Get your patient on Hydroxyzine Hydrochloride
Hydroxyzine Hydrochloride prescribing information
INDICATIONS AND USAGE
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested.
Useful in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses, and in histamine-mediated pruritus.
As a sedative when used as premedication and following general anesthesia, hydroxyzine may potentiate meperidine and barbiturates, so their use in pre-anesthetic adjunctive therapy should be modified on an individual basis. Atropine and other belladonna alkaloids are not affected by the drug. Hydroxyzine is not known to interfere with the action of digitalis in any way and it may be used concurrently with this agent.
The effectiveness of hydroxyzine as an antianxiety agent for long term use, that is more than 4 months, has not been assessed by systematic clinical studies. The physician should reassess periodically the usefulness of the drug for the individual patient.
DOSAGE AND ADMINISTRATION
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested: in adults, 50 mg to 100 mg four times daily; children under 6 years, 50 mg daily in divided doses and children over 6 years, 50 mg to 100 mg daily in divided doses.
For use in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses, and in histamine-mediated pruritus: in adults, 25 mg three times to four times daily; children under 6 years, 50 mg daily in divided doses and children over 6 years, 50 mg to 100 mg daily in divided doses.
As a sedative when used as a premedication and following general anesthesia: 50 mg to 100 mg in adults, and 0.6 mg/kg in children.
When treatment is initiated by the intramuscular route of administration, subsequent doses may be administered orally.
As with all medications, the dosage should be adjusted according to the patient’s response to therapy.
CONTRAINDICATIONS
Oral hydroxyzine hydrochloride is contraindicated in patients with known hypersensitivity to hydroxyzine hydrochloride products, and in patients with known hypersensitivity to cetirizine hydrochloride or levocetirizine hydrochloride.
Hydroxyzine is contraindicated in patients with a prolonged QT interval.
Hydroxyzine, when administered to the pregnant mouse, rat and rabbit, induced fetal abnormalities in the rat and mouse at doses substantially above the human therapeutic range. Clinical data in human beings are inadequate to establish safety in early pregnancy. Until such data are available, hydroxyzine is contraindicated in early pregnancy.
Hydroxyzine is contraindicated for patients who have shown a previous hypersensitivity to it.
ADVERSE REACTIONS
Side effects reported with the administration of hydroxyzine hydrochloride are usually mild and transitory in nature.
Skin and appendages: Oral hydroxyzine hydrochloride is associated with Acute Generalized Exanthematous Pustulosis (AGEP), fixed drug eruptions, pruritis, rash and urticaria in post marketing reports.
Anticholinergic: Dry mouth.
Central Nervous System: Drowsiness is usually transitory and may disappear in a few days of continued therapy or upon reduction of the dose. Involuntary motor activity including rare instances of tremor and convulsions have been reported, usually with doses considerably higher than those recommended. Clinically significant respiratory depression has not been reported at recommended doses.
Cardiac System: QT prolongation, Torsade de Pointes.
To report SUSPECTED ADVERSE REACTIONS, contact Kesin Pharma at 1-833-537-4679 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Hydroxyzine hydrochloride, USP is designated chemically as (±)-2-[2-[4-( p -Chloro-α-phenylbenzyl)-1-piperazinyl]ethoxy]ethanol dihydrochloride. Its structural formula is as follows:
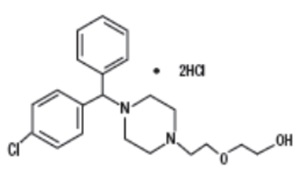
C 21 H 27 ClN 2 O 2 •2HCl M.W. 447.83 g/mol
Inactive ingredients for hydroxyzine hydrochloride oral solution, USP are: flavadew coolmint flavour; sodium benzoate; sucrose; and purified water. Anhydrous citric acid is used for pH adjustment. The pH range is between 2.0 and 4.3.
CLINICAL PHARMACOLOGY
Hydroxyzine hydrochloride is unrelated chemically to the phenothiazines, reserpine, meprobamate, or the benzodiazepines.
Hydroxyzine is not a cortical depressant, but its action may be due to suppression of activity in certain key regions of the subcortical area of the central nervous system.
Primary skeletal muscle relaxation has been demonstrated experimentally. Bronchodilator activity, and antihistaminic and analgesic effects have been demonstrated experimentally and confirmed clinically. An antiemetic effect, both by the apomorphine test and the veriloid test, has been demonstrated. Pharmacological and clinical studies indicate that hydroxyzine in therapeutic dosage does not increase gastric secretion or acidity and in most cases has mild antisecretory activity.
Hydroxyzine is rapidly absorbed from the gastrointestinal tract and its clinical effects are usually noted within 15 minutes to 30 minutes after oral administration.
HOW SUPPLIED
Hydroxyzine hydrochloride oral solution, USP (Syrup) 10 mg per 5 mL (teaspoonful) is a clear colourless, flavadew coolmint flavoured liquid, supplied in the following: NDC 81033-031-05: 5 mL unit-dose cup NDC 81033-031-12: 12.5 mL unit-dose cup NDC 81033-031-44: Carton containing 40 unit-dose cups of 5 mL each NDC 81033-031-45: Carton containing 40 unit-dose cups of 12.5 mL each