Get your patient on Halobetasol Propionate - Halobetasol Propionate cream (Halobetasol Propionate)
Halobetasol Propionate - Halobetasol Propionate cream prescribing information
INDICATIONS AND USAGE
Halobetasol Propionate Cream, 0.05% is a super-high potency corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Treatment beyond two consecutive weeks is not recommended, and the total dosage should not exceed 50 g/week because of the potential for the drug to suppress the hypothalamic-pituitary-adrenal (HPA) axis. Use in children under 12 years of age is not recommended.
As with other highly active corticosteroids, therapy should be discontinued when control has been achieved. If no improvement is seen within 2 weeks, reassessment of the diagnosis may be necessary.
DOSAGE AND ADMINISTRATION
Apply a thin layer of Halobetasol Propionate Cream to the affected skin once or twice daily, as directed by your physician, and rub in gently and completely.
Halobetasol Propionate Cream is a super-high potency topical corticosteroid; therefore, treatment should be limited to two weeks, and amounts greater than 50 g/wk should not be used. As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, reassessment of diagnosis may be necessary.
Halobetasol Propionate Cream should not be used with occlusive dressings.
CONTRAINDICATIONS
Halobetasol Propionate Cream is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
ADVERSE REACTIONS
In controlled clinical trials, the most frequent adverse events reported for Halobetasol Propionate Cream included stinging, burning or itching in 4.4% of the patients. Less frequently reported adverse reactions were dry skin, erythema, skin atrophy, leukoderma, vesicles and rash.
The following additional local adverse reactions are reported infrequently with topical corticosteroids, and they may occur more frequently with high potency corticosteroids, such as Halobetasol Propionate Cream. These reactions are listed in an approximate decreasing order of occurrence: folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, striae and miliaria.
To report SUSPECTED ADVERSE REACTIONS, contact Cosette Pharmaceuticals, Inc. at 1-800-922-1038 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Halobetasol Propionate Cream, 0.05% contains halobetasol propionate, a synthetic corticosteroid for topical dermatological use. The corticosteroids constitute a class of primarily synthetic steroids used topically as an anti-inflammatory and antipruritic agent.
Chemically halobetasol propionate is 21-chloro-6α, 9-difluoro-11β, 17-dihydroxy-16β -methylpregna-1, 4-diene-3-20-dione, 17-propionate, C 25 H 31 ClF 2 O 5 . It has the following structural formula:
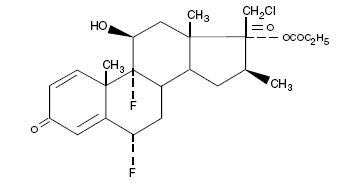
Halobetasol propionate has the molecular weight of 485. It is a white crystalline powder insoluble in water.
Each gram of Halobetasol Propionate Cream contains 0.5 mg/g of halobetasol propionate in a cream base of cetyl alcohol, glycerin, isopropyl isostearate, isopropyl palmitate, steareth-21, diazolidinyl urea, methylchloroisothiazolinone, (and) methylisothiazolinone and water.
CLINICAL PHARMACOLOGY
Like other topical corticosteroids, halobetasol propionate has anti-inflammatory, antipruritic and vasoconstrictive actions. The mechanism of the anti-inflammatory activity of the topical corticosteroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A 2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A 2 .
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle and the integrity of the epidermal barrier. Occlusive dressings with hydrocortisone for up to 24 hours have not been demonstrated to increase penetration; however, occlusion of hydrocortisone for 96 hours markedly enhances penetration. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption.
Human and animal studies indicate that less than 6% of the applied dose of halobetasol propionate enters the circulation within 96 hours following topical administration of Halobetasol Propionate Cream.
Studies performed with Halobetasol Propionate Cream indicate that it is in the super-high range of potency as compared with other topical corticosteroids.
HOW SUPPLIED
Halobetasol Propionate Cream, 0.05% is supplied in thefollowing tube sizes: 15 g (NDC 0713-0640-15) 50 g (NDC 0713-0640-86)
STORAGE
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
Distributed by: Cosette Pharmaceuticals, Inc. South Plainfield, NJ 07080
8-0640CPLNC2 Rev. 08/2022 VC7645