exenatide was present in the milk of lactating mice. However, due to species-specific differences in lactation physiology, the clinical relevance of these data is not clear (see Data) . The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for exenatide and any potential adverse effects on the breastfed child from exenatide or from the underlying maternal condition.
Data
In lactating mice subcutaneously injected twice a day with exenatide, the concentration of exenatide in milk was up to 2.5% of the concentration in maternal plasma.
Pediatric Use
The safety and effectiveness of exenatide have not been established in pediatric patients.
Effectiveness of exenatide was not demonstrated in a randomized, double-blind, placebo-controlled study conducted in 120 pediatric patients (78 received exenatide and 42 received placebo) aged 10 to 17 years with type 2 diabetes mellitus.
Geriatric Use
Population pharmacokinetic analysis of patients ranging from 22 to 73 years of age suggests that age does not influence the pharmacokinetic properties of exenatide [see Clinical Pharmacology (12.3) ] . Exenatide was studied in 282 patients 65 years of age or older and in 16 patients 75 years of age or older. No differences in safety or effectiveness were observed between these patients and younger patients. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection in the elderly based on renal function.
Renal Impairment
Exenatide is not recommended for use in patients with end-stage renal disease or severe renal impairment (creatinine clearance < 30 mL/min) and should be used with caution in patients with renal transplantation. In patients with end-stage renal disease receiving dialysis, single doses of exenatide 5 mcg were not well-tolerated due to gastrointestinal adverse reactions [see Clinical Pharmacology (12.3) ] .
Hepatic Impairment
No pharmacokinetic study has been performed in patients with a diagnosis of acute or chronic hepatic impairment. Because exenatide is cleared primarily by the kidney, hepatic dysfunction is not expected to affect blood concentrations of exenatide [see Clinical Pharmacology (12.3) ] .
CONTRAINDICATIONS
Exenatide injection is contraindicated in patients with:
- A prior severe hypersensitivity reaction to exenatide or to any of the excipients in exenatide injection. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with exenatide injection [see Warnings and Precautions (5.7) ] .
- A history of drug-induced immune-mediated thrombocytopenia from exenatide products. Serious bleeding, which may be fatal, from drug-induced immune-mediated thrombocytopenia has been reported with exenatide use [see Warnings and Precautions (5.8) ].
WARNINGS AND PRECAUTIONS
Acute Pancreatitis
Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with glucagon-like peptide-1 (GLP-1) receptor agonists, including exenatide [see Adverse Reactions (6.2) ]. After initiation of exenatide, observe patients carefully for signs and symptoms of pancreatitis (including persistent severe abdominal pain, sometimes radiating to the back and which may or may not be accompanied by vomiting). If pancreatitis is suspected, discontinue exenatide and initiate appropriate management.
Never Share an Exenatide Injection Pen Between Patients
Exenatide injection pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens.
Hypoglycemia with Concomitant Use of Insulin Secretagogues or Insulin
Patients receiving exenatide in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia including severe hypoglycemia [see Adverse Reactions (6) and Drug Interactions (7) ] .
The risk of hypoglycemia may be lowered by a reduction in the dose of sulfonylurea (or other concomitantly administered insulin secretagogue) or insulin. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia.
Acute Kidney Injury Due to Volume Depletion
There have been post-marketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with GLP-1 receptor agonists, exenatide [see Adverse Reactions (6.2) ]. The majority of the reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea [see Adverse Reactions (6) ].
Monitor renal function in patients reporting adverse reactions to exenatide that could lead to volume depletion, especially during dosage initiation and escalation of exenatide.
Exenatide is not recommended in patients with severe renal impairment (creatinine clearance <30 mL/min) or end-stage renal disease and should be used with caution in patients with renal transplantation [see Use in Specific Populations (8.6) ] .
Severe Gastrointestinal Adverse Reactions
Use of GLP-1 receptor agonists, including exenatide, has been associated with gastrointestinal adverse reactions, sometimes severe [see Adverse Reactions (6) ]. Exenatide is not recommended in patients with severe gastroparesis.
Immunogenicity
Patients may develop antibodies to exenatide following treatment with exenatide. Antibody levels were measured in 90% of subjects in the 30-week, 24-week, and 16-week placebo-controlled studies and the 30-week comparator-controlled study of exenatide. In 3%, 4%, 1% and 1% of these patients, respectively, antibody formation was associated with an attenuated glycemic response. If there is worsening glycemic control or failure to achieve targeted glycemic control, alternative antidiabetic therapy should be considered [see Adverse Reactions (6.1) ] .
Hypersensitivity
There have been post-marketing reports of serious hypersensitivity reactions (e.g., anaphylaxis and angioedema) in patients treated with exenatide. If a hypersensitivity reaction occurs, the patient should discontinue exenatide and other suspect medications and promptly seek medical advice. Inform and closely monitor patients with a history of anaphylaxis or angioedema with another GLP-1 receptor agonist for allergic reactions, because it is unknown whether such patients will be predisposed to anaphylaxis with exenatide [see Adverse Reactions (6.2) ] .
Drug-Induced Thrombocytopenia
Serious bleeding, which may be fatal, from drug-induced immune-mediated thrombocytopenia has been reported in the post-marketing setting with exenatide use. Drug-induced thrombocytopenia is an immune-mediated reaction, with exenatide-dependent anti-platelet antibodies. In the presence of exenatide, these antibodies cause platelet destruction. If drug-induced thrombocytopenia is suspected, discontinue exenatide immediately and do not re-expose the patient to exenatide [see Adverse Reactions (6.2) ] .
Acute Gallbladder Disease
Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and post-marketing. In a clinical study with exenatide, 1.9% of exenatide-treated patients and 1.4% of placebo-treated patients reported an acute event of gallbladder disease, such as cholelithiasis or cholecystitis. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated.
Pulmonary Aspiration During General Anesthesia or Deep Sedation
Exenatide delays gastric emptying [see Clinical Pharmacology (12.2) ] . There have been rare post-marketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations.
Available data are insufficient to inform recommendations to mitigate the risk of pulmonary aspiration during general anesthesia or deep sedation in patients taking exenatide, including whether modifying preoperative fasting recommendations or temporarily discontinuing exenatide could reduce the incidence of retained gastric contents. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking exenatide.
ADVERSE REACTIONS
The following serious adverse reactions are described below or elsewhere in the prescribing information:
- Acute Pancreatitis [see Warnings and Precautions (5.1) ]
- Never Share an Exenatide Pen Between Patients [see Warnings and Precautions (5.2) ]
- Hypoglycemia with Concomitant Use of Insulin Secretagogues or Insulin [see Warnings and Precautions (5.3) ]
- Acute Kidney Injury Due to Volume Depletion [see Warnings and Precautions (5.4) ]
- Severe Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.5) ]
- Immunogenicity [see Warnings and Precautions (5.6) ]
- Hypersensitivity [see Warnings and Precautions (5.7) ]
- Drug-Induced Thrombocytopenia [see Warnings and Precautions (5.8) ]
- Acute Gallbladder Disease [see Warnings and Precautions (5.9) ]
- Pulmonary Aspiration During General Anesthesia or Deep Sedation [see Warnings and Precautions (5.10) ]
Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Hypoglycemia
Table 1 summarizes the incidence and rate of hypoglycemia with exenatide in six placebo-controlled clinical trials.
Table 1: Incidence (%) and Rate of Hypoglycemia when Exenatide was used as Monotherapy or with Concomitant Antidiabetic Therapy in Six Placebo-Controlled Clinical Trials •
| Placebo BID | Exenatide 5 mcg BID | Exenatide 10 mcg BID | |
|---|---|---|---|
Monotherapy (24 Weeks) | |||
N | 77 | 77 | 78 |
% Overall | 1.3% | 5.2% | 3.8% |
Rate (episodes/patient-year) | 0.03 | 0.21 | 0.52 |
% Severe | 0.0% | 0.0% | 0.0% |
With Metformin (30 Weeks) | |||
N | 113 | 110 | 113 |
% Overall | 5.3% | 4.5% | 5.3% |
Rate (episodes/patient-year) | 0.12 | 0.13 | 0.12 |
% Severe | 0.0% | 0.0% | 0.0% |
With a Sulfonylurea (30 Weeks) | |||
N | 123 | 125 | 129 |
% Overall | 3.3% | 14.4% | 35.7% |
Rate (episodes/patient-year) | 0.07 | 0.64 | 1.61 |
% Severe | 0.0% | 0.0% | 0.0% |
With Metformin and a Sulfonylurea (30 Weeks) | |||
N | 247 | 245 | 241 |
% Overall | 12.6% | 19.2% | 27.8% |
Rate (episodes/patient-year) | 0.58 | 0.78 | 1.71 |
% Severe | 0.0% | 0.4% | 0.0% |
With a Thiazolidinedione (16 Weeks) | |||
N | 112 | not evaluated | 121 |
% Overall | 7.1% | not evaluated | 10.7% |
Rate (episodes/patient-years) | 0.56 | not evaluated | 0.98 |
% Severe | 0.0% | not evaluated | 0.0% |
With Insulin Glargine with or without Metformin and/or Thiazolidinedione (30 Weeks) † | |||
N | 122 | not evaluated | 137 |
% Overall | 29.5% | not evaluated | 24.8% |
Rate (episodes/patient-years) | 1.58 | not evaluated | 1.61 |
% Severe | 0.8% | not evaluated | 0.0% |
• A hypoglycemic episode was recorded if a patient reported symptoms of hypoglycemia with or without a blood glucose value consistent with hypoglycemia. Severe hypoglycemia was defined as an event with symptoms consistent with hypoglycemia requiring the assistance of another person and associated with either a documented blood glucose value < 54 mg/dL or prompt recovery after treatment for hypoglycemia. † When exenatide was initiated in combination with insulin glargine, the dose of insulin glargine was decreased by 20% in patients with an HbA 1c ≤ 8.0% to minimize the risk of hypoglycemia. See Table 10 for insulin dose titration algorithm. N = number of Intent-to-Treat subjects in each treatment group. | |||
Immunogenicity
Antibodies were assessed in 90% of subjects in the 30-week, 24-week, and 16-week studies of exenatide. In the 30-week controlled trials of exenatide add-on to metformin and/or sulfonylurea, antibodies were assessed at 2- to 6-week intervals. The mean antibody titer peaked at Week 6 and was reduced by 55% by Week 30. Three hundred and sixty patients (38%) had low titer antibodies (< 625) to exenatide at 30 weeks. The level of glycemic control (HbA 1c ) in these patients was generally comparable to that observed in the 534 patients (56%) without antibody titers. An additional 59 patients (6%) had higher titer antibodies (≥ 625) at 30 weeks. Of these patients, 32 (3% overall) had an attenuated glycemic response to exenatide; the remaining 27 (3% overall) had a glycemic response comparable to that of patients without antibodies.
In the 16-week trial of exenatide add-on to thiazolidinediones, with or without metformin, 36 patients (31%) had low titer antibodies to exenatide at 16 weeks. The level of glycemic control in these patients was generally comparable to that observed in the 69 patients (60%) without antibody titer. An additional 10 patients (9%) had higher titer antibodies at 16 weeks. Of these patients, 4 (4% overall) had an attenuated glycemic response to exenatide; the remaining 6 (5% overall) had a glycemic response comparable to that of patients without antibodies.
In the 24-week trial of exenatide used as monotherapy, 40 patients (28%) had low titer antibodies to exenatide at 24 weeks. The level of glycemic control in these patients was generally comparable to that observed in the 101 patients (70%) without antibody titers. An additional 3 patients (2%) had higher titer antibodies at 24 weeks. Of these patients, 1 (1% overall) had an attenuated glycemic response to exenatide; the remaining 2 (1% overall) had a glycemic response comparable to that of patients without antibodies.
Antibodies to exenatide were not assessed in the 30-week placebo-controlled trial of exenatide used in combination with insulin glargine.
In the 30-week comparator-controlled trial of exenatide used in combination with insulin glargine and metformin, 60 patients (20%) had low titer antibodies to exenatide at 30 weeks. The level of glycemic control in these patients was generally comparable to that observed in the 234 patients (77%) without antibody titers. An additional 10 patients (3%) had higher titer antibodies at 30 weeks. Of these patients, 2 (1% overall) had an attenuated glycemic response to exenatide; the remaining 8 (3% overall) had a glycemic response comparable to that of patients without antibodies.
Two hundred and ten patients with antibodies to exenatide in the exenatide clinical trials were tested for the presence of cross-reactive antibodies to GLP-1 and/or glucagon. No treatment-emergent cross-reactive antibodies were observed across the range of titers.
Other Adverse Reactions
Monotherapy
For the 24-week placebo-controlled study of exenatide used as a monotherapy, Table 2 summarizes adverse reactions (excluding hypoglycemia) occurring with an incidence ≥ 2% and occurring more frequently in exenatide-treated patients compared with placebo-treated patients.
Table 2: Treatment-Emergent Adverse Reactions ≥ 2% Incidence with Exenatide used as Monotherapy (excluding Hypoglycemia) •
Monotherapy | Placebo BID N=77 % | All Exenatide BID N=155 % |
Nausea | 0 | 8 |
Vomiting | 0 | 4 |
Dyspepsia | 0 | 3 |
• In a 24-week placebo-controlled trial. BID = twice daily. | ||
Adverse reactions reported in ≥ 1.0% to < 2.0% of patients receiving exenatide and reported more frequently than with placebo included decreased appetite, diarrhea and dizziness. The most frequently reported adverse reaction associated with exenatide, nausea, occurred in a dose-dependent fashion.
Two of the 155 patients treated with exenatide withdrew due to adverse reactions of headache and nausea. No placebo-treated patients withdrew due to adverse reactions.
Cholelithiasis and cholecystitis
In a clinical study with exenatide, 1.9% of exenatide-treated patients and 1.4% of placebo-treated patients reported an acute event of gallbladder disease, such as cholelithiasis or cholecystitis.
Combination Therapy
Add-On to Metformin and/or Sulfonylurea
In the three 30-week controlled trials of exenatide add-on to metformin and/or sulfonylurea, adverse reactions (excluding hypoglycemia) with an incidence ≥ 2% and occurring more frequently in exenatide-treated patients compared with placebo-treated patients are summarized in Table 3.
Table 3: Treatment-Emergent Adverse Reactions ≥ 2% Incidence and Greater Incidence with Exenatide Treatment used with Metformin and/or a Sulfonylurea (excluding Hypoglycemia) •
Placebo BID N=483 % | All Exenatide BID N=963 % | |
Nausea | 18 | 44 |
Vomiting | 4 | 13 |
Diarrhea | 6 | 13 |
Feeling Jittery | 4 | 9 |
Dizziness | 6 | 9 |
Headache | 6 | 9 |
Dyspepsia | 3 | 6 |
Asthenia | 2 | 4 |
Gastroesophageal Reflux Disease | 1 | 3 |
Hyperhidrosis | 1 | 3 |
• In three 30-week placebo-controlled clinical trials. BID = twice daily. | ||
Adverse reactions reported in ≥ 1.0% to < 2.0% of patients receiving exenatide and reported more frequently than with placebo included decreased appetite. Nausea was the most frequently reported adverse reaction and occurred in a dose-dependent fashion. With continued therapy, the frequency and severity decreased over time in most of the patients who initially experienced nausea. Patients in the long-term uncontrolled open-label extension studies at 52 weeks reported no new types of adverse reactions than those observed in the 30-week controlled trials.
The most common adverse reactions leading to withdrawal for exenatide-treated patients were nausea (3% of patients) and vomiting (1%). For placebo-treated patients, < 1% withdrew due to nausea and none due to vomiting.
Add-On to Thiazolidinedione with or without Metformin
For the 16-week placebo-controlled study of exenatide add-on to a thiazolidinedione, with or without metformin, Table 4 summarizes the adverse reactions (excluding hypoglycemia) with an incidence of ≥ 2% and occurring more frequently in exenatide-treated patients compared with placebo-treated patients.
Table 4: Treatment-Emergent Adverse Reactions ≥ 2% Incidence with Exenatide used with a Thiazolidinedione (TZD), with or without Metformin (MET) (excluding Hypoglycemia) •
With a TZD or TZD/MET | Placebo N=112 % | All Exenatide BID N=121 % |
Nausea | 15 | 40 |
Vomiting | 1 | 13 |
Dyspepsia | 1 | 7 |
Diarrhea | 3 | 6 |
Gastroesophageal Reflux Disease | 0 | 3 |
• In a 16-week placebo-controlled clinical trial. BID = twice daily. | ||
Adverse reactions reported in ≥ 1.0% to < 2.0% of patients receiving exenatide and reported more frequently than with placebo included decreased appetite. Chills (n=4) and injection-site reactions (n=2) occurred only in exenatide-treated patients. The two patients who reported an injection-site reaction had high titers of antibodies to exenatide. Two serious adverse events (chest pain and chronic hypersensitivity pneumonitis) were reported in the exenatide arm. No serious adverse events were reported in the placebo arm.
The most common adverse reactions leading to withdrawal for exenatide-treated patients were nausea (9%) and vomiting (5%). For placebo-treated patients, < 1% withdrew due to nausea.
Add-On to Insulin Glargine with or without Metformin and/or Thiazolidinedione (Placebo-Controlled)
For the 30-week placebo-controlled study of exenatide as add-on to insulin glargine with or without oral antihyperglycemic medications, Table 5 summarizes adverse reactions (excluding hypoglycemia) occurring with an incidence ≥ 2% and occurring more frequently in exenatide-treated patients compared with placebo-treated patients.
Table 5: Treatment-Emergent Adverse Reactions ≥ 2% Incidence with Exenatide used with Insulin Glargine with or without Oral Antihyperglycemic Medications (excluding Hypoglycemia) •
With Insulin Glargine | Placebo N=122 % | All Exenatide BID N=137 % |
Nausea | 8 | 41 |
Vomiting | 4 | 18 |
Diarrhea | 8 | 18 |
Headache | 4 | 14 |
Constipation | 2 | 10 |
Dyspepsia | 2 | 7 |
Asthenia | 1 | 5 |
Abdominal Distension | 1 | 4 |
Decreased Appetite | 0 | 3 |
Flatulence | 1 | 2 |
Gastroesophageal Reflux Disease | 1 | 2 |
• In a 30-week placebo-controlled clinical trial. BID = twice daily. | ||
The most frequently reported adverse reactions leading to withdrawal for exenatide-treated patients were nausea (5.1%) and vomiting (2.9%). No placebo-treated patients withdrew due to nausea or vomiting.
Post-marketing Experience
The following additional adverse reactions have been reported during post-approval use of exenatide or other formulations of exenatide. Because these events are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood : Drug-induced thrombocytopenia.
Drug Interactions: International normalized ratio (INR) increased with concomitant warfarin use sometimes associated with bleeding [see Drug Interactions (7) ] .
Gastrointestinal: Nausea, vomiting and/or diarrhea resulting in dehydration; abdominal distension, abdominal pain, eructation, constipation, flatulence, ileus, acute pancreatitis, hemorrhagic and necrotizing pancreatitis sometimes resulting in death.
Hepatobiliary: Cholecystitis, cholelithiasis requiring cholecystectomy.
Hypersensitivity: Injection-site reactions, generalized pruritus and/or urticaria, macular or papular rash, angioedema, anaphylactic reaction.
Neurologic: Dysgeusia; somnolence, dysesthesia.
Pulmonary: Pulmonary aspiration has occurred in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation.
Renal: Altered renal function, including increased serum creatinine, renal impairment, worsened chronic renal failure or acute renal failure (sometimes requiring hemodialysis), kidney transplant and kidney transplant dysfunction.
Skin and Subcutaneous Tissue: Alopecia.
DRUG INTERACTIONS
Table 6: Clinically Relevant Interactions with Exenatide
Concomitant Use of Insulin Secretagogues or Insulin | |
Clinical Impact | Exenatide promotes insulin release from pancreatic beta-cells in the presence of elevated glucose concentrations. The risk of hypoglycemia is increased when exenatide is used in combination with insulin secretagogues (e.g., sulfonylureas) or insulin [see Warnings and Precautions (5.3) and Adverse Reactions (6) ] . |
Intervention | When initiating exenatide, consider reducing the dose of concomitantly administered insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
Warfarin | |
Clinical Impact | In a drug interaction study, exenatide did not have a significant effect on INR [see Clinical Pharmacology (12.3) ] . There have been post-marketing reports for exenatide of increased INR with concomitant use of warfarin, sometimes associated with bleeding [see Adverse Reactions (6.2) ] . |
Intervention | In patients taking warfarin, the prothrombin time should be monitored more frequently after initiation or alteration of exenatide therapy. Once a stable prothrombin time has been documented, the prothrombin time can be monitored at the intervals recommended for patients taking warfarin. |
Orally Administered Drugs (e.g., acetaminophen) | |
Clinical Impact | Exenatide slows gastric emptying. Therefore, exenatide has the potential to reduce the rate of absorption of orally administered drugs [see Clinical Pharmacology (12.3) ]. |
Intervention | Use caution when administering oral medications with exenatide where a slower rate of oral absorption may be clinically meaningful. For oral medications that are dependent on threshold concentrations for efficacy, such as contraceptives and antibiotics, patients should be advised to take those drugs at least 1 hour before exenatide injection. If such drugs are to be administered with food, patients should be advised to take them with a meal or snack when exenatide is not administered [see Clinical Pharmacology (12.3) ] . |
DESCRIPTION
Exenatide is a synthetic peptide, GLP-1 receptor agonist, that was originally identified in the lizard Heloderma suspectum .
Exenatide is a 39-amino acid peptide amide. Exenatide has the empirical formula C 184 H 282 N 50 O 60 S and molecular weight of 4186.6 Daltons. The amino acid sequence for exenatide is shown below.
H-His-Gly-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Leu-Ser-Lys-Gln-Met-Glu-Glu-Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-Trp-Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser-Gly-Ala-Pro-Pro-Pro-Ser-NH 2
Exenatide injection, USP is supplied for subcutaneous administration as a sterile, preserved isotonic solution in a glass cartridge that has been assembled in a pen-injector (pen). Each milliliter (mL) contains 250 micrograms (mcg) synthetic exenatide, USP; 2.2 mg metacresol as an antimicrobial preservative; mannitol as a tonicity-adjusting agent; and glacial acetic acid and sodium acetate trihydrate in water for injection as a buffering solution at pH 4.5. Two prefilled pens are available to deliver unit doses of 5 mcg per dose or 10 mcg per dose. Each prefilled pen will deliver 60 doses to provide for 30 days of twice daily administration (BID). Each prefilled device is filled with volume to allow delivery of 1.2 mL or 2.4 mL. Each device contains additional volume to allow for troubleshooting the device 4 times.
CLINICAL PHARMACOLOGY
Mechanism of Action
Incretins, such as glucagon-like peptide-1 (GLP-1), enhance glucose-dependent insulin secretion and exhibit other antihyperglycemic actions following their release into the circulation from the gut. Exenatide is a GLP-1 receptor agonist that enhances glucose-dependent insulin secretion by the pancreatic beta-cell, suppresses inappropriately elevated glucagon secretion, and slows gastric emptying.
The amino acid sequence of exenatide partially overlaps that of human GLP-1. Exenatide has been shown to bind and activate the human GLP-1 receptor in vitro . This leads to an increase in both glucose-dependent synthesis of insulin, and in vivo secretion of insulin from pancreatic beta cells, by mechanisms involving cyclic AMP and/or other intracellular signaling pathways.
Exenatide improves glycemic control by reducing fasting and postprandial glucose concentrations in patients with type 2 diabetes through the actions described below.
Pharmacodynamics
Glucose-Dependent Insulin Secretion
Exenatide has acute effects on pancreatic beta-cell responsiveness to glucose leading to insulin release predominantly in the presence of elevated glucose concentrations. This insulin secretion subsides as blood glucose concentrations decrease and approach euglycemia. However, exenatide does not impair the normal glucagon response to hypoglycemia.
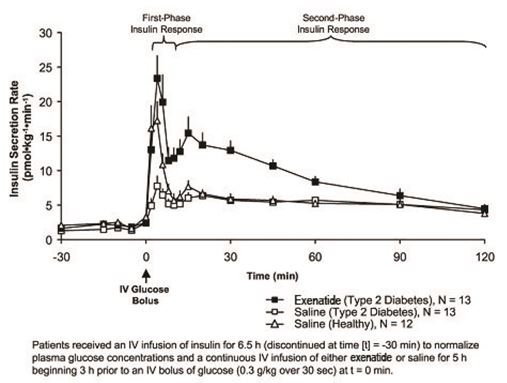
First-Phase Insulin Response
In healthy individuals, robust insulin secretion occurs during the first 10 minutes following intravenous (IV) glucose administration. This secretion, known as the “first-phase insulin response,” is characteristically absent in patients with type 2 diabetes. The loss of the first-phase insulin response is an early beta-cell defect in type 2 diabetes. Administration of exenatide at therapeutic plasma concentrations restored first-phase insulin response to an IV bolus of glucose in patients with type 2 diabetes (Figure 1). Both first-phase insulin secretion and second-phase insulin secretion were significantly increased in patients with type 2 diabetes treated with exenatide compared with saline (p < 0.001 for both).
Figure 1: Mean (+SEM) Insulin Secretion Rate during Infusion of Exenatide or Saline in Patients with Type 2 Diabetes and during Infusion of Saline in Healthy Subjects
Glucagon Secretion
In patients with type 2 diabetes, exenatide moderates glucagon secretion and lowers serum glucagon concentrations during periods of hyperglycemia. Lower glucagon concentrations lead to decreased hepatic glucose output and decreased insulin demand.
Gastric Emptying
Exenatide slows gastric emptying, thereby reducing the rate at which meal-derived glucose appears in the circulation.
Food Intake
In both animals and humans, administration of exenatide has been shown to reduce food intake.
Postprandial Glucose
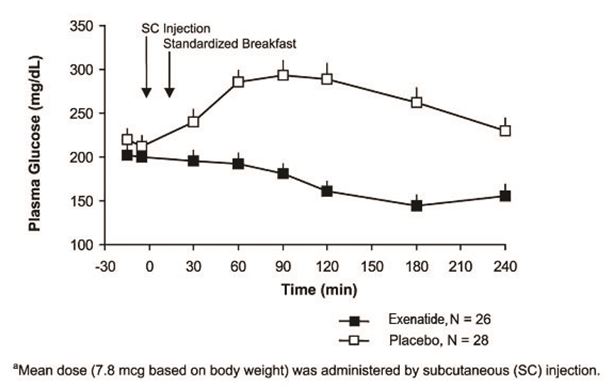
In patients with type 2 diabetes, exenatide reduces postprandial plasma glucose concentrations (Figure 2).
Figure 2: Mean (+SEM) Postprandial Plasma Glucose Concentrations on Day 1 of Exenatide a Treatment in Patients with Type 2 Diabetes Treated with Metformin, a Sulfonylurea, or Both (N = 54)
Fasting Glucose
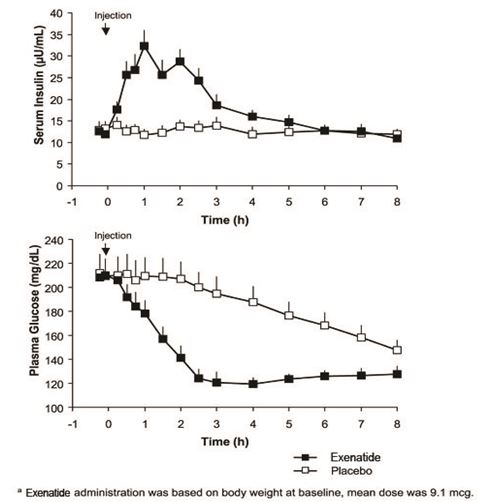
In a single-dose crossover study in patients with type 2 diabetes and fasting hyperglycemia, immediate insulin release followed injection of exenatide. Plasma glucose concentrations were significantly reduced with exenatide compared with placebo (Figure 3).
Figure 3: Mean (+SEM) Serum Insulin and Plasma Glucose Concentrations Following a One-Time Injection of Exenatide a or Placebo in Fasting Patients with Type 2 Diabetes (N = 12)
Cardiac Electrophysiology
The effect of exenatide 10 mcg subcutaneously on QTc interval was evaluated in a randomized, placebo-, and active-controlled (moxifloxacin 400 mg) crossover thorough QTc study in 62 healthy subjects. In this study with demonstrated ability to detect small effects, the upper bound of the 90% confidence interval for the largest placebo-adjusted, baseline-corrected QTc was below 10 msec. Thus, exenatide (10 mcg single-dose) was not associated with clinically meaningful prolongation of the QTc interval.
Pharmacokinetics
Absorption
Following SC administration to patients with type 2 diabetes, exenatide reaches median peak plasma concentrations in 2.1 hours. The mean peak exenatide concentration (C max ) was 211 pg/mL and overall mean area under the time-concentration curve (AUC 0-inf ) was 1,036 pg•h/mL following SC administration of a 10-mcg dose of exenatide. Exenatide exposure (AUC) increased proportionally over the therapeutic dose range of 5 mcg to 10 mcg. The C max values increased less than proportionally over the same range. Similar exposure is achieved with SC administration of exenatide in the abdomen, thigh, or upper arm.
Distribution
The mean apparent volume of distribution of exenatide following SC administration of a single-dose of exenatide is 28.3 L.
Metabolism and Elimination
Nonclinical studies have shown that exenatide is predominantly eliminated by glomerular filtration with subsequent proteolytic degradation. The mean apparent clearance of exenatide in humans is 9.1 L/hour and the mean terminal half-life is 2.4 hours. These pharmacokinetic characteristics of exenatide are independent of the dose. In most individuals, exenatide concentrations are measurable for approximately 10 hours post-dose.
Drug Interactions
Acetaminophen
When 1,000 mg acetaminophen elixir was given with 10 mcg exenatide (0 hour) and 1 hour, 2 hours and 4 hours after exenatide injection, acetaminophen AUCs were decreased by 21%, 23%, 24% and 14%, respectively; C max was decreased by 37%, 56%, 54% and 41%, respectively; T max was increased from 0.6 hour in the control period to 0.9 hour, 4.2 hours, 3.3 hours and 1.6 hours, respectively. Acetaminophen AUC, C max and T max were not significantly changed when acetaminophen was given 1 hour before exenatide injection.
Digoxin
Administration of repeated doses of exenatide (10 mcg BID) 30 minutes before oral digoxin (0.25 mg once daily) decreased the C max of digoxin by 17% and delayed the T max of digoxin by approximately 2.5 hours; however, the overall steady-state pharmacokinetic exposure (e.g., AUC) of digoxin was not changed.
Lovastatin
Administration of exenatide (10 mcg BID) 30 minutes before a single oral dose of lovastatin (40 mg) decreased the AUC and C max of lovastatin by approximately 40% and 28%, respectively, and delayed the T max by about 4 hours compared with lovastatin administered alone. In the 30-week controlled clinical trials of exenatide, the use of exenatide in patients already receiving HMG CoA reductase inhibitors was not associated with consistent changes in lipid profiles compared to baseline.
Lisinopril
In patients with mild to moderate hypertension stabilized on lisinopril (5 mg/day to 20 mg/day), exenatide (10 mcg BID) did not alter steady-state C max or AUC of lisinopril. Lisinopril steady-state T max was delayed by 2 hours. There were no changes in 24-hour mean systolic and diastolic blood pressure.
Oral Contraceptives
The effect of exenatide (10 mcg BID) on single and on multiple-doses of a combination oral contraceptive (30 mcg ethinyl estradiol plus 150 mcg levonorgestrel) was studied in healthy female subjects. Repeated daily doses of the oral contraceptive (OC) given 30 minutes after exenatide administration decreased the C max of ethinyl estradiol and levonorgestrel by 45% and 27%, respectively and delayed the T max of ethinyl estradiol and levonorgestrel by 3.0 hours and 3.5 hours, respectively, as compared to the oral contraceptive administered alone. Administration of repeated daily doses of the OC one hour prior to exenatide administration decreased the mean C max of ethinyl estradiol by 15% but the mean C max of levonorgestrel was not significantly changed as compared to when the OC was given alone. Exenatide did not alter the mean trough concentrations of levonorgestrel after repeated daily dosing of the oral contraceptive for both regimens. However, the mean trough concentration of ethinyl estradiol was increased by 20% when the OC was administered 30 minutes after exenatide administration injection as compared to when the OC was given alone. The effect of exenatide on OC pharmacokinetics is confounded by the possible food effect on OC in this study. Therefore, OC products should be administered at least one hour prior to exenatide injection.
Warfarin
Administration of warfarin (25 mg) 35 minutes after repeated doses of exenatide (5 mcg BID on Days 1 to 2 and 10 mcg BID on Days 3 to 9) in healthy volunteers delayed warfarin T max by approximately 2 hours. No clinically relevant effects on C max or AUC of S - and R -enantiomers of warfarin were observed. Exenatide did not significantly alter the pharmacodynamic properties (e.g., international normalized ratio) of warfarin [see Drug Interactions (7) ] .
Specific Populations
Patients with Renal Impairment
Pharmacokinetics of exenatide was studied in subjects with normal, mild, or moderate renal impairment and subjects with end-stage renal disease. In subjects with mild to moderate renal impairment (creatinine clearance 30 mL/min to 80 mL/min), exenatide exposure was similar to that of subjects with normal renal function. However, in subjects with end-stage renal disease receiving dialysis, mean exenatide exposure increased by 3.37-fold compared to that of subjects with normal renal function [see Use in Specific Populations (8.6) ] .
Patients with Hepatic Impairment
No pharmacokinetic study has been performed in patients with a diagnosis of acute or chronic hepatic impairment [see Use in Specific Populations (8.7) ] .
Age
Population pharmacokinetic analysis of patients ranging from 22 to 73 years of age suggests that age does not influence the pharmacokinetic properties of exenatide [see Use in Specific Population (8.5) ] .
Male and Female Patients
Population pharmacokinetic analysis of male and female patients suggests that gender does not influence the distribution and elimination of exenatide.
Racial or Ethnic Groups
Population pharmacokinetic analysis of samples from White, Hispanic or Latino ethnicity, Asian and Black or African American patients suggests that race has no significant influence on the pharmacokinetics of exenatide.
Body Mass Index
Population pharmacokinetic analysis of patients with body mass indices (BMI) ≥ 30 kg/m 2 and < 30 kg/m 2 suggests that BMI has no significant effect on the pharmacokinetics of exenatide.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
A 104-week carcinogenicity study was conducted in male and female rats at doses of 18, 70, or 250 mcg/kg/day administered by bolus SC injection. Benign thyroid C-cell adenomas were observed in female rats at all exenatide doses. The incidences in female rats were 8% and 5% in the two control groups and 14%, 11% and 23% in the low-, medium- and high-dose groups with systemic exposures of 5-, 22-, and 130-times, respectively, the human exposure resulting from the maximum recommended dose of 20 mcg/day, based on plasma area under the curve (AUC).
In a 104-week carcinogenicity study in mice at doses of 18, 70, or 250 mcg/kg/day administered by bolus SC injection, no evidence of tumors was observed at doses up to 250 mcg/kg/day, a systemic exposure up to 95-times the human exposure resulting from the maximum recommended dose of 20 mcg/day, based on AUC.
Exenatide was not mutagenic or clastogenic, with or without metabolic activation, in the Ames bacterial mutagenicity assay or chromosomal aberration assay in Chinese hamster ovary cells. Exenatide was negative in the in vivo mouse micronucleus assay.
In mouse fertility studies with SC doses of 6, 68, or 760 mcg/kg/day, males were treated for 4 weeks prior to and throughout mating, and females were treated 2 weeks prior to mating and throughout mating until gestation day 7. No adverse effect on fertility was observed at 760 mcg/kg/day, a systemic exposure 390-times the human exposure resulting from the maximum recommended dose of 20 mcg/day, based on AUC.
CLINICAL STUDIES
Exenatide has been studied as monotherapy and in combination with metformin, a sulfonylurea, a thiazolidinedione, a combination of metformin and a sulfonylurea, a combination of metformin and a thiazolidinedione, or in combination with insulin glargine with or without metformin and/or thiazolidinedione.
Monotherapy
In a randomized, double-blind, placebo-controlled trial of 24 weeks duration, exenatide 5 mcg BID (n=77), exenatide 10 mcg BID (n=78), or placebo BID (n=77) was used as monotherapy in patients with entry HbA 1c ranging from 6.5% to 10%. All patients assigned to exenatide initially received 5 mcg BID for 4 weeks. After 4 weeks, those patients either continued to receive exenatide 5 mcg BID or had their dose increased to 10 mcg BID. Patients assigned to placebo received placebo BID throughout the trial. Exenatide or placebo was injected subcutaneously before the morning and evening meals. The majority of patients (68%) were White, 26% were West Asian, 3% were Hispanic or Latino ethnicity, 3% were Black or African American and 0.4% were of East Asian ethnicity.
The primary endpoint was the change in HbA 1c from baseline to Week 24 (or the last value at time of early discontinuation). Compared to placebo, exenatide 5 mcg BID and 10 mcg BID resulted in statistically significant reductions in HbA 1c from baseline at Week 24 (Table 7).
Table 7: Results of 24-Week Placebo-Controlled Trial of Exenatide used as Monotherapy
Placebo BID | Exenatide 5 mcg BID | Exenatide 10 mcg • BID | |
Intent-to-Treat Population (N) | 77 | 77 | 78 |
HbA 1c (%), Mean | |||
Baseline | 7.8 | 7.9 | 7.8 |
Change at Week 24 † | -0.2 | -0.7 | -0.9 |
Difference from placebo † (95% CI) | -0.5 [-0.9, -0.2] ‡ | -0.7 [-1.0, -0.3] | |
Proportion Achieving HbA 1c < 7% | 38% | 48% | 53% |
Body Weight (kg), Mean | |||
Baseline | 86.1 | 85.1 | 86.2 |
Change at Week 24 † | -1.5 | -2.7 | -2.9 |
Difference from placebo † (95% CI) | -1.3 [-2.3, -0.2] | -1.5 [-2.5, -0.4] | |
Fasting Serum Glucose § (mg/dL), Mean | |||
Baseline | 159 | 166 | 155 |
Change at Week 24 † | -5 | -17 | -19 |
Difference from placebo † (95% CI) | -12 [-23.2, -1.3] | -14 [-24.5, -2.5] | |
• Exenatide 5 mcg twice daily (BID) for 1 month followed by 10 mcg BID for 5 months before the morning and evening meals. † Least squares means are adjusted for screening HbA 1c strata and baseline value of the dependent variable. ‡ p < 0.01, treatment vs. placebo. § Measured using the hexokinase-based glucose method. BID = twice daily. | |||
On average, there were no adverse effects of exenatide on blood pressure or lipids.
Combination Therapy with Oral Antihyperglycemic Medicines
Three 30-week, double-blind, placebo-controlled trials were conducted to evaluate the safety and efficacy of exenatide in patients with type 2 diabetes whose glycemic control was inadequate with metformin alone, a sulfonylurea alone, or metformin in combination with a sulfonylurea. In addition, a 16-week, placebo-controlled trial was conducted where exenatide was added to existing thiazolidinedione (pioglitazone or rosiglitazone) treatment, with or without metformin, in patients with type 2 diabetes with inadequate glycemic control.
In the 30-week trials, after a 4-week placebo lead-in period, patients were randomly assigned to receive exenatide 5 mcg BID, exenatide 10 mcg BID, or placebo BID before the morning and evening meals, in addition to their existing oral antidiabetic agent. All patients assigned to exenatide initially received 5 mcg BID for 4 weeks. After 4 weeks, those patients either continued to receive exenatide 5 mcg BID or had their dose increased to 10 mcg BID. Patients assigned to placebo received placebo BID throughout the study. A total of 1,446 patients were randomized in the three 30-week trials: 991 (69%) were White, 224 (16%) were Hispanic or Latino and 174 (12%) were of Black of African American ethnicity. Mean HbA 1c values at baseline for the trials ranged from 8.2% to 8.7%.
In the placebo-controlled trial of 16 weeks duration, exenatide (n=121) or placebo (n=112) was added to existing thiazolidinedione (pioglitazone or rosiglitazone) treatment, with or without metformin. Randomization to exenatide or placebo was stratified based on whether the patients were receiving metformin. Exenatide treatment was initiated at a dose of 5 mcg BID for 4 weeks then increased to 10 mcg BID for 12 more weeks. Patients assigned to placebo received placebo BID throughout the study. Exenatide or placebo was injected subcutaneously before the morning and evening meals. In this trial, 79% of patients were taking a thiazolidinedione and metformin and 21% were taking a thiazolidinedione alone. The majority of patients (84%) were White, 8% were Hispanic or Latino ethnicity and 3% were of Black or African American ethnicity. The mean baseline HbA 1c values were 7.9% for exenatide and placebo.
The primary endpoint in each study was the mean change in HbA 1c from baseline to study end (or early discontinuation). Table 8 summarizes the study results for the 30- and 16-week clinical trials.
Table 8: Results of 30-Week and 16-Week Placebo-Controlled Trials of Exenatide used in Combination with Oral Antidiabetic Agents
Placebo BID | Exenatide 5 mcg BID | Exenatide 10 mcg • BID | |
In Combination with Metformin (30 Weeks) | |||
Intent-to-Treat Population (N) | 113 | 110 | 113 |
HbA 1c (%), Mean | |||
Baseline | 8.2 | 8.3 | 8.2 |
Change at Week 30 † | 0.0 | -0.5 | -0.9 |
Difference from placebo † (95% CI) | -0.5 [-0.7, -0.2] ‡ | -0.9 [-1.1, -0.6] ‡ | |
Proportion Achieving HbA 1c < 7% | 12% | 32% | 40% |
Body Weight (kg), Mean | |||
Baseline | 99.9 | 100.0 | 100.9 |
Change at Week 30 † | -0.2 | -1.3 | -2.6 |
Difference from placebo † (95% CI) | -1.1 [-2.2, -0.0] | -2.4 [-3.5, -1.3] | |
Fasting Plasma Glucose § (mg/dL), Mean | |||
Baseline | 169 | 176 | 168 |
Change at Week 30 † | +14 | -5 | -10 |
Difference from placebo † (95% CI) | -20 [-32, -7] | -24 [-37, -12] | |
In Combination with a Sulfonylurea (30 Weeks) | |||
Intent-to-Treat Population (N) | 123 | 125 | 129 |
HbA 1c (%), Mean | |||
Baseline | 8.7 | 8.5 | 8.6 |
Change at Week 30 † | +0.1 | -0.5 | -0.9 |
Difference from placebo † (95% CI) | -0.6 [-0.9, -0.3] ‡ | -1.0 [-1.3, -0.7] ‡ | |
Proportion Achieving HbA 1c < 7% | 10% | 25% | 36% |
Body Weight (kg), Mean | |||
Baseline | 99.1 | 94.9 | 95.2 |
Change at Week 30 † | -0.8 | -1.1 | -1.6 |
Difference from placebo † (95% CI) | -0.3 [-1.1, 0.6] | -0.9 [-1.7, -0.0] | |
Fasting Plasma Glucose § (mg/dL), Mean | |||
Baseline | 194 | 180 | 178 |
Change at Week 30 † | +6 | -5 | -11 |
Difference from placebo † (95% CI) | -11 [-25, 3] | -17 [-30, -3] | |
In Combination with Metformin and a Sulfonylurea (30 Weeks) | |||
Intent-to-Treat Population (N) | 247 | 245 | 241 |
HbA 1c (%), Mean | |||
Baseline | 8.5 | 8.5 | 8.5 |
Change at Week 30 † | +0.1 | -0.7 | -0.9 |
Difference from placebo † (95% CI) | -0.8 [-1.0, -0.6] ‡ | -1.0 [-1.2, -0.8] ‡ | |
Proportion Achieving HbA 1c < 7% | 8% | 25% | 31% |
Body Weight (kg), Mean | |||
Baseline | 99.1 | 96.9 | 98.4 |
Change at Week 30 † | -0.9 | -1.6 | -1.6 |
Difference from placebo † (95% CI) | -0.7 [-1.2, -0.2] | -0.7 [-1.3, -0.2] | |
Fasting Plasma Glucose § (mg/dL), Mean | |||
Baseline | 181 | 182 | 178 |
Change at Week 30 † | +13 | -11 | -12 |
Difference from placebo † (95% CI) | -24 [-33, -15] | -25 [-34, -16] | |
In Combination with a Thiazolidinedione or a Thiazolidinedione plus Metformin (16 Weeks) | |||
Intent-to-Treat Population (N) | 112 | Dose not studied | 121 |
HbA 1c (%), Mean | |||
Baseline | 7.9 | Dose not studied | 7.9 |
Change at Week 16 † | +0.1 | Dose not studied | -0.7 |
Difference from placebo † (95% CI) | Dose not studied | -0.9 [-1.1, -0.7] ‡ | |
Proportion Achieving HbA 1c < 7% | 15% | Dose not studied | 51% |
Body Weight (kg), Mean | |||
Baseline | 96.8 | Dose not studied | 97.5 |
Change at Week 16 † | 0.0 | Dose not studied | -1.5 |
Difference from placebo † (95% CI) | Dose not studied | -1.5 [-2.2, -0.7] | |
Fasting Serum Glucose § (mg/dL), Mean | |||
Baseline | 159 | Dose not studied | 164 |
Change at Week 16 † | +4 | Dose not studied | -21 |
Difference from placebo † (95% CI) | Dose not studied | -25 [-33, -16] | |
• Exenatide 5 mcg twice daily for 1 month followed by 10 mcg BID for 6 months for the 30-week trials or 10 mcg BID for 3 months in the 16-week trial before the morning and evening meals. † Least squares means are adjusted for baseline HbA 1c strata or value, investigator site, baseline value of the dependent variable (if applicable) and background antihyperglycemic therapy (if applicable). ‡ p < 0.01, treatment vs. placebo. § Measured using the hexokinase-based glucose method. BID = twice daily. | |||
HbA 1c
The addition of exenatide to a regimen of metformin, a sulfonylurea, or both, resulted in statistically significant reductions from baseline in HbA 1c compared with patients receiving placebo added to these agents in the three controlled trials (Table 8).
In the 16-week trial of exenatide add-on to thiazolidinediones, with or without metformin, exenatide resulted in statistically significant reductions from baseline in HbA 1c compared with patients receiving placebo (Table 8).
Postprandial Glucose
Postprandial glucose was measured after a mixed meal tolerance test in 9.5% of patients participating in the 30-week add-on to metformin, add-on to sulfonylurea, and add-on to metformin in combination with sulfonylurea clinical trials. In this pooled subset of patients, exenatide reduced postprandial plasma glucose concentrations in a dose-dependent manner. The mean (SD) change in 2-hour postprandial glucose concentration following administration of exenatide at Week 30 relative to baseline was −63 (65) mg/dL for 5 mcg BID (n=42), −71 (73) mg/dL for 10 mcg BID (n=52) and +11 (69) mg/dL for placebo BID (n=44).
Combination with Insulin Glargine
30-Week Placebo-Controlled Trial
A 30-week, double-blind, placebo-controlled trial was conducted to evaluate the efficacy and safety of exenatide (n=137) versus placebo (n=122) when added to titrated insulin glargine, with or without metformin and/or thiazolidinedione, in patients with type 2 diabetes with inadequate glycemic control.
All patients assigned to exenatide initially received 5 mcg BID for 4 weeks. After 4 weeks, those patients assigned to exenatide had their dose increased to 10 mcg BID. Patients assigned to placebo received placebo BID throughout the trial. Exenatide or placebo was injected subcutaneously before the morning and evening meals. Patients with an HbA 1c ≤ 8.0% decreased their pre-study dose of insulin glargine by 20% and patients with an HbA 1c ≥ 8.1% maintained their current dose of insulin glargine. Five weeks after initiating randomized treatment, insulin doses were titrated with guidance from the investigator toward predefined fasting glucose targets according to the dose titration algorithm provided in Table 9. The majority of patients (78%) were White, 10% were American Indian or Alaska Native, 9% were Black or African American, 3% were Asian, and 0.8% were of multiple origins.
The primary endpoint was the change in HbA 1c from baseline to Week 30. Compared to placebo, exenatide 10 mcg BID resulted in statistically significant reductions in HbA 1c from baseline at Week 30 (Table 9) in patients receiving titrated insulin glargine.
Table 9: 30-Week Placebo-Controlled Trial of Exenatide Used in Combination with Insulin Glargine with or without Metformin and/or Thiazolidinediones
Placebo BID + Titrated Insulin Glargine | Exenatide 10 mcg • BID + Titrated Insulin Glargine | |
Intent-to-Treat Population (N) | 122 | 137 |
HbA 1c (%), Mean | ||
Baseline | 8.5 | 8.3 |
Change at Week 30 † | -1.0 | -1.7 |
Difference from placebo † (95% CI) | -0.7 [-1.0, -0.5] ¶ | |
Proportion Achieving HbA 1c < 7% | 29% | 56% |
Body Weight (kg), Mean | ||
Baseline | 93.8 | 95.4 |
Change at Week 30 ‡ | 1.0 | -1.8 |
Difference from placebo ‡ (95% CI) | -2.7 [-3.7, -1.7] ¶ | |
Fasting Serum Glucose § (mg/dL), Mean | ||
Baseline | 133 | 132 |
Change at Week 30 ‡ | -16 | -23 |
Difference from placebo ‡ (95% CI) | -7 [-18, 3] | |
• Exenatide 5 mcg twice daily for 1 month followed by 10 mcg BID for 5 months for the 30-week trial. † Least squares means are based on a mixed model adjusting for treatment, pooled investigator, visit, baseline HbA 1c value and treatment by visit, where subject is treated as a random effect. ‡ Least squares means are based on a mixed model adjusting for treatment, pooled investigator, visit, baseline HbA 1c stratum, baseline value of the dependent variable (where applicable) and treatment by visit, where subject is treated as a random effect. § Patients in both groups titrated insulin glargine dose to achieve optimal fasting glucose concentrations. ¶ p < 0.01, treatment vs. placebo. BID = twice daily. | ||
Table 10: Dosing Algorithm for Titration of Insulin Glargine •
Fasting Plasma Glucose Values (mg/dL) | Dose Change (U) |
< 56 † | -4 |
56 to 72 † | -2 |
73 to 99 ‡ | 0 |
100 to 119 ‡ | +2 |
120 to 139 ‡ | +4 |
140 to 179 ‡ | +6 |
≥ 180 ‡ | +8 |
Abbreviations: U = units. • Adapted from Riddle et al. 2003. † Value for at least 1 fasting plasma glucose measurement since the last assessment. ‡ Based on the average of fasting plasma glucose measurements taken over the prior 3 to 7 days. The increase in the total daily dose should not have exceeded more than 10 units per day or 10% of the current total daily dose, whichever was greater. | |
30-Week Comparator-Controlled Noninferiority Trial
A 30 week, open-label, active comparator-controlled, noninferiority study was conducted to evaluate the safety and efficacy of exenatide (n=315) versus titrated insulin lispro (n=312) on a background of optimized basal insulin glargine and metformin in patients with type 2 diabetes with inadequate glycemic control.
Following a 12-week basal insulin optimization (BIO) phase, subjects with an HbA 1c > 7.0% entered a 30-week intervention phase and were randomized to add either exenatide or insulin lispro to their existing regimen of insulin glargine and metformin. Insulin glargine was titrated to a target fasting plasma glucose of 72 to 100 mg/dL.
All patients assigned to exenatide initially received 5 mcg BID for four weeks. After four weeks, their dose was increased to 10 mcg BID. Patients in the exenatide-treated arm with an HbA 1c ≤ 8.0% at the end of the BIO phase decreased their insulin glargine dose by at least 10%.
All patients assigned to insulin lispro three times daily (TID) maintained their prior total daily insulin dose at baseline; however, the initial insulin lispro dose was ⅓ to 1/2; of the total daily insulin dose with the insulin glargine dose reduced accordingly. The insulin lispro dose was titrated based on preprandial glucose values.
The majority of patients (87%) were White, 7% were American Indian or Alaska Native, 5% were Asian, and < 1% were of Black or African American ethnicity.
The primary endpoint was the change in HbA 1c from baseline to Week 30. Both exenatide 10 mcg BID and titrated lispro provided a mean reduction in HbA 1c at Week 30 that met the pre-specified non-inferiority margin of 0.4%.
Table 11: 30-Week Comparator-Controlled Trial of Exenatide used in Combination with Insulin Glargine and Metformin
Titrated Insulin Lispro TID + Titrated Insulin Glargine | Exenatide 10 mcg • BID + Titrated Insulin Glargine | |
Intent-to-Treat Population (N) | 312 | 315 |
HbA 1c (%), Mean | ||
Baseline | 8.2 | 8.3 |
Change at Week 30 † # | −1.1 | −1.1 |
Difference from Insulin Lispro † # (95% CI) | −0.0 [−0.2, 0.1] | |
Body Weight (kg), Mean | ||
Baseline | 89.3 | 89.9 |
Change at Week 30 † # | 1.9 | −2.6 |
Difference from Insulin Lispro † # (95% CI) | −4.5 [−5.2, −3.9] | |
Fasting Serum Glucose ‡ (mg/dL), Mean | ||
Baseline | 126 | 130 |
Change at Week 30 † # | 5 | −7 |
Difference from Insulin Lispro † # (95% CI) | −12 [−19, −4] | |
• Exenatide 5 mcg BID for 1 month followed by 10 mcg BID for 5 months for the 30-week trial. † Least squares means are based on a mixed model adjusting for treatment, country, prior use of sulfonylurea (yes/no), visit, corresponding baseline, and treatment by visit interaction, where subject is treated as a random effect. # Data at 30 weeks are available from 88% and 84% of the intent-to-treat subjects in the Lispro and Exenatide groups, respectively. ‡ Patients titrated insulin glargine or insulin lispro dose to achieve prespecified target fasting and preprandial glucose concentrations. BID = twice daily. TID = three times daily. | ||
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Exenatide Injection, USP is supplied as a clear, colorless sterile solution for subcutaneous injection containing 250 mcg/mL exenatide, USP.
The following packages are available:
5 mcg per dose, in a single-patient-use prefilled pen containing 300 mcg/1.2 mL (250mcg/mL), 60 doses: NDC 70121-1685-1
10 mcg per dose, in a single-patient-use prefilled pen containing 600 mcg/2.4 mL (250mcg/mL), 60 doses: NDC 70121-1686-1
Storage and Handling
- Store exenatide injection in the refrigerator at 36 ○ to 46 ○ F (2 ○ to 8 ○ C).
- After first use, exenatide injection can be kept at a temperature not to exceed 77 ○ F (25 ○ C).
- Do not freeze. Do not use exenatide injection if it has been frozen.
- Protect exenatide injection from light.
- Discard the pen 30 days after first use, even if some drug remains in the pen.
- Use a puncture-resistant container to discard the needles. Do not reuse or share needles.
Mechanism of Action
Incretins, such as glucagon-like peptide-1 (GLP-1), enhance glucose-dependent insulin secretion and exhibit other antihyperglycemic actions following their release into the circulation from the gut. Exenatide is a GLP-1 receptor agonist that enhances glucose-dependent insulin secretion by the pancreatic beta-cell, suppresses inappropriately elevated glucagon secretion, and slows gastric emptying.
The amino acid sequence of exenatide partially overlaps that of human GLP-1. Exenatide has been shown to bind and activate the human GLP-1 receptor in vitro . This leads to an increase in both glucose-dependent synthesis of insulin, and in vivo secretion of insulin from pancreatic beta cells, by mechanisms involving cyclic AMP and/or other intracellular signaling pathways.
Exenatide improves glycemic control by reducing fasting and postprandial glucose concentrations in patients with type 2 diabetes through the actions described below.