Get your patient on Hydrochlorothiazide - Hydrochlorothiazide tablet (Hydrochlorothiazide)
Hydrochlorothiazide - Hydrochlorothiazide tablet prescribing information
INDICATIONS AND USAGE
Hydrochlorothiazide tablets USP are indicated as adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
Hydrochlorothiazide tablets USP have also been found useful in edema due to various forms of renal dysfunction such as nephrotic syndrome, acute glomerulonephritis, and chronic renal failure.
Hydrochlorothiazide tablets USP are indicated in the management of hypertension either as the sole therapeutic agent or to enhance the effectiveness of other antihypertensive drugs in the more severe forms of hypertension.
Use in pregnancy Routine use of diuretics during normal pregnancy is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy and there is no satisfactory evidence that they are useful in the treatment of toxemia.
Edema during pregnancy may arise from pathologic causes or from the physiologic and mechanical consequences of pregnancy. Thiazides are indicated in pregnancy when edema is due to pathologic causes, just as they are in the absence of pregnancy (see PRECAUTIONS, Pregnancy ). Dependent edema in pregnancy, resulting from restriction of venous return by the gravid uterus, is properly treated through elevation of the lower extremities and use of support stockings. Use of diuretics to lower intravascular volume in this instance is illogical and unnecessary. During normal pregnancy there is hypervolemia which is not harmful to the fetus or the mother in the absence of cardiovascular disease. However, it may be associated with edema, rarely generalized edema. If such edema causes discomfort, increased recumbency will often provide relief. Rarely this edema may cause extreme discomfort which is not relieved by rest. In these instances, a short course of diuretic therapy may provide relief and be appropriate.
DOSAGE AND ADMINISTRATION
Therapy should be individualized according to patient response. Use the smallest dosage necessary to achieve the required response.
CONTRAINDICATIONS
Anuria. Hypersensitivity to this product or to other sulfonamide-derived drugs.
ADVERSE REACTIONS
The following adverse reactions have been reported and, within each category, are listed in order of decreasing severity.
Body as a Whole Weakness.
Cardiovascular Hypotension including orthostatic hypotension (may be aggravated by alcohol, barbiturates, narcotics or antihypertensive drugs).
Digestive Pancreatitis, jaundice (intrahepatic cholestatic jaundice), diarrhea, vomiting, sialadenitis, cramping, constipation, gastric irritation, nausea, anorexia.
Hematologic Aplastic anemia, agranulocytosis, leucopenia, hemolytic anemia, thrombocytopenia.
Hypersensitivity Anaphylactic reactions, necrotizing angiitis (vasculitis and cutaneous vasculitis), respiratory distress including pneumonitis and pulmonary edema, photosensitivity, fever, urticaria, rash, purpura.
Metabolic Electrolyte imbalance (see PRECAUTIONS ), hyperglycemia, glycosuria, hyperuricemia
Musculoskeletal Muscle spasm.
Nervous System/Psychiatric Vertigo, paresthesias, dizziness, headache, restlessness.
Renal Renal failure, renal dysfunction, interstitial nephritis (see WARNINGS )
Skin Erythema multiforme including Stevens-Johnson syndrome, exfoliative dermatitis including toxic epidermal necrolysis, alopecia.
Special Senses Transient blurred vision, xanthopsia.
Urogenital Impotence.
Whenever adverse reactions are moderate or severe, thiazide dosage should be reduced or therapy withdrawn.
Postmarketing Experience Non-melanoma Skin Cancer
Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
CALL YOUR DOCTOR FOR MEDICAL ADVICE ABOUT SIDE EFFECTS. YOU MAY REPORT SIDE EFFECTS TO THE FDA AT 1-800-FDA-1088 OR LEADING PHARMA, LLC AT 1-844-740-7500.
DESCRIPTION
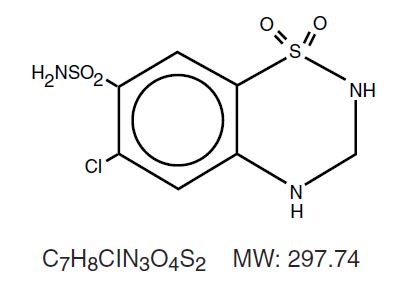
Hydrochlorothiazide USP is a diuretic and antihypertensive. It is the 3,4-dihydro derivative of chlorothiazide. It is chemically designated as 6-chloro-3,4-dihydro-2 H -1,2,4-benzothiadiazine -7-sulfonamide 1,1-dioxide and has the following structural formula:
Hydrochlorothiazide USP is a white, or practically white, crystalline powder which is slightly soluble in water, freely soluble in sodium hydroxide solution, in n-butylamine, and in dimethylformamide; sparingly soluble in methanol, insoluble in ether, in chloroform, and in dilute mineral acids. Each tablet for oral administration contains 12.5 mg, 25 mg and 50 mg of hydrochlorothiazide USP respectively. In addition, each tablet contains the following inactive ingredients: FD&C yellow #6, lactose anhydrous, magnesium stearate, microcrystalline cellulose, sodium starch glycolate, and stearic acid.
CLINICAL PHARMACOLOGY
The mechanism of the antihypertensive effect of thiazides is unknown. Hydrochlorothiazide does not usually affect normal blood pressure.
Hydrochlorothiazide affects the distal renal tubular mechanism of electrolyte reabsorption. At maximal therapeutic dosage all thiazides are approximately equal in their diuretic efficacy.
Hydrochlorothiazide increases excretion of sodium and chloride in approximately equivalent amounts. Natriuresis may be accompanied by some loss of potassium and bicarbonate.
After oral use diuresis begins within 2 hours, peaks in about 4 hours and lasts about 6 to12 hours.
HOW SUPPLIED
Hydrochlorothiazide tablets USP, 12.5 mg are available as peach color, round, flat face, beveled edge tablets, debossed “EP” above and “155” below on one side, and plain on the other side containing 12.5 mg of hydrochlorothiazide USP, packaged in bottles of 100 (NDC 69315-155-01) and 1000 (NDC 69315-155-10) tablets.
Hydrochlorothiazide tablets USP, 25 mg are available as peach color, round, flat face, beveled edge bisected tablets, debossed "EP" above bisect and "131" below bisect on one side and plain on the other side containing 25 mg of hydrochlorothioazide USP, packaged in bottles of 100 (NDC 69315-131-01), 1000 (NDC 69315-131-10) and 5000 (NDC 69315-131-50) tablets.
Hydrochlorothiazide tablets USP, 50 mg are available as peach color, round, flat face, beveled edge, bisected tablets, debossed "EP" above bisect and "130" below bisect on one side and plain on the other side containing 50 mg of hydrochlorothiazide USP, packaged in bottles of 100 (NDC 69315-130-01) and 1000 (NDC 69315-130-10) tablets.
Dispense in a well-closed container as defined in the USP, with a child-resistant closure (as required).
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured by: Leading Pharma, LLC Fairfield, NJ 07004
Rev.06 04/24