Get your patient on Gallifrey - Norethindrone Acetate tablet (Norethindrone Acetate)
Gallifrey - Norethindrone Acetate tablet prescribing information
INDICATIONS AND USAGE
Gallifrey (Norethindrone Acetate Tablets, USP), 5 mg
Gallifrey is indicated for the treatment of secondary amenorrhea, endometriosis, and abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology, such as submucous fibroids or uterine cancer. Gallifrey is not intended, recommended or approved to be used with concomitant estrogen therapy in postmenopausal women for endometrial protection.
DOSAGE AND ADMINISTRATION
Gallifrey (Norethindrone Acetate Tablets, USP), 5 mg
Therapy with Gallifrey tablets must be adapted to the specific indications and therapeutic response of the individual patient.
Secondary amenorrhea, abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology: 2.5 mg to 10 mg Gallifrey may be given daily for 5 to 10 days to produce secretory transformation of an endometrium that has been adequately primed with either endogenous or exogenous estrogen.
Progestin withdrawal bleeding usually occurs within three to seven days after discontinuing Gallifrey therapy. Patients with a past history of recurrent episodes of abnormal uterine bleeding may benefit from planned menstrual cycling with Gallifrey.
Endometriosis: Initial daily dosage of 5 mg Gallifrey for two weeks. Dosage should be increased by 2.5 mg per day every two weeks until 15 mg per day of Gallifrey is reached. Therapy may be held at this level for six to nine months or until annoying breakthrough bleeding demands temporary termination.
CONTRAINDICATIONS
Known or suspected pregnancy. There is no indication for norethindrone acetate tablets in pregnancy. (See PRECAUTIONS .)
Undiagnosed vaginal bleeding
Known, suspected or history of cancer of the breast
Active deep vein thrombosis, pulmonary embolism or history of these conditions
Active or recent (e.g., within the past year) arterial thromboembolic disease (e.g., stroke, myocardial infarction)
Impaired liver function or liver disease
As a diagnostic test for pregnancy
Hypersensitivity to any of the drug components
ADVERSE REACTIONS
See WARNINGS and PRECAUTIONS .
The following adverse reactions have been observed in women taking progestins:
Breakthrough bleeding
Spotting
Change in menstrual flow
Amenorrhea
Edema
Changes in weight (decreases, increases)
Changes in the cervical squamo-columnar junction and cervical secretions
Cholestatic jaundice
Rash (allergic) with and without pruritus
Melasma or chloasma
Clinical depression
Acne
Breast enlargement/tenderness
Headache/migraine
Urticaria
Abnormalities of liver tests (i.e., AST, ALT, Bilirubin)
Decreased HDL cholesterol and increased LDL/HDL ratio
Mood swings
Nausea
Insomnia
Anaphylactic/anaphylactoid reactions
Thrombotic and thromboembolic events (e.g., deep vein thrombosis, pulmonary embolism, retinal vascular thrombosis, cerebral thrombosis and embolism)
Optic neuritis (which may lead to partial or complete loss of vision)
Drug Interactions
No pharmacokinetic drug interaction studies investigating any drug-drug interactions with norethindrone acetate tablets have been conducted.
DESCRIPTION
Gallifrey (Norethindrone acetate tablets, USP), 5 mg oral tablets.
Gallifrey, (17-hydroxy-19-nor-17α-pregn-4-en-20-yn-3-one acetate), a synthetic, orally active progestin, is the acetic acid ester of norethindrone. It is a white, or creamy white, crystalline powder.
Gallifrey tablets contain the following inactive ingredients: lactose anhydrous, lactose monohydrate, magnesium stearate, and microcrystalline cellulose.
The USP Dissolution Test is pending.
CLINICAL PHARMACOLOGY
Norethindrone acetate induces secretory changes in an estrogen-primed endometrium. On a weight basis, it is twice as potent as norethindrone.
PHARMACOKINETICS
Absorption
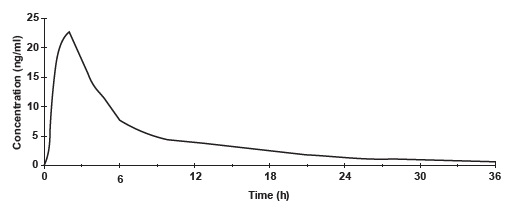
Norethindrone acetate is completely and rapidly deacetylated to norethindrone (NET) after oral administration, and the disposition of norethindrone acetate is indistinguishable from that of orally administered norethindrone. Norethindrone acetate is rapidly absorbed from norethindrone acetate tablets, with maximum plasma concentration of norethindrone generally occurring at about 2 hours post-dose. The pharmacokinetic parameters of norethindrone following single oral administration of norethindrone acetate tablets in 29 healthy female volunteers are summarized in Table 1.
| AUC = area under the curve, C max = maximum plasma concentration, t max = time at maximum plasma concentration, t 1/2 = half-life, SD = standard deviation |
Norethindrone acetate tablets (n=29) Arithmetic Mean ± SD |
Norethindrone (NET) |
AUC (0-inf)(ng/ml•h) 166.9 ± 56.28 |
C max (ng/ml) 26.19 ± 6.19 |
t max (h) 1.83 ± 0.58 |
t 1/2 (h) 8.51 ± 2.19 |
Figure 1. Mean Plasma Concentration Profile after a Single Dose of 5 mg Administered to 29 Healthy Female Volunteers under Fasting Conditions
Effect of Food
The effect of food administration on the pharmacokinetics of norethindrone acetate tablets has not been studied.
Distribution
Norethindrone is 36% bound to sex hormone-binding globulin (SHBG) and 61% bound to albumin. Volume of distribution of norethindrone is about 4 L/kg.
Metabolism
Norethindrone undergoes extensive biotransformation, primarily via reduction, followed by sulfate and glucuronide conjugation. The majority of metabolites in the circulation are sulfates, with glucuronides accounting for most of the urinary metabolites.
Excretion
Plasma clearance value for norethindrone is approximately 0.4 L/hr/kg. Norethindrone is excreted in both urine and feces, primarily as metabolites. The mean terminal elimination half-life of norethindrone following a single dose administration of norethindrone acetate tablets is approximately 9 hours.
SPECIAL POPULATIONS
Geriatrics
The effect of age on the pharmacokinetics of norethindrone after norethindrone acetate tablets administration has not been evaluated.
Race
The effect of race on the disposition of norethindrone after norethindrone acetate tablets administration has not been evaluated.
Renal Insufficiency
The effect of renal disease on the disposition of norethindrone after norethindrone acetate tablets administration has not been evaluated. In premenopausal women with chronic renal failure undergoing peritoneal dialysis who received multiple doses of an oral contraceptive containing ethinyl estradiol and norethindrone, plasma norethindrone concentration was unchanged compared to concentrations in premenopausal women with normal renal function.
Hepatic Insufficiency
The effect of hepatic disease on the disposition of norethindrone after norethindrone acetate tablets administration has not been evaluated. However, norethindrone acetate tablets are contraindicated in markedly impaired liver function or liver disease.
Drug Interactions
No pharmacokinetic drug interaction studies investigating any drug-drug interactions with norethindrone acetate tablets have been conducted.
HOW SUPPLIED
Gallifrey (Norethindrone acetate tablets, USP) is available as:
5 mg: White to off white, oval, flat-faced, beveled edge tablet debossed with 257 on one side of the tablet and a score (functional scoring) on the other side.
Available as follows:
Bottle of 50 tablets NDC 70700-326-50
Store at 20º to 25ºC (68º to 77ºF). [See USP Controlled Room Temperature.]
PHARMACIST: Dispense a Patient Information Leaflet with each prescription.