Get your patient on Hydralazine Hydrochloride - Hydralazine Hydrochloride injection (Hydralazine Hydrochloride)
Hydralazine Hydrochloride - Hydralazine Hydrochloride injection prescribing information
INDICATIONS AND USAGE
Severe essential hypertension when the drug cannot be given orally or when there is an urgent need to lower blood pressure.
DOSAGE AND ADMINISTRATION
When there is urgent need, therapy in the hospitalized patient may be initiated intramuscularly or as a rapid intravenous bolus injection directly into the vein. Hydralazine hydrochloride injection should be used only when the drug cannot be given orally. The usual dose is 20 to 40 mg, repeated as necessary.
Certain patients (especially those with marked renal damage) may require a lower dose. Blood pressure should be checked frequently. It may begin to fall within a few minutes after injection, with the average maximal decrease occurring in 10 to 80 minutes. In cases where there has been increased intracranial pressure, lowering the blood pressure may increase cerebral ischemia. Most patients can be transferred to oral hydralazine hydrochloride within 24 to 48 hours.
The product should be used immediately after the vial is opened. It should not be added to infusion solutions. Hydralazine hydrochloride injection may discolor upon contact with metal; discolored solutions should be discarded.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Hypersensitivity to hydralazine; coronary artery disease; mitral valvular rheumatic heart disease.
ADVERSE REACTIONS
Adverse reactions with hydralazine hydrochloride are usually reversible when dosage is reduced. However, in some cases it may be necessary to discontinue the drug.
The following adverse reactions have been observed, but there has not been enough systematic collection of data to support an estimate of their frequency.
Common: Headache, anorexia, nausea, vomiting, diarrhea, palpitations, tachycardia, angina pectoris.
Less Frequent: Digestive: constipation, paralytic ileus.
Cardiovascular: hypotension, paradoxical pressor response, edema.
Respiratory: dyspnea.
Neurologic: peripheral neuritis, evidenced by paresthesia, numbness and tingling; dizziness; tremors; muscle cramps; psychotic reactions characterized by depression, disorientation, or anxiety.
Genitourinary: difficulty in urination.
Hematologic: blood dyscrasias, consisting of reduction in hemoglobin and red cell count, leukopenia, agranulocytosis, purpura; lymphadenopathy; splenomegaly.
Hypersensitive Reactions : rash, urticaria, pruritus, fever, chills, arthralgia, eosinophilia and rarely hepatitis.
Other: nasal congestion, flushing, lacrimation, conjunctivitis.
DESCRIPTION
Hydralazine hydrochloride injection, USP is an antihypertensive available in a 1 mL vial for intravenous and intramuscular administration. Hydralazine hydrochloride injection, USP is a sterile, nonpyrogenic clear, colorless solution.
Each mL contains:
Active: Hydralazine hydrochloride USP, 20 mg
Preservatives: Methylparaben NF, 0.65 mg; Propylparaben NF, 0.35 mg
Inactives: Propylene glycol USP, 103.6 mg; Sodium hydroxide and/or Hydrochloric acid USP to adjust pH (3.4 to 4.4) and Water for injection.
Hydralazine hydrochloride, USP is 1-hydrazinophthalazine monohydrochloride and its structural formula is:
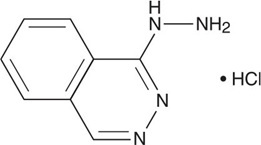
Molecular Formula C 8 H 8 N 4 •HCl
Hydralazine hydrochloride, USP is a white to off-white, odorless crystalline powder. It is soluble in water, practically insoluble in alcohol and in ether. It melts at about 275ºC, with decomposition and has a molecular weight of 196.64.
CLINICAL PHARMACOLOGY
Although the precise mechanism of action of hydralazine is not fully understood, the major effects are on the cardiovascular system. Hydralazine apparently lowers blood pressure by exerting a peripheral vasodilating effect through a direct relaxation of vascular smooth muscle. Hydralazine, by altering cellular calcium metabolism, interferes with the calcium movements within the vascular smooth muscle that are responsible for initiating or maintaining the contractile state.
The peripheral vasodilating effect of hydralazine results in decreased arterial blood pressure (diastolic more than systolic), decreased peripheral vascular resistance, and an increased heart rate, stroke volume, and cardiac output. The preferential dilatation of arterioles, as compared to veins, minimizes postural hypotension and promotes the increase in cardiac output. Hydralazine usually increases renin activity in plasma, presumably as a result of increased secretion of renin by the renal juxtaglomerular cells in response to reflex sympathetic discharge. This increase in renin activity leads to the production of angiotensin II, which then causes stimulation of aldosterone and consequent sodium reabsorption. Hydralazine also maintains or increases renal and cerebral blood flow.
The average maximal decrease in blood pressure usually occurs 10 to 80 minutes after administration of hydralazine hydrochloride injection. No other pharmacokinetic data on hydralazine hydrochloride injection are available.
HOW SUPPLIED
Hydralazine Hydrochloride Injection USP, 20 mg/mL is clear, colorless solution and is supplied as follows;
20 mg per mL 1 mL Single-Dose Vials
Packaged in a Carton of 25 NDC 55150-400-25
Storage: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Discard unused portion.
The vial stopper is not made with natural rubber latex.
Distributed by: Eugia US LLC 279 Princeton-Hightstown Rd. E. Windsor, NJ 08520 Manufactured by: Eugia Pharma Specialities Limited Hyderabad – 500032 India Revised: January 2023