Get your patient on Feirza 1/20 - Norethindrone Acetate And Ethinyl Estradiol Tablets Usp, 1 Mg/0.02 Mg And Ferrous Fumarate Tablets, 75 Mg (Norethindrone Acetate And Ethinyl Estradiol Tablets Usp, 1 Mg/0.02 Mg And Ferrous Fumarate Tablets, 75 Mg)
Feirza 1/20 - Norethindrone Acetate And Ethinyl Estradiol Tablets Usp, 1 Mg/0.02 Mg And Ferrous Fumarate Tablets, 75 Mg prescribing information
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs, including norethindrone acetate and ethinyl estradiol tablets, are contraindicated in women who are over 35 years of age and smoke (see CONTRAINDICATIONS and WARNINGS ).
INDICATIONS AND USAGE
Feirza 1/20 is indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Oral contraceptives are highly effective. Table I lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization, depends upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates.
| Adapted from RA Hatcher et al, Reference 7. | ||
% Of Women Experiencing an Unintended Pregnancy in the First Year of Continuous Use | ||
Method | Lowest Expected The authors’ best guess of the percentage of women expected to experience an accidental pregnancy among couples who initiate a method (not necessarily for the first time) and who use it consistently and correctly during the first year if they do not stop for any other reason. | Typical This term represents “typical” couples who initiate use of a method (not necessarily for the first time), who experience an accidental pregnancy during the first year if they do not stop use for any other reason. |
(No contraception) | (85) | (85) |
Oral contraceptives | 3 | |
combined | 0.1 | N/A N/A - Data not available. |
progestin only | 0.5 | N/A |
Diaphragm with spermicidal cream or jelly | 6 | 20 |
Spermicides alone (foam, creams, gels, vaginal suppositories, and vaginal film) | 6 | 26 |
Vaginal Sponge | ||
nulliparous | 9 | 20 |
parous | 20 | 40 |
Implant | 0.05 | 0.05 |
Injection: depot medroxyprogesterone acetate | 0.3 | 0.3 |
IUD | ||
progesterone T | 1.5 | 2 |
copper T 380A | 0.6 | 0.8 |
LNg 20 | 0.1 | 0.1 |
Condom without spermicides | ||
female | 5 | 21 |
male | 3 | 14 |
Cervical Cap with spermicidal cream or jelly | ||
nulliparous | 9 | 20 |
parous | 26 | 40 |
Periodic abstinence (all methods) | 1 to 9 | 25 |
Withdrawal | 4 | 19 |
Female sterilization | 0.5 | 0.5 |
Male sterilization | 0.10 | 0.15 |
DOSAGE AND ADMINISTRATION
The tablet blister pack has been designed to make oral contraceptive dosing as easy and as convenient as possible. The tablets are arranged in four rows of seven tablets each, with the days of the week appearing on the tablet blister above the first row of tablets.
Note: Each blister card has been preprinted with the days of the week, starting with Sunday, to facilitate a Sunday-Start regimen. Six different day label strips have been provided with the Detailed Patient & Brief Summary Patient Package Insert in order to accommodate a Day-1 Start regimen. If the patient is using the Day-1 Start regimen, she should place the self-adhesive day label strip that corresponds to her starting day over the preprinted days.
Important: The patient should be instructed to use an additional method of protection until after the first week of administration in the initial cycle when utilizing the Sunday-Start regimen.
The possibility of ovulation and conception prior to initiation of use should be considered.
Dosage and Administration for 28-Day Dosage Regimen
To achieve maximum contraceptive effectiveness, Feirza 1/20 should be taken exactly as directed and at intervals not exceeding 24 hours.
Feirza 1/20 provides a continuous administration regimen consisting of 21 white to off-white tablets of norethindrone acetate and ethinyl estradiol and 7 brown non-hormone containing tablets of ferrous fumarate. The ferrous fumarate tablets are present to facilitate ease of drug administration via a 28-day regimen and do not serve any therapeutic purpose. There is no need for the patient to count days between cycles because there are no “off-tablet days.”
A. Sunday-Start Regimen: The patient begins taking the first white to off-white tablet from the top row of the blister (labeled Sunday) on the first Sunday after menstrual flow begins. When the menstrual flow begins on Sunday, the first white to off-white tablet is taken on the same day. The patient takes one white to off-white tablet daily for 21 days. The last white to off-white tablet in the blister pack will be taken on a Saturday. Upon completion of all 21 white to off-white tablets, and without interruption, the patient takes one brown tablet daily for 7 days. Upon completion of this first course of tablets, the patient begins a second course of 28-day tablets, without interruption, the next day (Sunday), starting with the Sunday white to off-white tablet in the top row. Adhering to this regimen of one white to off-white tablet daily for 21 days, followed without interruption by one brown tablet daily for seven days, the patient will start all subsequent cycles on a Sunday.
B. Day-1 Start Regimen: The first day of menstrual flow is Day 1. The patient places the self-adhesive day label strip that corresponds to her starting day over the preprinted days on the blister card. She starts taking one white to off-white tablet daily, beginning with the first white to off-white tablet in the top row. After the last white to off-white tablet (at the end of the third row) has been taken, the patient will then take the brown tablets for a week (7 days). For all subsequent cycles, the patient begins a new 28 tablet regimen on the eighth day after taking her last white to off-white tablet, again starting with the first tablet in the top row after placing the appropriate day label strip over the preprinted days on the tablet blister. Following this regimen of 21 white to off-white tablets and 7 brown tablets, the patient will start all subsequent cycles on the same day of the week as the first course.
Tablets should be taken regularly with a meal or at bedtime. It should be stressed that efficacy of medication depends on strict adherence to the dosage schedule.
Special Notes on Administration
Menstruation usually begins two or three days, but may begin as late as the fourth or fifth day, after the brown tablets have been started. In any event, the next course of tablets should be started without interruption. If spotting occurs while the patient is taking white to off-white tablets, continue medication without interruption.
If the patient forgets to take one or more white to off-white tablets, the following is suggested:
One tablet is missed
take tablet as soon as remembered
take next tablet at the regular time
Two consecutive tablets are missed (week 1 or week 2)
take two tablets as soon as remembered
take two tablets the next day
use another birth control method for seven days following the missed tablets
Two consecutive tablets are missed (week 3)
Sunday-Start Regimen:
take one tablet daily until Sunday
discard remaining tablets
start new pack of tablets immediately (Sunday)
use another birth control method for seven days following the missed tablets
Day-1 Start Regimen:
discard remaining tablets
start new pack of tablets that same day
use another birth control method for seven days following the missed tablets
Three (or more) consecutive tablets are missed
Sunday-Start Regimen:
take one tablet daily until Sunday
discard remaining tablets
start new pack of tablets immediately (Sunday)
use another birth control method for seven days following the missed tablets
Day-1 Start Regimen:
discard remaining tablets
start new pack of tablets that same day
use another birth control method for seven days following the missed tablets
The possibility of ovulation occurring increases with each successive day that scheduled white to off-white tablets are missed. While there is little likelihood of ovulation occurring if only one white to off-white tablet is missed, the possibility of spotting or bleeding is increased. This is particularly likely to occur if two or more consecutive white to off-white tablets are missed.
If the patient forgets to take any of the seven brown tablets in week four, those brown tablets that were missed are discarded and one brown tablet is taken each day until the pack is empty. A back-up birth control method is not required during this time. A new pack of tablets should be started no later than the eighth day after the last white to off-white tablet was taken.
In the rare case of bleeding which resembles menstruation, the patient should be advised to discontinue medication and then begin taking tablets from a new tablet blister pack on the next Sunday or the first day (Day-1), depending on her regimen. Persistent bleeding which is not controlled by this method indicates the need for reexamination of the patient, at which time nonfunctional causes should be considered.
Use of Oral Contraceptives in the Event of a Missed Menstrual Period
If the patient has not adhered to the prescribed dosage regimen, the possibility of pregnancy should be considered after the first missed period and oral contraceptives should be withheld until pregnancy has been ruled out.
If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out before continuing the contraceptive regimen.
After several months on treatment, bleeding may be reduced to a point of virtual absence. This reduced flow may occur as a result of medication, in which event it is not indicative of pregnancy.
CONTRAINDICATIONS
Oral contraceptives are contraindicated in women who currently have the following conditions:
Thrombophlebitis or thromboembolic disorders
A past history of deep vein thrombophlebitis or thromboembolic disorders
Cerebral vascular or coronary artery disease
Current diagnosis of, or history of, breast cancer, which may be hormone sensitive
Carcinoma of the endometrium or other known or suspected estrogen-dependent neoplasia
Undiagnosed abnormal genital bleeding
Cholestatic jaundice of pregnancy or jaundice with prior pill use
Hepatic adenomas or carcinomas
Are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see Warnings , RISK OF LIVER ENZYME ELEVATIONS WITH CONCOMITANT HEPATITIS C TREATMENT ).
ADVERSE REACTIONS
An increased risk of the following serious adverse reactions has been associated with the use of oral contraceptives (see WARNINGS section):
Thrombophlebitis
Arterial thromboembolism
Pulmonary embolism
Myocardial infarction
Cerebral hemorrhage
Cerebral thrombosis
Hypertension
Gallbladder disease
Hepatic adenomas or benign liver tumors
Post Marketing Experience
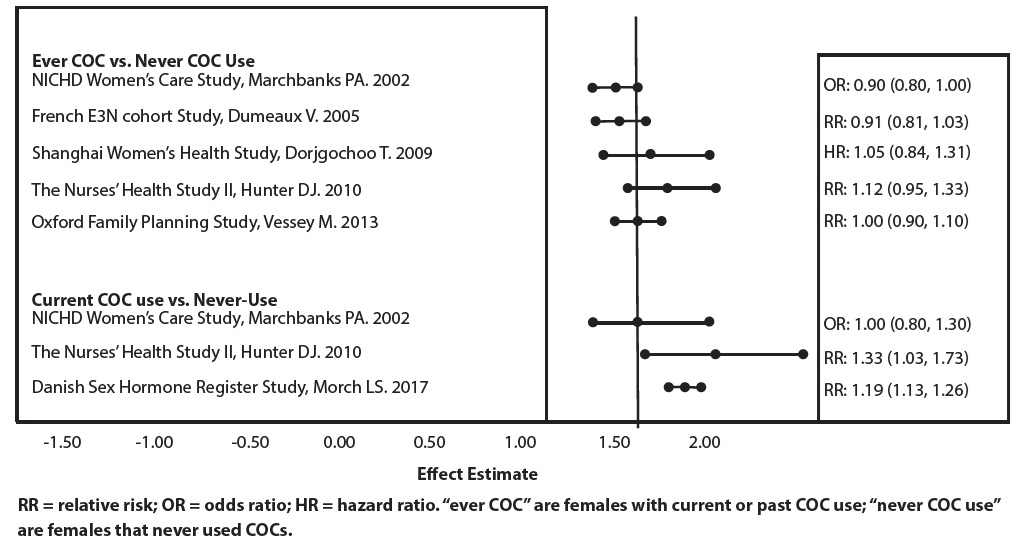
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 1) (70-74).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 1) (70, 73, 75). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
FIGURE 1: RELEVANT STUDIES OF RISK OF BREAST CANCER WITH COMBINED ORAL CONTRACEPTIVES
There is evidence of an association between the following conditions and the use of oral contraceptives, although additional confirmatory studies are needed:
Mesenteric thrombosis
Retinal thrombosis
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug-related:
Nausea
Vomiting
Gastrointestinal symptoms (such as abdominal cramps and bloating)
Breakthrough bleeding
Spotting
Change in menstrual flow
Amenorrhea
Temporary infertility after discontinuation of treatment
Edema
Melasma which may persist
Breast changes: tenderness, enlargement, secretion
Change in weight (increase or decrease)
Change in cervical erosion and secretion
Diminution in lactation when given immediately postpartum
Cholestatic jaundice
Migraine
Rash (allergic)
Depression
Reduced tolerance to carbohydrates
Vaginal candidiasis
Change in corneal curvature (steepening)
Intolerance to contact lenses
The following adverse reactions have been reported in users of oral contraceptives and the association has been neither confirmed nor refuted:
Pre-menstrual syndrome
Cataracts
Changes in appetite
Cystitis-like syndrome
Headache
Nervousness
Dizziness
Hirsutism
Loss of scalp hair
Erythema multiforme
Erythema nodosum
Hemorrhagic eruption
Vaginitis
Porphyria
Impaired renal function
Hemolytic uremic syndrome
Budd-Chiari syndrome
Acne
Changes in libido
Colitis
To report SUSPECTED ADVERSE REACTIONS, contact Xiromed, LLC at 844-XIROMED (844-947-6633) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Feirza 1/20 (Norethindrone Acetate and Ethinyl Estradiol Tablets, USP and Ferrous Fumarate Tablets) is a progestogen-estrogen combination.
Feirza 1/20 provides a continuous dosage regimen consisting of 21 oral contraceptive tablets and seven ferrous fumarate tablets. The ferrous fumarate tablets are present to facilitate ease of drug administration via a 28-day regimen, are non-hormonal, and do not serve any therapeutic purpose.
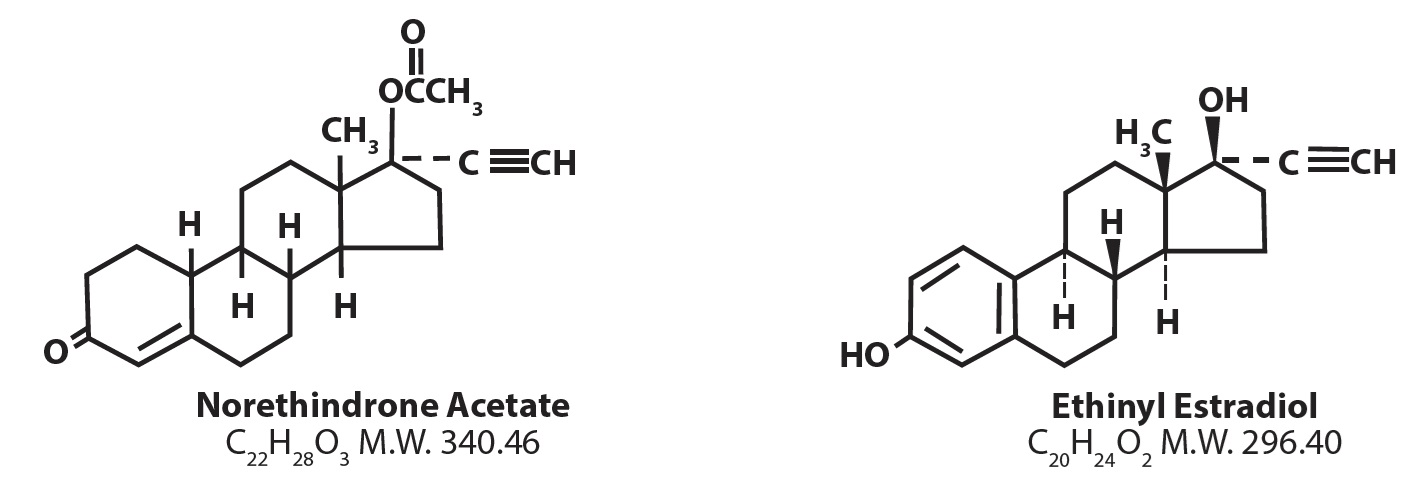
Each white to off-white tablet contains norethindrone acetate (19-Norpregn-4-en-20-yn-3-one, 17-(acetyloxy)-, (17α)), 1 mg; ethinyl estradiol (19-Norpregna-1,3,5(10)-trien-20-yne-3, 17-diol, (17α)-), 20 mcg. Also contains compressible sugar, croscarmellose sodium, dl -α-tocopherol, lactose anhydrous, lactose monohydrate, magnesium stearate, povidone K-25, sodium lauryl sulphate.
The structural formulas are as follows:
Each brown placebo tablet contains colloidal silicon dioxide, croscarmellose sodium, ferrous fumarate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone K-90. The ferrous fumarate tablets do not serve any therapeutic purpose.
CLINICAL PHARMACOLOGY
Combination oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations include changes in the cervical mucus (which increase the difficulty of sperm entry into the uterus) and the endometrium (which reduce the likelihood of implantation).
Pharmacokinetics
The pharmacokinetics of norethindrone acetate and ethinyl estradiol tablets have not been characterized; however, the following pharmacokinetic information regarding norethindrone acetate and ethinyl estradiol is taken from the literature.
Absorption
Norethindrone acetate appears to be completely and rapidly deacetylated to norethindrone after oral administration, since the disposition of norethindrone acetate is indistinguishable from that of orally administered norethindrone (1). Norethindrone acetate and ethinyl estradiol are subject to first-pass metabolism after oral dosing, resulting in an absolute bioavailability of approximately 64% for norethindrone and 43% for ethinyl estradiol (1-3).
Distribution
Volume of distribution of norethindrone and ethinyl estradiol ranges from 2 to 4 L/kg (1 to 3). Plasma protein binding of both steroids is extensive (greater than 95%); norethindrone binds to both albumin and sex hormone binding globulin, whereas ethinyl estradiol binds only to albumin (4).
Metabolism
Norethindrone undergoes extensive biotransformation, primarily via reduction, followed by sulfate and glucuronide conjugation. The majority of metabolites in the circulation are sulfates, with glucuronides accounting for most of the urinary metabolites (5). A small amount of norethindrone acetate is metabolically converted to ethinyl estradiol. Ethinyl estradiol is also extensively metabolized, both by oxidation and by conjugation with sulfate and glucuronide. Sulfates are the major circulating conjugates of ethinyl estradiol and glucuronides predominate in urine. The primary oxidative metabolite is 2-hydroxy ethinyl estradiol, formed by the CYP3A4 isoform of cytochrome P450. Part of the first-pass metabolism of ethinyl estradiol is believed to occur in gastrointestinal mucosa. Ethinyl estradiol may undergo enterohepatic circulation (6).
Excretion
Norethindrone and ethinyl estradiol are excreted in both urine and feces, primarily as metabolites (5, 6). Plasma clearance values for norethindrone and ethinyl estradiol are similar (approximately 0.4 L/hr/kg) (1-3).
Special Population
Race:
The effect of race on the disposition of norethindrone acetate and ethinyl estradiol tablets has not been evaluated.
Renal Insufficiency
The effect of renal disease on the disposition of norethindrone acetate and ethinyl estradiol tablets has not been evaluated. In premenopausal women with chronic renal failure undergoing peritoneal dialysis who received multiple doses of an oral contraceptive containing ethinyl estradiol and norethindrone, plasma ethinyl estradiol concentrations were higher and norethindrone concentrations were unchanged compared to concentrations in premenopausal women with normal renal function.
Hepatic Insufficiency
The effect of hepatic disease on the disposition of norethindrone acetate and ethinyl estradiol tablets has not been evaluated. However, ethinyl estradiol and norethindrone may be poorly metabolized in patients with impaired liver function.
Drug-Drug Interactions
Numerous drug-drug interactions have been reported for oral contraceptives. A summary of these is found under PRECAUTIONS , Drug Interactions .
HOW SUPPLIED
Feirza 1/20 (Norethindrone Acetate and Ethinyl Estradiol Tablets USP, 1 mg/20 mcg and Ferrous Fumarate Tablets, 75 mg) is packaged in cartons of 3 pouches (NDC 70700-307-85); each pouch (NDC 70700-307-84) contains a blister pack of 28 tablets. Each blister pack contains 21 white to off-white, round, flat-faced, unscored tablet debossed with 239 on one side and plain on the other side and 7 brown, round, flat-faced, unscored tablets debossed with 291 on one side and plain on the other side. Each brown tablet contains 75 mg ferrous fumarate.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
INSTRUCTIONS TO PATIENT
TABLET BLISTER PACK
The Feirza 1/20 blister pack has been designed to make oral contraceptive dosing as easy and as convenient as possible. The tablets are arranged in four rows of seven tablets each, with the days of the week appearing above the first row of tablets.
If your TABLET BLISTER PACK contains: | You are taking: |
21 white to off-white tablets and 7 brown tablets | Feirza 1/20 |
Each white to off-white tablet contains 1 mg norethindrone acetate and 20 mcg ethinyl estradiol.
Each brown tablet contains 75 mg ferrous fumarate, and is intended to help you remember to take the tablets correctly. These brown tablets are not intended to have any health benefit.
DIRECTIONS
To remove a tablet, press down on it with your thumb or finger. The tablet will drop through the back of the tablet blister. Do not press with your thumbnail, fingernail, or any other sharp object.
HOW TO TAKE THE PILL
IMPORTANT POINTS TO REMEMBER |
BEFORE YOU START TAKING YOUR PILLS:
1. BE SURE TO READ THESE DIRECTIONS:
Before you start taking your pills.
Anytime you are not sure what to do.
2. THE RIGHT WAY TO TAKE THE PILL IS TO TAKE ONE PILL EVERY DAY AT THE SAME TIME. If you miss pills you could get pregnant. This includes starting the pack late. The more pills you miss, the more likely you are to get pregnant.
3. MANY WOMEN HAVE SPOTTING OR LIGHT BLEEDING, OR MAY FEEL SICK TO THEIR STOMACH DURING THE FIRST 1 to 3 PACKS OF PILLS.
If you do have spotting or light bleeding or feel sick to your stomach, do not stop taking the pill. The problem will usually go away. If it doesn't go away, check with your doctor or clinic.
4. MISSING PILLS CAN ALSO CAUSE SPOTTING OR LIGHT BLEEDING, even when you make up these missed pills. On the days you take 2 pills to make up for missed pills, you could also feel a little sick to your stomach.
5. IF YOU HAVE VOMITING OR DIARRHEA, for any reason, or IF YOU TAKE SOME MEDICINES, including some antibiotics, your birth control pills may not work as well. Use a back-up birth control method (such as condoms or foam) until you check with your doctor or clinic.
6. IF YOU HAVE TROUBLE REMEMBERING TO TAKE THE PILL, talk to your doctor or clinic about how to make pill-taking easier or about using another method of birth control.
7. IF YOU HAVE ANY QUESTIONS OR ARE UNSURE ABOUT THE INFORMATION IN THIS LEAFLET, call your doctor or clinic.
BEFORE YOU START TAKING YOUR PILLS |
1. DECIDE WHAT TIME OF DAY YOU WANT TO TAKE YOUR PILL. It is important to take it at about the same time every day.
2. LOOK AT YOUR PILL PACK TO SEE IF IT HAS 28 PILLS:
The 28-Day pill pack has 21 “active” white to off-white pills (with hormones) to take for 3 weeks, followed by 1 week of reminder brown pills (without hormones).
3. ALSO FIND:
1) where on the pack to start taking pills,
2) in what order to take the pills (follow the arrows), and
3) the week numbers as shown in the following picture:
Feirza 1/20 will contain: 21 WHITE TO OFF-WHITE PILLS for WEEKS 1, 2, and 3 . WEEK 4 will contain BROWN PILLS ONLY .

4. BE SURE YOU HAVE READY AT ALL TIMES:
ANOTHER KIND OF BIRTH CONTROL (such as condoms or foam) to use as a back-up in case you miss pills.
An EXTRA, FULL PILL PACK.
WHEN TO START THE FIRST PACK OF PILLS |
You have a choice of which day to start taking your first pack of pills. Decide with your doctor or clinic which is the best day for you. Pick a time of day which will be easy to remember.
DAY-1 START:
1. Pick the day label strip that starts with the first day of your period. (This is the day you start bleeding or spotting, even if it is almost midnight when the bleeding begins.)
2. Place this day label strip on the tablet blister card over the area that has the days of the week (starting with Sunday) printed on the blister card.

3. Take the first “active” white to off-white pill of the first pack during the first 24 hours of your period .
4. You will not need to use a back-up method of birth control, since you are starting the pill at the beginning of your period.
SUNDAY START:
1. Take the first “active” white to off-white pill of the first pack on the Sunday after your period starts , even if you are still bleeding. If your period begins on Sunday, start the pack that same day.
2.000000000000000e+00 Use another method of birth control as a back-up method if you have sex anytime from the Sunday you start your first pack until the next Sunday (7 days). Condoms or foam are good back-up methods of birth control.
WHAT TO DO DURING THE MONTH |
1. TAKE ONE PILL AT THE SAME TIME EVERY DAY UNTIL THE PACK IS EMPTY.
Do not skip pills even if you are spotting or bleeding between monthly periods or feel sick to your stomach (nausea).
Do not skip pills even if you do not have sex very often.
2. WHEN YOU FINISH A PACK OR SWITCH YOUR BRAND OF PILLS:
Start the next pack on the day after your last “reminder” pill. Do not wait any days between packs.
WHAT TO DO IF YOU MISS PILLS |
If you MISS 1 white to off-white “active” pill:
1. Take it as soon as you remember. Take the next pill at your regular time. This means you may take 2 pills in 1 day.
2. You do not need to use a back-up birth control method if you have sex.
If you MISS 2 white to off-white “active” pills in a row in WEEK 1 OR WEEK 2 of your pack:
1. Take 2 pills on the day you remember and 2 pills the next day.
2. Then take 1 pill a day until you finish the pack.
3. You COULD GET PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms or foam) as a back-up method of birth control until you have taken a white to off-white “active” pill every day for 7 days.
If you MISS 2 white to off-white “active” pills in a row in THE 3 rd WEEK :
1.000000000000000e+00 If you are a Day-1 Starter:
THROW OUT the rest of the pill pack and start a new pack that same day.
If you are a Sunday Starter:
Keep taking 1 pill every day until Sunday. On Sunday, THROW OUT the rest of the pack and start a new pack of pills that same day.
2. You may not have your period this month, but this is expected. However, if you miss your period 2 months in a row, call your doctor or clinic because you might be pregnant.
3. You COULD GET PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms or foam) as a back-up method of birth control until you have taken a white to off-white “active” pill every day for 7 days.
If you MISS 3 OR MORE white to off-white “active” pills in a row (during the first 3 weeks):
1.000000000000000e+00 If you are a Day-1 Starter:
THROW OUT the rest of the pill pack and start a new pack that same day.
If you are a Sunday Starter:
Keep taking 1 pill every day until Sunday. On Sunday, THROW OUT the rest of the pack and start a new pack of pills that same day.
2. You may not have your period this month, but this is expected. However, if you miss your period 2 months in a row, call your doctor or clinic because you might be pregnant.
3. You COULD GET PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms or foam) as a back-up method of birth control until you have taken a white to off-white “active” pill every day for 7 days.
A REMINDER FOR THOSE ON 28-DAY PACKS: IF YOU FORGET ANY OF THE 7 BROWN “REMINDER” PILLS IN WEEK 4: THROW AWAY THE PILLS YOU MISSED. KEEP TAKING 1 PILL EACH DAY UNTIL THE PACK IS EMPTY. YOU DO NOT NEED A BACK-UP METHOD. |
FINALLY, IF YOU ARE STILL NOT SURE WHAT TO DO ABOUT THE PILLS YOU HAVE MISSED:
Use a BACK-UP METHOD anytime you have sex.
KEEP TAKING ONE WHITE TO OFF-WHITE “ACTIVE” PILL EACH DAY until you can reach your doctor or clinic.
PREGNANCY DUE TO PILL FAILURE
The incidence of pill failure resulting in pregnancy is approximately 1% (i.e., one pregnancy per 100 women per year) if taken every day as directed, but more typical failure rates are about 3%. If failure does occur, the risk to the fetus is minimal.
PREGNANCY AFTER STOPPING THE PILL
There may be some delay in becoming pregnant after you stop using oral contraceptives, especially if you had irregular menstrual cycles before you used oral contraceptives. It may be advisable to postpone conception until you begin menstruating regularly once you have stopped taking the pill and desire pregnancy.
There does not appear to be any increase in birth defects in newborn babies when pregnancy occurs soon after stopping the pill.
OVERDOSAGE
Serious ill effects have not been reported following ingestion of large doses of oral contraceptives by young children. Overdosage may cause nausea and withdrawal bleeding in females. In case of overdosage, contact your healthcare provider or pharmacist.
OTHER INFORMATION
Your healthcare provider will take a medical and family history and examine you before prescribing oral contraceptives. The physical examination may be delayed to another time if you request it and your healthcare provider believes that it is a good medical practice to postpone it. You should be reexamined at least once a year. Be sure to inform your healthcare provider if there is a family history of any of the conditions listed previously in this leaflet. Be sure to keep all appointments with your healthcare provider, because this is a time to determine if there are early signs of side effects of oral contraceptive use.
Do not use the drug for any condition other than the one for which it was prescribed. This drug has been prescribed specifically for you; do not give it to others who may want birth control pills.
HEALTH BENEFITS FROM ORAL CONTRACEPTIVES
In addition to preventing pregnancy, use of oral contraceptives may provide certain benefits. They are:
Menstrual cycles may become more regular
Blood flow during menstruation may be lighter and less iron may be lost. Therefore, anemia due to iron deficiency is less likely to occur
Pain or other symptoms during menstruation may be encountered less frequently
Ectopic (tubal) pregnancy may occur less frequently
Noncancerous cysts or lumps in the breast may occur less frequently
Acute pelvic inflammatory disease may occur less frequently
Oral contraceptive use may provide some protection against developing two forms of cancer: cancer of the ovaries and cancer of the lining of the uterus.
If you want more information about birth control pills, ask your doctor or pharmacist. They have a more technical leaflet called the "Physician Insert," which you may wish to read.
Remembering to take tablets according to schedule is stressed because of its importance in providing you the greatest degree of protection.
MISSED MENSTRUAL PERIODS FOR BOTH DOSAGE REGIMENS
At times there may be no menstrual period after a cycle of pills. Therefore, if you miss one menstrual period but have taken the pills exactly as you were supposed to , continue as usual into the next cycle. If you have not taken the pills correctly and miss a menstrual period, you may be pregnant and should stop taking oral contraceptives until your doctor or healthcare provider determines whether or not you are pregnant. Until you can get to your doctor or healthcare provider, use another form of contraception. If two consecutive menstrual periods are missed, you should stop taking pills until it is determined whether or not you are pregnant. Although there does not appear to be any increase in birth defects in newborn babies if you become pregnant while using oral contraceptives, you should discuss the situation with your doctor or healthcare provider.
Periodic Examination
Your doctor or healthcare provider will take a complete medical and family history before prescribing oral contraceptives. At that time and about once a year thereafter, he or she will generally examine your blood pressure, breasts, abdomen, and pelvic organs (including a Papanicolaou smear, i.e., test for cancer).
Keep this and all drugs out of the reach of children.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Rx only
Manufactured for:
Xiromed, LLC
Florham Park, NJ 07932
Made in India
Code No.: GUJ-DRUGS/G/28/1297
PI-307-00
Revised: 08/2024