Get your patient on Hyrnuo (Sevabertinib)
Hyrnuo prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Patient education
Administration guides
Patient support program
Clinical information
Insurance resources
Prior authorization & coverage support
Dosage & administration
DOSAGE AND ADMINISTRATION
Patient Selection
Select patients for treatment of locally advanced or metastatic non-squamous NSCLC based on the presence of HER2 ( ERBB2 ) TKD activating mutations in tumor specimens [see Clinical Studies (14) ] .
Information on FDA-approved tests is available at http://www.fda.gov/CompanionDiagnostics.
Recommended Dosage
The recommended dosage of HYRNUO is 20 mg orally twice daily with food, until disease progression or unacceptable toxicity [see Clinical Pharmacology (12.3) ] . Swallow tablets whole. Do not cut, crush, or chew tablets.
Missed Dose
If a dose is missed, take the missed dose as soon as you remember prior to the next scheduled dose. Do not take 2 doses at the same time to make up for the missed dose.
Vomited Dose
If a dose is vomited, do not take an additional dose. Resume dosing at the next scheduled time.
Dosage Modifications for Adverse Reactions
The recommended dosage reductions for adverse reactions are provided in Table 1.
| Dose Reduction | Dosage Modification |
|---|---|
| First | 10 mg twice daily |
| Second | 10 mg once daily |
| Permanently discontinue HYRNUO in patients who are unable to tolerate 10 mg once daily. | |
The recommended dosage modifications for adverse reactions are provided in Table 2.
| Adverse Reaction | Severity Grades based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Version 5.0. | Dosage Modification |
|---|---|---|
| Diarrhea [see Warnings and Precautions (5.1) ] | Intolerable Grade 2 or Grade 3 |
|
| Grade 4 |
| |
| Hepatotoxicity [see Warnings and Precautions (5.2) ] | Grade 2, 3 or 4 ALT and/or AST withoutincreased total bilirubin or Grade 3 total bilirubin |
|
| ALT or AST ≥ 3× ULN withtotal bilirubin ≥ 2× ULN or Grade 4 total bilirubin |
| |
| Interstitial lung disease (ILD)/pneumonitis [see Warnings and Precautions (5.3) ] | Any Grade |
|
| Ocular toxicity [see Warnings and Precautions (5.4) ] | Grade 2 |
|
| Grade 3 or Grade 4 |
| |
| Pancreatic Enzyme Elevation [see Warnings and Precautions (5.5) ] | Grade 3 |
|
| Grade 4 |
| |
| Other adverse reactions [see Adverse Reactions (6.1) ] | Intolerable or recurrent Grade 2 or Grade 3 |
|
| Grade 4 | Permanently discontinue HYRNUO. |
Dosage Modifications for Strong CYP3A Inhibitors
Avoid concomitant use of strong CYP3A inhibitors. If concomitant use cannot be avoided, reduce HYRNUO dosage as shown in Table 3. After the CYP3A inhibitor has been discontinued for 3 to 5 elimination half-lives, resume the HYRNUO dosage that was used prior to initiating the inhibitor [see Drug Interactions (7.1) ].
| Current Dosage | Recommended Dosage |
|---|---|
| 20 mg twice daily | 10 mg twice daily |
| 10 mg twice daily | 10 mg once daily |
| 10 mg once daily | Withhold HYRNUO until strong CYP3A inhibitor is discontinued |

Hyrnuo prescribing information
INDICATIONS AND USAGE
HYRNUO is indicated for the treatment of adult patients with locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC) whose tumors have HER2 ( ERBB2 ) tyrosine kinase domain (TKD) activating mutations, as detected by an FDA-approved test [see Dosage and Administration (2.1) ] , and who have received a prior systemic therapy.
This indication is approved under accelerated approval based on objective response rate (ORR) and duration of response (DOR) [see Clinical Studies (14) ] . Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
DOSAGE AND ADMINISTRATION
Patient Selection
Select patients for treatment of locally advanced or metastatic non-squamous NSCLC based on the presence of HER2 ( ERBB2 ) TKD activating mutations in tumor specimens [see Clinical Studies (14) ] .
Information on FDA-approved tests is available at http://www.fda.gov/CompanionDiagnostics.
Recommended Dosage
The recommended dosage of HYRNUO is 20 mg orally twice daily with food, until disease progression or unacceptable toxicity [see Clinical Pharmacology (12.3) ] . Swallow tablets whole. Do not cut, crush, or chew tablets.
Missed Dose
If a dose is missed, take the missed dose as soon as you remember prior to the next scheduled dose. Do not take 2 doses at the same time to make up for the missed dose.
Vomited Dose
If a dose is vomited, do not take an additional dose. Resume dosing at the next scheduled time.
Dosage Modifications for Adverse Reactions
The recommended dosage reductions for adverse reactions are provided in Table 1.
| Dose Reduction | Dosage Modification |
|---|---|
| First | 10 mg twice daily |
| Second | 10 mg once daily |
| Permanently discontinue HYRNUO in patients who are unable to tolerate 10 mg once daily. | |
The recommended dosage modifications for adverse reactions are provided in Table 2.
| Adverse Reaction | Severity Grades based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Version 5.0. | Dosage Modification |
|---|---|---|
| Diarrhea [see Warnings and Precautions (5.1) ] | Intolerable Grade 2 or Grade 3 |
|
| Grade 4 |
| |
| Hepatotoxicity [see Warnings and Precautions (5.2) ] | Grade 2, 3 or 4 ALT and/or AST withoutincreased total bilirubin or Grade 3 total bilirubin |
|
| ALT or AST ≥ 3× ULN withtotal bilirubin ≥ 2× ULN or Grade 4 total bilirubin |
| |
| Interstitial lung disease (ILD)/pneumonitis [see Warnings and Precautions (5.3) ] | Any Grade |
|
| Ocular toxicity [see Warnings and Precautions (5.4) ] | Grade 2 |
|
| Grade 3 or Grade 4 |
| |
| Pancreatic Enzyme Elevation [see Warnings and Precautions (5.5) ] | Grade 3 |
|
| Grade 4 |
| |
| Other adverse reactions [see Adverse Reactions (6.1) ] | Intolerable or recurrent Grade 2 or Grade 3 |
|
| Grade 4 | Permanently discontinue HYRNUO. |
Dosage Modifications for Strong CYP3A Inhibitors
Avoid concomitant use of strong CYP3A inhibitors. If concomitant use cannot be avoided, reduce HYRNUO dosage as shown in Table 3. After the CYP3A inhibitor has been discontinued for 3 to 5 elimination half-lives, resume the HYRNUO dosage that was used prior to initiating the inhibitor [see Drug Interactions (7.1) ].
| Current Dosage | Recommended Dosage |
|---|---|
| 20 mg twice daily | 10 mg twice daily |
| 10 mg twice daily | 10 mg once daily |
| 10 mg once daily | Withhold HYRNUO until strong CYP3A inhibitor is discontinued |
DOSAGE FORMS AND STRENGTHS
Tablets: red brown film-coated, round, biconvex tablets debossed with "SE" on one side and "10" on the other side containing 10 mg of sevabertinib.
USE IN SPECIFIC POPULATIONS
- Lactation : Advise not to breastfeed. (8.2 )
Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action [see Clinical Pharmacology (12.1) ] , HYRNUO can cause fetal harm when administered to a pregnant woman. There are no available data on the use of HYRNUO in pregnant women to inform a drug-associated risk. In embryo-fetal development studies, oral administration of sevabertinib to pregnant rats during the period of organogenesis resulted in alterations to growth at maternal exposures ≥0.18 times the human exposure based on area under the curve (AUC) at the clinical dose of 20 mg twice daily. Animal studies with disrupted or depleted HER2/EGFR and in vitro assays have demonstrated that inhibition of HER2 and/or EGFR results in structural abnormalities, alteration to growth, and embryo-fetal and infant mortality (see Data ) . Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In embryo-fetal development studies, sevabertinib was administered orally to pregnant rats during the period of organogenesis from gestation day 6 to 17 at doses ranging from 1.5 to 11 mg/kg/day. Sevabertinib treatment resulted in maternal toxicity (reduced body weight and body weight gain) and a reduction in fetal weights at ≥6 mg/kg/day (≥0.18 times the human exposure based on AUC at the clinical dose).
Additional Nonclinical Data
A literature-based assessment of the effects on reproduction in mouse models with disrupted or depleted HER2 / EGFR demonstrated that HER2/EGFR is critically important in reproductive and developmental processes including blastocyst implantation, placental development, and embryo-fetal/postnatal survival and development.
In a human-induced pluripotent stem cell-based assay, sevabertinib reduced cardiomyocyte and hepatocyte differentiation markers.
Lactation
Risk Summary
There are no data on the presence of sevabertinib or its metabolites in human milk or their effects on a breastfed child or on milk production. In rats, sevabertinib or its metabolites are excreted in milk (see Data ) . Because of the potential for serious adverse reactions in breastfed children from HYRNUO, advise women not to breastfeed during treatment with HYRNUO and for 1 week after the last dose.
Data
Animal Data
Following administration of radiolabeled sevabertinib to lactating rats, sevabertinib or its metabolites were excreted in milk. Sevabertinib-derived radioactivity concentrations were 13- to 26-times higher in milk than in plasma. Approximately 1.3% of the administered dose of sevabertinib-derived radioactivity was excreted into the milk.
Females and Males of Reproductive Potential
HYRNUO can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1) ] .
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to initiating HYRNUO.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with HYRNUO and for 1 week after the last dose.
Males
Advise males with female partners of reproductive potential to use effective contraception during treatment with HYRNUO and for 1 week after the last dose.
Pediatric Use
The safety and effectiveness of HYRNUO have not been established in pediatric patients.
Geriatric Use
Of the 268 patients with locally advanced or metastatic NSCLC harboring HER2 activating mutations who received HYRNUO at 20 mg twice daily in the SOHO-01study, 43% were 65 years and over and 13% were 75 years and over. No overall differences in effectiveness were observed between these older and younger patients. Grade 3 diarrhea was observed in 23% of patients age ≥75 years and 14% of patients <75 years old.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Diarrhea : At the first sign of diarrhea or increased bowel movement frequency, instruct patients to start an antidiarrheal treatment, and to increase their fluid and electrolyte intake. Interrupt, reduce the dose, or permanently discontinue HYRNUO based on severity. (5.1 )
- Hepatotoxicity : Monitor liver function tests including ALT, AST, and total bilirubin at baseline prior to administration of HYRNUO, every 2 weeks during the first month, and then monthly thereafter as clinically indicated, with more frequent testing in patients who develop transaminase elevations. Interrupt, reduce the dose, or permanently discontinue HYRNUO based on severity. (5.2 )
- Interstitial Lung Disease (ILD)/Pneumonitis : Monitor patients for new or worsening symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, fever). Discontinue HYRNUO upon confirmation of ILD/pneumonitis. (5.3 )
- Ocular Toxicity : Promptly refer patients presenting with new or worsening eye symptoms to an ophthalmologist. Interrupt, reduce the dose or permanently discontinue HYRNUO based on severity. (5.4 )
- Pancreatic Enzyme Elevation : Monitor amylase and lipase regularly during treatment. Interrupt, reduce the dose, or permanently discontinue HYRNUO based on severity. (5.5 )
- Embryo-fetal toxicity : Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. (5.6 , 8.1 , 8.3 )
Diarrhea
HYRNUO can cause severe diarrhea that can lead to dehydration and electrolyte imbalances. In the pooled safety population [see Adverse Reactions (6.1) ] , diarrhea was reported in 86% of patients who received HYRNUO including Grade 3 in 15%. The median time to first onset of any grade diarrhea was four days. Dosage interruptions occurred in 15% of patients, and dose reductions occurred in 12% of patients.
At the first sign of diarrhea or increased bowel movement frequency, instruct patients to start an antidiarrheal treatment (e.g., loperamide [refer to full Prescribing Information] ), and to increase their fluid and electrolyte intake. Interrupt, reduce the dose, or permanently discontinue HYRNUO based on severity [see Dosage and Administration (2.3) ] .
Hepatotoxicity
HYRNUO can cause severe hepatotoxicity characterized by elevations of liver function tests. In the pooled safety population [see Adverse Reactions (6.1) ], based on adverse reaction data, hepatotoxicity occurred in 24% of patients treated with HYRNUO including 3% Grade 3. Based on laboratory data, 35% of patients treated with HYRNUO experienced increased alanine aminotransferase (ALT), including 2.3% Grade 3. Increased aspartate aminotransferase (AST) occurred in 35% of patients treated with HYRNUO, including 2.3% Grade 3. Increased bilirubin occurred in 12% of patients treated with HYRNUO. The median time to first onset of AST or ALT elevation was 1.4 (range 0.2 to 14.5) months. HYRNUO was interrupted for an adverse reaction of hepatotoxicity in 4.1% of patients, the dose was reduced in 4.1% and permanently discontinued in 0.4%.
Monitor liver function tests including ALT, AST, and total bilirubin at baseline prior to the first administration of HYRNUO, every 2 weeks for the first month, and then monthly thereafter as clinically indicated, with more frequent testing in patients who develop transaminase elevations. Interrupt, reduce the dose, or permanently discontinue HYRNUO based on the severity of the adverse reaction [see Dosage and Administration (2.3) ] .
Interstitial Lung Disease/Pneumonitis
HYRNUO can cause severe interstitial lung disease (ILD)/pneumonitis. In the pooled safety population [see Adverse Reactions (6.1) ], ILD/pneumonitis occurred in two patients (0.7%) treated with HYRNUO, including 0.4% Grade 3. One patient required interruption of HYRNUO.
Monitor patients for new or worsening symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, fever). Discontinue HYRNUO upon confirmation of ILD/pneumonitis [see Dosage and Administration (2.3) ] .
Ocular Toxicity
HYRNUO can cause ocular toxicity. In the pooled safety population [see Adverse Reactions (6.1) ], ocular toxicity occurred in 14% of patients treated with HYRNUO, including 11% Grade 1, 2.6% Grade 2 and 0.4% Grade 3 (one case of corneal epithelial microcysts with temporary unilateral blindness).
Promptly refer patients presenting with new or worsening eye symptoms to an ophthalmologist. Interrupt, reduce the dose or permanently discontinue HYRNUO based on severity [see Dosage and Administration (2.3) ].
Pancreatic Enzyme Elevation
HYRNUO can cause elevations of amylase and lipase levels . In the pooled safety population [see Adverse Reactions (6.1) ] , based on laboratory data, increased amylase occurred in 32% of patients treated with HYRNUO, including 3.2% Grade 3 or 4. Increased lipase elevation occurred in 40% of patients treated with HYRNUO, including 10% Grade 3 or 4. Two patients (0.7%) required interruption of HYRNUO due to increased lipase and 3 (1.1%) required interruption of HYRNUO due to increased amylase. The median time to onset of increased amylase/lipase was 1.4 months (range 0.2 to 17 months).
Monitor amylase and lipase regularly during treatment with HYRNUO. Interrupt, reduce the dose, or permanently discontinue HYRNUO based on severity [see Dosage and Administration (2.3) ].
Embryo-fetal toxicity
Based on findings from animal studies and its mechanism of action, HYRNUO can cause fetal harm when administered to a pregnant woman. In embryo-fetal development studies, oral administration of sevabertinib to pregnant rats during the period of organogenesis resulted in alterations to growth at maternal exposures ≥0.18 times the human exposure based on area under the curve (AUC) at the clinical dose of 20 mg twice daily. Animal studies with disrupted or depleted HER2/EGFR and in vitro assays have demonstrated that inhibition of HER2 and/or EGFR results in structural abnormalities, alteration to growth, and embryo-fetal and infant mortality.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with HYRNUO and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with HYRNUO and for 1 week after the last dose [see Use in Specific Populations (8.1 , 8.3) ] .
ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Diarrhea [see Warnings and Precautions (5.1) ]
- Hepatotoxicity [see Warnings and Precautions (5.2) ]
- Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.3) ]
- Ocular Toxicity [see Warnings and Precautions (5.4) ]
- Pancreatic Enzyme Elevation [see Warnings and Precautions (5.5) ]
Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population in the WARNINGS AND PRECAUTIONS reflects exposure to HYRNUO at 20 mg orally twice daily in 268 patients with locally advanced or metastatic NSCLC harboring HER2 and/or other mutations from the SOHO-01 study [see Clinical Studies (14) ] . Among 268 patients who received HYRNUO, 35% were exposed for greater than 6 months and 12% were exposed for greater than 1 year. In this pooled safety population, the most common (>20%) adverse reactions were diarrhea, rash, stomatitis, and paronychia. The most common (≥2%) Grade 3 or 4 laboratory abnormalities were decreased potassium, increased lipase, decreased lymphocyte count, decreased sodium, increased amylase, increased ALT, and increased AST.
The safety of HYRNUO at 20 mg orally twice daily was evaluated in 136 patients with locally advanced or metastatic NSCLC harboring HER2 activating mutations who had received prior systemic therapy in the SOHO-01 study [see Clinical Studies (14) ] . Among 136 patients who received HYRNUO, 46% were exposed greater than 6 months and 15% were exposed for greater than 1 year. The median age of patients who received HYRNUO was 62 years (range: 29 to 91); 63% female; 65% Asian, 27% White, 3.7% Black or African American; and 2.2% were of Hispanic or Latino ethnicity.
The most common adverse reactions (>20%) in patients who received HYRNUO were diarrhea, rash, paronychia, stomatitis, and nausea. The most common Grade 3 or 4 laboratory abnormalities (≥2%) were potassium decreased, lipase increased, lymphocyte count decreased, sodium decreased, amylase increased, aspartate aminotransferase (AST) increased, and alanine aminotransferase (ALT) increased.
Serious adverse reactions occurred in 31% of patients who received HYRNUO. Serious adverse reactions in ≥2% of patients were diarrhea (6%), pneumonia (3.7%), dyspnea (2.2%), and pleural effusion (2.2%).
Permanent discontinuation of HYRNUO due to an adverse reaction occurred in 3.7% of patients. Adverse reactions which resulted in permanent discontinuation were corneal epithelial microcysts, hepatic function abnormal, electrocardiogram QT prolonged, pain in extremity and dyspnea (0.7%, 1 patient each).
Dosage interruptions of HYRNUO due to an adverse reaction occurred in 46% of patients. Adverse reactions which resulted in dosage interruptions in >3% of patients were diarrhea, hypokalemia, nausea, decreased appetite, and pneumonia.
Dose reductions of HYRNUO due to adverse reactions occurred in 28% of patients. Adverse reactions which resulted in dose reductions in >2% of patients were diarrhea, rash, and hypokalemia.
Table 4 summarizes the adverse reactions in SOHO-01 (Groups D and E).
| Adverse Reaction Graded per NCI CTCAE version 5. | HYRNUO N = 136 | |
|---|---|---|
| All Grades (%) | Grade 3 or 4 All were Grade 3, except for dyspnea (0.7%, Grade 4). (%) | |
| Gastrointestinal disorders | ||
| Diarrhea Includes diarrhea, enterocolitis. | 87 | 18 |
| Stomatitis Includes cheilitis, mouth ulceration, mucosal inflammation, stomatitis. | 29 | 1.5 |
| Nausea | 21 | 1.5 |
| Vomiting | 15 | 2.2 |
| Abdominal pain Includes abdominal pain, abdominal pain upper. | 10 | 0 |
| Skin and subcutaneous tissue disorders | ||
| Rash Includes dermatitis acneiform, eczema, eczema asteatotic, palmar-plantar erythrodysaesthesia syndrome, rash, rash erythematous, rash maculopapular, rash pruritic, rash pustular, skin exfoliation. | 66 | 1.5 |
| Paronychia Includes ingrowing nail, nail disorder, onychoclasis, onycholysis, onychomadesis, paronychia. | 33 | 0 |
| Dry skin Includes dry skin, xeroderma. | 20 | 0 |
| Pruritus | 14 | 1.5 |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 18 | 2.9 |
| Investigations | ||
| Weight decreased | 19 | 0.7 |
| General disorders and administration site conditions | ||
| Fatigue Includes asthenia, fatigue. | 13 | 0.7 |
| Eye disorders | ||
| Ocular toxicity Includes blindness unilateral, cataract, conjunctivitis, conjunctivitis allergic, corneal epithelial microcysts, dry eye, eye discharge, eye pain, lacrimation increased, ocular hyperemia, ocular hypertension, ocular toxicity, vision blurred, visual acuity reduced, visual impairment, xerophthalmia. | 16 | 0.7 |
| Respiratory disorders | ||
| Dyspnea Includes dyspnea, dyspnea exertional. | 10 | 1.5 |
Clinically relevant adverse reactions in <10% of patients who received HYRNUO included edema (8%), cardiac arrhythmia (6%; includes arrhythmia, atrioventricular block complete, electrocardiogram QT prolonged, sinus bradycardia, sinus tachycardia, supraventricular extrasystoles, supraventricular tachycardia, tachycardia) and alopecia (3.7%).
Table 5 summarizes the laboratory abnormalities observed in SOHO-01 (Groups D and E).
| Laboratory Abnormality | HYRNUO N=136 The denominator used to calculate the rate varied from 103 to 135 based on the number of patients with a baseline value and at least one post-treatment value. | |
|---|---|---|
| All Grades (%) Graded per NCI CTCAE version 5 using only numeric values. | Grade 3 or 4 All were Grade 3, except for calcium decreased (0.7%, Grade 4) and amylase increased (1.5%; Grade 4) (%) | |
| Hematology | ||
| Hemoglobin decreased | 47 | 1.5 |
| Lymphocyte count decreased | 32 | 6 |
| White blood cell decreased | 21 | 0.7 |
| Chemistry | ||
| Lipase increased | 48 | 12 |
| Potassium decreased | 45 | 13 |
| Aspartate aminotransferase increased | 41 | 3 |
| Magnesium decreased | 40 | 0 |
| Alanine aminotransferase increased | 37 | 3 |
| Glucose increased Graded per NCI CTCAE version 4.03 using only numeric values. | 36 | 0.7 |
| Albumin decreased | 32 | 1.5 |
| Amylase increased | 31 | 3.8 |
| Calcium decreased | 28 | 1.5 |
| Creatinine increased | 27 | 0 |
| Sodium decreased | 26 | 4.4 |
| Alkaline phosphatase increased | 24 | 0 |
| Triglycerides increased | 22 | 0 |
Laboratory abnormalities in <20% of patients who received HYRNUO include blood bilirubin increased (14%; all were Grades 1 and 2).
DRUG INTERACTIONS
- Strong CYP3A Inhibitors: Avoid concomitant use with strong CYP3A inhibitors. If concomitant use cannot be avoided, reduce HYRNUO dosage. (2.4 , 7.1 ).
- Moderate CYP3A Inhibitors: Monitor patients for increased HYRNUO-associated adverse reactions (2.3 , 7.1 )
- Strong and Moderate CYP3A Inducers: Avoid concomitant use with strong or moderate CYP3A inducers. (7.1 )
- Certain CYP3A Substrates: Avoid concomitant use with CYP3A substrates where minimal increases in concentration may lead to serious adverse reactions unless otherwise recommended in the Prescribing Information of the CYP3A substrate. (7.2 )
- Certain P-gp Substrates: Refer to the Prescribing Information for P-gp substrates where minimal increases in concentration may lead to serious adverse reactions (7.2 )
Effect of Other Drugs on HYRNUO
Table 6 describes drug interactions where concomitant use of another drug affects HYRNUO.
| Strong and Moderate CYP3A Inhibitors | |
| Prevention or management | Strong CYP3A Inhibitors :
|
Moderate CYP3A Inhibitors :
| |
| Mechanism and Clinical Effect |
|
| Strong and Moderate CYP3A Inducers | |
| Prevention or management |
|
| Mechanism and Clinical Effect |
|
Effects of HYRNUO on Other Drugs
Table 7 describes drug interactions where concomitant use of HYRNUO affects another drug.
| Certain CYP3A Substrates | |
| Prevention or management |
|
| Mechanism and Clinical Effect |
|
| Certain P-gp Substrates | |
| Prevention or management |
|
| Mechanism and Clinical Effect |
|
| CYP1A1 Substrates | |
| Prevention or management |
|
| Mechanism and Clinical Impact |
|
DESCRIPTION
HYRNUO tablets contain sevabertinib, a kinase inhibitor. The chemical name of the drug substance is 3-(3-chloro-2-methoxyanilino)-2-{3-[(2S)-1,4-dioxan-2-ylmethoxy]pyridin-4-yl}-1,5,6,7-tetrahydro-4H-pyrrolo[3,2-c]pyridin-4-one hydrate. The molecular formula is C 24 H 25 ClN 4 O 5 (anhydrate) and the molecular weight is 484.93 g/mol (anhydrate).
The structural formula is shown below:
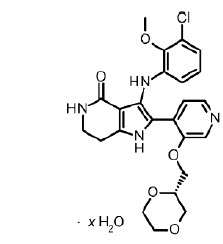
Sevabertinib is present as a non-stoichiometric hydrate as a white to off-white to yellow to pinkish powder. It is slightly soluble in aqueous solution at pH 2, and practically insoluble in aqueous solutions at pH 4.5 and above.
The strength of HYRNUO is based on the anhydrate form. Each HYRNUO tablet for oral use contains 10 mg of sevabertinib. The inactive ingredients are: cellulose microcrystalline, crospovidone, lactose monohydrate, and magnesium stearate. The tablet film coating contains ferric oxide red, hypromellose 5 cP, and macrogol 3350.
CLINICAL PHARMACOLOGY
Mechanism of Action
Sevabertinib is a reversible kinase inhibitor of human epidermal growth factor receptor 2 (HER2). It also exhibits activity against epidermal growth factor receptor (EGFR).
In vitro, sevabertinib inhibited the phosphorylation of HER2 and downstream signaling in cancer cells with HER2 alterations and proliferation of cancer cells overexpressing wild-type HER2 or harboring HER2 mutations.
In vivo, sevabertinib demonstrated antitumor activity in subcutaneous mouse xenograft models derived from human NSCLC tumors harboring an activating HER2 exon 20 mutation.
Pharmacodynamics
Exposure-Response Relationship
Higher sevabertinib exposure, across the dose range of 10 to 80 mg total daily dose (0.25 to 2 times the recommended dosage), was associated with an increased incidence of diarrhea (all grade and Grade ≥3) and rash.
Cardiac Electrophysiology
At 2 times the maximum recommended dose, a mean increase in the QTc interval >20 ms was not observed.
Pharmacokinetics
Sevabertinib pharmacokinetics were observed at steady state in patients with advanced NSCLC harboring activating HER2 or EGFR mutations at the approved recommended dosage and are presented as mean (CV%), unless otherwise specified.
Sevabertinib maximum concentration (C max ) is 902 (45%) ng/mL and total systemic exposure (AUC) is 6,640 (50%) ng•h/mL. Sevabertinib C max and AUC increase in a dose-proportional manner across the dose range of 10 mg to 80 mg (0.25 to 2 times the approved recommended total daily dose). Sevabertinib accumulation is approximately 1.7-fold for AUC and 1.3-fold for C max at the approved recommended dosage. Steady state is achieved within 3 days.
Absorption
Sevabertinib median (min, max) time to maximum concentrations (T max ) is approximately 2 hours (0.5, 8.2 hours) after a single dose.
Effect of Food
Sevabertinib C max decreases by 56% and AUC decreases by 28% with a high-fat meal (1000 calories, 50% fat) in healthy subjects. No clinically significant differences in sevabertinib pharmacokinetics were observed following administration of a low-fat meal (400 calories, 25% fat).
Distribution
Sevabertinib apparent volume of distribution is 28 L (42%). Sevabertinib plasma protein binding is 95%. The blood-to-plasma concentration ratio is 0.6.
Elimination
Sevabertinib effective half-life is approximately 8 hours (33%) with an apparent clearance of 3.1 L/hour (38%).
Metabolism
Sevabertinib is primarily metabolized by CYP3A (major), CYP1A1 (minor), and glucuronidation (minor).
Excretion
After a single oral dose of radiolabeled sevabertinib 40 mg to healthy subjects, approximately 84% of the dose was recovered in feces (14% unchanged) and approximately 10% in urine (1.3% unchanged).
Specific Populations
No clinically significant effects in the pharmacokinetics of sevabertinib were observed based on age (18 to 91 years), race (27% White, 65% Asian, 2.7% Black/African American), sex, body weight (29 to 155 kg), smoking status, eGFR 30 to < 90 mL/min, or mild hepatic impairment (AST > ULN and total bilirubin ≤ ULN; or total bilirubin >1 to 1.5× ULN and any AST). The effect of severe renal impairment (eGFR 15 to <30 mL/min), end-stage renal disease (eGFR <15 mL/min), moderate hepatic impairment (total bilirubin >1.5 to 3× ULN and any AST) or severe hepatic impairment (total bilirubin > 3× ULN and any AST) on sevabertinib pharmacokinetics is unknown.
Drug Interaction Studies
Clinical Studies
Strong CYP3A Inhibitors:Sevabertinib AUC increased 2.3-fold and C max 1.6-fold following concomitant use of itraconazole (strong CYP3A inhibitor) 200 mg once daily.
Strong CYP3A Inducers:Sevabertinib AUC decreased by 79% and C max by 57% following concomitant use of carbamazepine (strong CYP3A inducer) 600 mg once daily.
CYP3A Substrates:Midazolam (CYP3A substrate) AUC increased 2-fold and C max 1.8-fold following concomitant use of HYRNUO 20 mg twice daily.
P-gp Substrates:Dabigatran etexilate (P-gp substrate) AUC increased 1.4-fold following concomitant use of HYRNUO 20 mg twice daily.
BCRP Substrates:Rosuvastatin (BCRP substrate) AUC increased 1.3-fold and C max 1.4-fold following concomitant use of HYRNUO 20 mg twice daily.
Other Drugs:No clinically significant differences in sevabertinib pharmacokinetics were observed when used concomitantly with esomeprazole (proton pump inhibitor).
In Vitro studies
CYP450 Enzymes:Sevabertinib inhibits CYP1A1 and CYP2C8 but does not inhibit CYP2A6, CYP2C9, CYP1A2, CYP2B6, CYP2D6, CYP2C19, or CYP2E1. Sevabertinib does not induce CYP1A2, CYP2B6, or CYP2C19.
Transporter Systems:Sevabertinib is a substrate of P-gp, and BCRP. Sevabertinib inhibits MATE1 and MATE2-K but does not inhibit OATP1B1, OATP1B3, MRP2, OAT1, OAT3, OCT1, or OCT2.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies have not been conducted with sevabertinib.
Mutagenesis
Sevabertinib was not genotoxic in a bacterial reverse mutation (Ames) and an in vitro micronucleus assay, or an in vivo micronucleus assay in rats.
Impairment of Fertility
Fertility studies have not been conducted with sevabertinib.
CLINICAL STUDIES
Locally Advanced or Metastatic Non-squamous NSCLC with HER2 TKD Activating Mutations
The efficacy of HYRNUO was evaluated in SOHO-01 (NCT05099172), an open-label, single-arm, multicenter, multi-cohort clinical study. Eligible patients (Groups D and E) were required to have previously treated locally advanced or metastatic NSCLC with HER2 ( ERBB2 ) activating mutations and have an Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 or 1. HER2 ( ERBB2 ) activating mutations were determined in tumor tissue or plasma by local laboratories prior to enrollment. Patients with treated, stable and asymptomatic brain metastases were eligible. Patients with symptomatic CNS metastases, clinically significant cardiac disease, and history of steroid dependent interstitial lung disease (ILD)/pneumonitis were excluded.
Patients received HYRNUO 20 mg orally twice daily until disease progression or unacceptable toxicity. The major efficacy outcomes were confirmed objective response rate (ORR) and duration of response (DOR), as assessed by Blinded Independent Central Review (BICR) using RECIST v1.1.
The efficacy population included 70 patients from Group D, and 52 patients from Group E, with advanced non-squamous NSCLC with HER2 ( ERBB2 ) tyrosine kinase domain (TKD) activating mutations based on prospective local testing. Of the 122 patients in these combined cohorts, tumor tissue samples from 67.2% (82/122) of patients were retrospectively tested using Oncomine™ Dx Target Test (Life Technologies Corporation). While 92.7% (76/82) of samples were positive for HER2 ( ERBB2 ) TKD activating mutations, 7.3% (6/82) were unevaluable, and there were no samples with negative status for HER2 ( ERBB2 ) TKD activating mutations.
NSCLC Previously Treated, Naïve to HER2-Targeted Therapy: Group D
Efficacy was evaluated in 70 patients with locally advanced or metastatic non-squamous NSCLC with HER2 ( ERBB2 ) TKD activating mutations who had received prior systemic therapy but were naïve to therapy targeting HER2 mutations. Baseline demographic and disease characteristics of the efficacy population were: median age 59 years (range 29 to 77 years); 67% female; 70% Asian, 23% White, 1.4% Black or African American, 6% race not reported; 2.9% were of Hispanic or Latino ethnicity. Patients had an ECOG performance status of either 0 (39%) or 1 (61%); 69% were never-smokers, 29% were former smokers and 2.9% were current smokers. All patients had adenocarcinoma histology. Ninety-one percent (91%) of patients had stage IV disease and 20% had stable brain metastases. The median number of prior therapies was 1 (range 1 to 8); 94% of patients received prior platinum-based chemotherapy, 71% received prior immunotherapy, and 69% received both in combination. Among the patients, 70% of patients had a Y772_A775dup (YVMA) exon 20 insertion.
Efficacy results for SOHO-01 Group D are presented in Table 8.
| Efficacy Parameter | HYRNUO N=70 |
|---|---|
| CI – Confidence Interval | |
| Objective Response Rate (ORR) , (95% CI) | 71% (59, 82) |
| Complete Response | 2.9% |
| Partial Response | 69% |
| Duration of Response (DOR) Observed proportion of responding patients with duration of response beyond landmark time. | N=50 |
| Median, months (95% CI) Kaplan-Meier estimate. | 9.2 (6.3, 15.0) |
| DOR ≥6 months | 54% |
| DOR ≥12 months | 18% |
NSCLC Previously Treated, Including Prior HER2 Targeted Antibody Drug Conjugates (ADCs): Group E
Efficacy was evaluated in 52 patients with locally advanced or metastatic non-squamous NSCLC with HER2 ( ERBB2 ) TKD activating mutations who had received prior systemic therapy including HER2-targeted ADCs.
Baseline demographic and disease characteristics of this efficacy population were: median age 65 years (range 35 to 91 years); 67% female; 62% Asian, 27% White, 6% Black or African American, 6% race not reported; 1.9% were of Hispanic or Latino ethnicity. Patients had an ECOG performance status of either 0 (29%) or 1 (71%); 65% were never-smokers, and 35% were former smokers. All patients had adenocarcinoma histology. Eighty-five percent (85%) of patients had stage IV disease and 29% had stable brain metastases. The median number of prior therapies was 2 (range 1 to 8), 77% of patients received prior platinum-based chemotherapy, 56% received prior immunotherapy, and 56% received both in combination. Among the patients, 77% of patients had a Y772_A775dup (YVMA) exon 20 insertion.
The ORR was 38% (95% CI 25, 53), with 6% of patients having a complete response and 33% of patients having a partial response. The median DOR was 7 months (95% CI 5.6, NE); ranging from 1+ to 17.2+ months based on the observed DOR. The observed proportion of responding patients with DOR of ≥6 months and ≥12 months was 60% and 10%, respectively.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
HYRNUO 10 mg tablets are supplied as red brown film-coated, round, biconvex tablets debossed with "SE" on one side and "10" on the other side.
HYRNUO tablets are packaged in a HDPE bottle of 120 tablets closed with a child-resistant screw cap.
NDC 50419-397-01
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [ see USP Controlled Room Temperature ].
Mechanism of Action
Sevabertinib is a reversible kinase inhibitor of human epidermal growth factor receptor 2 (HER2). It also exhibits activity against epidermal growth factor receptor (EGFR).
In vitro, sevabertinib inhibited the phosphorylation of HER2 and downstream signaling in cancer cells with HER2 alterations and proliferation of cancer cells overexpressing wild-type HER2 or harboring HER2 mutations.
In vivo, sevabertinib demonstrated antitumor activity in subcutaneous mouse xenograft models derived from human NSCLC tumors harboring an activating HER2 exon 20 mutation.