Get your patient on Hydralazine Hydrochloride - Hydralazine Hydrochloride tablet (Hydralazine Hydrochloride)
Hydralazine Hydrochloride - Hydralazine Hydrochloride tablet prescribing information
INDICATIONS & USAGE
Essential hypertension, alone or as an adjunct.
DOSAGE & ADMINISTRATION
Initiate therapy in gradually increasing dosages; adjust according to individual response. Start with 10 mg four times daily for the first 2 to 4 days, increase to 25 mg four times daily for the balance of the first week. For the second and subsequent weeks, increase dosage to 50 mg four times daily. For maintenance, adjust dosage to the lowest effective levels.
The incidence of toxic reactions, particularly the L.E. cell syndrome, is high in the group of patients receiving large doses of hydrALAZINE hydrochloride tablets.
In a few resistant patients, up to 300 mg of hydrALAZINE hydrochloride tablets daily may be required for a significant antihypertensive effect. In such cases, a lower dosage of hydrALAZINE hydrochloride tablets combined with a thiazide and/or reserpine or a beta blocker may be considered. However, when combining therapy, individual titration is essential to ensure the lowest possible therapeutic dose of each drug.
CONTRAINDICATIONS
Hypersensitivity to hydrALAZINE; coronary artery disease; mitral valvular rheumatic heart disease.
ADVERSE REACTIONS
Adverse reactions with hydrALAZINE are usually reversible when dosage is reduced. However, in some cases it may be necessary to discontinue the drug. The following adverse reactions have been observed, but there has not been enough systematic collection of data to support an estimate of their frequency.
Common headache, anorexia, nausea, vomiting, diarrhea, palpitations, tachycardia, angina pectoris.
Less Frequent Digestive: constipation, paralytic ileus.
Cardiovascular hypotension, paradoxical pressor response, edema.
Respiratory dyspnea.
Neurologic peripheral neuritis, evidenced by paresthesia, numbness, and tingling; dizziness; tremors; muscle cramps; psychotic reactions characterized by depression, disorientation, or anxiety.
Genitourinary difficulty in urination.
Hematologic blood dyscrasias, consisting of reduction in hemoglobin and red cell count, leukopenia, agranulocytosis, purpura; lymphadenopathy; splenomegaly.
Hypersensitive Reactions rash, urticaria, pruritus, fever, chills, arthralgia, eosinophilia, and rarely, hepatitis.
Other nasal congestion, flushing, lacrimation, conjunctivitis.
Drug/Drug Interactions
MAO inhibitors should be used with caution in patients receiving hydrALAZINE.
When other potent parenteral antihypertensive drugs, such as diazoxide, are used in combination with hydrALAZINE, patients should be continuously observed for several hours for any excessive fall in blood pressure. Profound hypotensive episodes may occur when diazoxide injection and hydrALAZINE are used concomitantly.
DESCRIPTION
HydrALAZINE hydrochloride USP, is an antihypertensive, for oral administration. Its chemical name is 1-hydrazinophthalazine monohydrochloride, and its structural formula is:
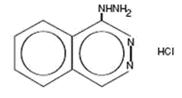
C 8 H 8 N 4.000000000000000e+00 HCl M.W. 196.64
HydrALAZINE hydrochloride USP is a white to off-white, odorless crystalline powder. It is soluble in water, slightly soluble in alcohol, and very slightly soluble in ether. It melts at about 275°C, with decomposition. Each tablet for oral administration contains 25 mg, 50 mg or 100 mg HydrALAZINE hydrochloride USP. Tablets also contain mannitol, microcrystalline cellulose, sodium starch glycolate, stearic acid and FD&C Yellow #6.
CLINICAL PHARMACOLOGY
Although the precise mechanism of action of hydrALAZINE is not fully understood, the major effects are on the cardiovascular system.
HydrALAZINE apparently lowers blood pressure by exerting a peripheral vasodilating effect through a direct relaxation of vascular smooth muscle. HydrALAZINE, by altering cellular calcium metabolism, interferes with the calcium movements within the vascular smooth muscle that are responsible for initiating or maintaining the contractile state.
The peripheral vasodilating effect of hydrALAZINE results in decreased arterial blood pressure (diastolic more than systolic); decreased peripheral vascular resistance; and an increased heart rate, stroke volume, and cardiac output. The preferential dilatation of arterioles, as compared to veins, minimizes postural hypotension and promotes the increase in cardiac output. HydrALAZINE usually increases renin activity in plasma, presumably as a result of increased secretion of renin by the renal juxtaglomerular cells in response to reflex sympathetic discharge. This increase in renin activity leads to the production of angiotensin II, which then causes stimulation of aldosterone and consequent sodium reabsorption. HydrALAZINE also maintains or increases renal and cerebral blood flow.
HydrALAZINE is rapidly absorbed after oral administration, and peak plasma levels are reached at 1 to 2 hours. Plasma levels of apparent hydrALAZINE decline with a half-life of 3 to 7 hours. Binding to human plasma protein is 87%. Plasma levels of hydrALAZINE vary widely among individuals. HydrALAZINE is subject to polymorphic acetylation; slow acetylators generally have higher plasma levels of hydrALAZINE and require lower doses to maintain control of blood pressure. HydrALAZINE undergoes extensive hepatic metabolism; it is excreted mainly in the form of metabolites in the urine.
HOW SUPPLIED
HydrALAZINE Hydrochloride Tablets USP are available as:
| 25 mg | orange, round, uncoated, biconvex tablet debossed 'C43' on one side and plain on other side |
| Bottles of 100………………………………………………...NDC 72241-004-05 Bottles of 1,000………………………………………………NDC 72241-004-11 | |
| 50 mg | orange, round, uncoated, biconvex tablet debossed 'C45' on one side and plain on other side |
| Bottles of 100………………………………………………...NDC 72241-005-05 Bottles of 1,000………………………………………………NDC 72241-005-11 | |
| 100 mg | orange, round, uncoated, biconvex tablet debossed 'C47' on one side and plain on other side |
| Bottles of 100………………………………………………...NDC 72241-006-05 Bottles of 1,000……………………………………………….NDC 72241-006-11 |
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required)
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN
Manufactured for :
Modavar Pharmaceuticals LLC. 1016, 16th Street, Suite 602, Washington, DC 20036 Toll free number : 800 688 4697
Manufactured By:
Cadila Pharmaceuticals Ltd. 1389, Dholka, Dist. Ahmedabad, Gujarat State, India.
Revised: June 2021