Get your patient on Folic Acid - Folic Acid injection, Solution (Folic Acid)
Folic Acid - Folic Acid injection, Solution prescribing information
INDICATIONS AND USAGE
Folic Acid Injection, USP alone is effective in the treatment of megaloblastic anemias due to a deficiency of folic acid as may be seen in tropical or nontropical sprue, in anemias of nutritional origin, pregnancy, infancy or childhood.
DOSAGE AND ADMINISTRATION
Parenteral Administration : Intramuscular (IM), intravenous (IV) and subcutaneous (SC) routes may be used if the disease is exceptionally severe or if gastrointestinal absorption may be, or is known to be, impaired.
Usual Therapeutic Dosage — In adults and children (regardless of age): up to 1 mg daily. Resistant cases may require larger doses.
Maintenance Level : When clinical symptoms have subsided and the blood picture has become normal, a maintenance level should be used, i.e., 0.1 mg for infants and up to 0.3 mg for children under four years of age, 0.4 mg for adults and children four or more years of age, and 0.8 mg for pregnant and lactating women, per day, but never less than 0.1 mg per day. Patient should be kept under close supervision and adjustment of the maintenance level made if relapse appears imminent.
In the presence of alcoholism, hemolytic anemia, anticonvulsant therapy or chronic infection, the maintenance level may need to be increased.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
ADVERSE REACTIONS
Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
To report SUSPECTED ADVERSE REACTIONS, contact XGen Pharmaceuticals DJB, Inc. at 1-866- 390-4411 or FDA at 1-800-FDA-1088 or www.fda. gov/medwatch.
DESCRIPTION
Folic Acid Injection, USP is a sterile, nonpyrogenic solution of sodium folate (prepared by the addition of sodium hydroxide to folic acid) in Water for Injection, USP intended for intramuscular (IM), intravenous (IV) or subcutaneous (SC) use.
Folic acid is a complex organic compound present in liver, yeast and other substances, which may be prepared synthetically. It is a yellow or yellowish orange, odorless crystalline powder. It is very slightly soluble in water, insoluble in alcohol, chloroform, ether; readily dissolves in dilute solutions of alkali hydroxides and carbonates. It is chemically designated as: L-Glutamic acid, N-[4-[[(2-amino-1-4-dihydro-4-oxo-6-pteridinyl) methyl] amino]benzoyl]-, and has the following structural formula.
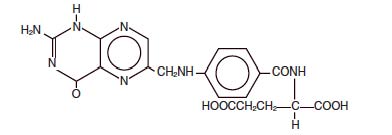
C 19 H 19 N 7 O 6 ; M.W. 441.40
Each mL contains: Sodium folate (equivalent to 5 mg folic acid); edetate disodium 2 mg; benzyl alcohol 15 mg (added as preservative); Water for Injection, USP q.s. Sodium hydroxide for pH adjustment (8.0 to 11.0).
CLINICAL PHARMACOLOGY
In man, an exogenous source of folate is required for nucleoprotein synthesis and maintenance of normal erythropoiesis. Folic acid, whether given by mouth or parenterally, stimulates specifically the production of red blood cells, white blood cells and platelets in persons suffering from certain megaloblastic anemias.
HOW SUPPLIED
Folic Acid Injection, USP 50 mg per 10 mL (5 mg per mL) is available as:
Product NDC: 39822-1100-1 10 mL Multiple Dose, in a flip-top vial, packaged individually.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
PROTECT FROM LIGHT.
Retain vial in carton until contents are used.
Manufactured in India.
Manufactured by: Anthea Pharma Private Limited Mfg. Lic: TS/SGY/2023-112201
Manufactured for: XGen Pharmaceuticals DJB, Inc. Big Flats, NY 14814

Revised: August 2025
FOLI-AN-PI-00
1500105