Get your patient on Glucagon - Glucagon (Glucagon)
Glucagon - Glucagon prescribing information
INDICATIONS AND USAGE
For the treatment of severe hypoglycemia: Glucagon is indicated as a treatment for severe hypoglycemia (low blood sugar) which may occur in patients with diabetes mellitus. Because patients with type 1 diabetes may have less of an increase in blood glucose levels compared with a stable type 2 patient, supplementary carbohydrate should be given as soon as possible, especially to a pediatric patient.
For use as a diagnostic aid: Glucagon is indicated as a diagnostic aid in the radiologic examination of the stomach, duodenum, small bowel, and colon when diminished intestinal motility would be advantageous.
Glucagon is as effective for this examination as are the anticholinergic drugs. However, as use of glucagon in combination with anticholinergic drugs may result in increased side effects, the use of glucagon in combination with anticholinergic drugs is not recommended.
DOSAGE AND ADMINISTRATION
General Instructions for Use:
- The diluent is provided for use only in the preparation of glucagon for parenteral injection and for no other use.
- Glucagon should not be used at concentrations greater than 1 mg/mL.
- Reconstituted glucagon should be used immediately. Discard any unused portion.
- Reconstituted glucagon solutions should be used only if they are clear and of a water-like consistency.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
Directions for Treatment of Severe Hypoglycemia: Severe hypoglycemia should be treated initially with intravenous glucose, if possible.
- If parenteral glucose can not be used, dissolve the lyophilized glucagon using the accompanying diluting solution and use immediately.
- For adults and for pediatric patients weighing more than 44 lb (20 kg), give 1 mg by subcutaneous, intramuscular, or intravenous injection.
- For pediatric patients weighing less than 44 lb (20 kg), give 0.5 mg or a dose equivalent to 20 to 30 mcg/kg.2-6
- Discard any unused portion.
- An unconscious patient will usually awaken within 15 minutes following the glucagon injection. If the response is delayed, there is no contraindication to the administration of an additional dose of glucagon; however, in view of the deleterious effects of cerebral hypoglycemia emergency aid should be sought so that parenteral glucose can be given.
- After the patient responds, supplemental carbohydrate should be given to restore liver glycogen and to prevent secondary hypoglycemia.
Directions for Use as a Diagnostic Aid: Dissolve the lyophilized glucagon using the accompanying diluting solution and use immediately. Discard any unused portion. The doses in the following table may be administered for relaxation of the stomach, duodenum, and small bowel, depending on the onset and duration of effect required for the examination. Since the stomach is less sensitive to the effect of glucagon, 0.5 mg intravenous or 2 mg intramuscular are recommended.
• Administration of 2 mg (2 units) doses produces a higher incidence of nausea and vomiting than do lower doses. | |||
| Dose | Route of Administration | Time of Onset of Action | Approximate Duration of Effect |
| 0.25-0.5 mg (0.25-0.5 units) | IV | 1 minute | 9-17 minutes |
| 1-mg (1 unit) | IM | 8-10 minutes | 12-27 minutes |
| 2 mg• (2 units) | IV | 1 minute | 22-25 minutes |
| 2 mg• (2 units) | IM | 4-7 minutes | 21-32 minutes |
For examination of the colon, it is recommended that a 2 mg (2 units) dose be administered intramuscularly approximately 10 minutes prior to the procedure. Colon relaxation and reduction of patient discomfort may allow the radiologist to perform a more satisfactory examination.
CONTRAINDICATIONS
Glucagon is contraindicated in patients with known hypersensitivity to it or in patients with known pheochromocytoma.
ADVERSE REACTIONS
Side effects may include nausea and vomiting. These reactions may also occur with hypoglycemia. Generalized allergic reactions have been reported (see WARNINGS ). In a three month controlled study of 75 volunteers comparing animal-sourced glucagon with glucagon manufactured through rDNA technology, no glucagon-specific antibodies were detected in either treatment group.
Necrolytic migratory erythema (NME) cases have been reported postmarketing in patients receiving continuous infusions of glucagon (see WARNINGS ).
DESCRIPTION
Glucagon for Injection (synthetic origin) is a polypeptide hormone identical to human glucagon that increases blood glucose and relaxes smooth muscle of the gastrointestinal tract. Glucagon is produced by solid state peptide synthesis and is highly purified.
Glucagon is a single-chain polypeptide that contains 29 amino acid residues and has a molecular weight of 3483.
The empirical formula is C 153 H 225 SN 43 O 49 S. The primary sequence of glucagon is shown below.
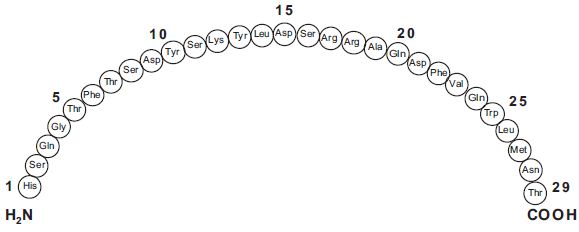
Crystalline glucagon (synthetic origin) is a white to off-white powder. It is relatively insoluble in water but is soluble at a pH of less than 3 or more than 9.5.
Glucagon is available for use intravenously, intramuscularly, or subcutaneously in a kit that contains a vial of sterile glucagon and a syringe of sterile diluent. The vial contains 1 mg of glucagon and 49 mg of lactose. Hydrochloric acid may have been added during manufacture to adjust the pH of the glucagon. One International Unit of glucagon is equivalent to 1 mg of glucagon.1 The diluent syringe contains 12 mg/mL of glycerin, Water For Injection, and hydrochloric acid.
CLINICAL PHARMACOLOGY
Glucagon increases blood glucose concentration and is used in the treatment of severe hypoglycemia. Glucagon acts only on liver glycogen, converting it to glucose.
Glucagon administered through a parenteral route relaxes smooth muscle of the stomach, duodenum, small bowel, and colon.
Pharmacokinetics
Glucagon has been studied following intramuscular, subcutaneous, and intravenous administration in adult volunteers. Administration of the intravenous glucagon showed dose proportionality of the pharmacokinetics between 0.25 and 2.0 mg. Calculations from a 1 mg dose showed a small volume of distribution (mean, 0.25 L/kg) and a moderate clearance (mean, 13.5 mL/min/kg). The half-life was short, ranging from 8 to 18 minutes.
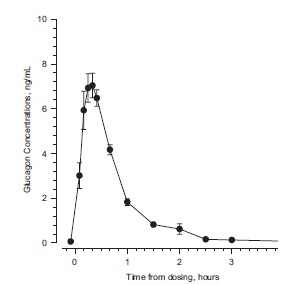
Maximum plasma concentrations of 7.9 ng/mL were achieved approximately 20 minutes after subcutaneous administration (see Figure 1A). With intramuscular dosing, maximum plasma concentrations of 6.9 ng/mL were attained approximately 13 minutes after dosing. Glucagon is extensively degraded in liver, kidney, and plasma. Urinary excretion of intact glucagon has not been measured.
Pharmacokinetics
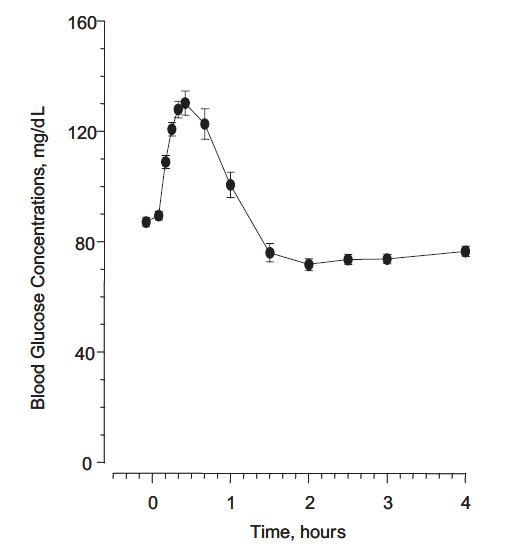
In a study of 25 volunteers, a subcutaneous dose of 1 mg glucagon resulted in a mean peak glucose concentration of 136 mg/dL 30 minutes after injection (see Figure 1B). Similarly, following intramuscular injection, the mean peak glucose level was 138 mg/dL, which occurred at 26 minutes after injection. No difference in maximum blood glucose concentration between animal-sourced and rDNA glucagon was observed after subcutaneous and intramuscular injection.
Figure 1 Mean (± SE) serum glucagon and blood glucose levels after subcutaneous injection of glucagon (1mg) in 25 normal volunteers
A
B
HOW SUPPLIED
Glucagon Emergency Kit for Low Blood Sugar (Glucagon for Injection) contains:
1 mg glucagon for injection (as a sterile, lyophilized white to off-white powder) in a single-dose vial and 1 mL of diluent for glucagon in a prefilled syringe. NDC 0548-5850-00
Stability and Storage: Before Reconstitution - Vials of glucagon, as well as the diluent for glucagon, may be stored at controlled room temperature 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature.]
The USP defines controlled room temperature by the following: A temperature maintained thermostatically that encompasses the usual and customary working environment of 20° to 25°C (68° to 77°F); that results in a mean kinetic temperature calculated to be not more than 25°C; and that allows for excursions between 15° and 30°C (59° and 86°F) that are experienced in pharmacies, hospitals, and warehouses.
After Reconstitution - Glucagon for Injection should be used immediately. Discard any unused portion.