Get your patient on Fluocinonide - Fluocinonide cream (Fluocinonide)
Fluocinonide - Fluocinonide cream prescribing information
INDICATIONS AND USAGE
Fluocinonide Cream USP, 0.05% (Emulsified Base) is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
DOSAGE AND ADMINISTRATION
Fluocinonide Cream, USP 0.05% (Emulsified Base) is generally applied to the affected area as a thin film from two to four times daily, as needed.
Occlusive dressings may be used for the management of psoriasis or recalcitrant conditions.
If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
CONTRAINDICATIONS
Fluocinonide Cream, USP 0.05% is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings. These reactions are listed in an approximate decreasing order of occurrence:
| Burning | Hypertrichosis | Maceration of the Skin |
| Itching | Acneiform Eruptions | Secondary Infection |
| Irritation | Hypopigmentation | Skin Atrophy |
| Dryness | Perioral Dermatitis | Striae |
| Folliculitis | Allergic Contact Dermatitis | Miliaria |
To Report SUSPECTED ADVERSE REACTIONS, contact Cosette Pharmaceuticals Inc. at 1-800-922-1038 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
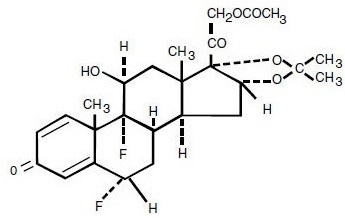
Fluocinonide Cream, USP 0.05% (Emulsified Base) is intended for topical administration. The active component is the corticosteroid fluocinonide, which is the 21-acetate ester of fluocinolone acetonide and has the chemical name pregna-1,4-diene-3,20-dione,21-(acetyloxy)-6,9-difluoro-11-hydroxy-16,17-[(1-methylethylidene)bis(oxy)]-, (6α ,11β,16α )-. It has the following chemical structure: Mol. Formula: C 26 H 32 F 2 O 7 Mol. Wt: 494.53
Fluocinonide Cream, USP 0.05% (Emulsified Base) contains fluocinonide 0.5 mg/g in a water-washable aqueous emollient base of cetyl alcohol, citric acid (anhydrous), mineral oil, polysorbate 60, propylene glycol, purified water, sorbitan monostearate, stearyl alcohol.
CLINICAL PHARMACOLOGY
Topical corticosteroids share anti-inflammatory, anti-pruritic and vasoconstrictive actions.
The mechanism of anti-inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
HOW SUPPLIED
Fluocinonide Cream, USP 0.05% (Emulsified Base) is supplied in 15 g (NDC 0713-0664-15), 30 g (NDC 0713-0664-31) and 60 g (NDC 0713-0664-60) tubes.
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature]. Do not refrigerate.
Distributed by:
Cosette Pharmaceuticals,Inc.
South Plainfield, NJ 07080
VC7673 Rev. 09/2022 8-0664CPLNC3